Abstract
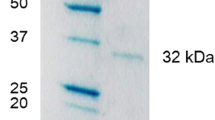
A solution of 10 g/L of sodium alginate (Satialgine® types used [Sanofi trademark]: SG800® and S1100® with manuronic/guluronic ratio of 0.5 and 1.2, respectively) containing invertase (0.08 g of protein/L) was dropped into 0.1 M CaCl2 solution buffered at pH 4.0, 7.0, or 8.0. The beads were left to harden in CaCl2 solution for 24 h. The high immobilization yield of 60% occurred with SG800 at pH8.0. The activity of soluble and insoluble invertase was measured against pH (2.8–8.0), sucrose concentration (4.5–45 mM), and temperature (30–60°C). Both forms presented an optimum pH of 4.6. However, the soluble invertase was stable at the overall pH interval studied, whereas insoluble invertase lost 30% of its original activity at pH > 5.0. At temperatures above 40°C, the insoluble form was more stable than the soluble one. The kinetic constants and activation energies (E a ) for free invertase were K M =41.2 mM, V max=0.10 mg of TRS/(min · mL), and E a 28 kJ/mol for entrapped invertase they were (K M ) ap =7.2 mM, (V max) ap =0.060 mg of TRS/(min · mL), and (E a )ap=24 kJ/mol.
Similar content being viewed by others
References
Crueger, A. (1990), in Microbial Enzymes and Biotechnology, Fogarty, W. M. and Kelly, C. T., eds., Elsevier, London, pp. 179–221.
Uhlig, H. (1990), in Enzymes in Industry, Gerhartz, W., ed., VCH, Weinheim, Germany, pp. 130–141.
Illanes, A. (1994), Biotecnologia de Enzimas, OAS, Washington, DC, pp. 152–153.
Adlercreutz, P. (1993), in Enzymes in Food Processing, Reed, G., Nagodawithana, T., eds., Academic, New York, pp. 103–119.
Vitolo, M. and Barros, D. P. (1992), Lebensm.-Wiss.u.-Technol. 25, 240–243.
Smidsrod, O. and Skjak-Braek, G. (1990), Trends Biotechnol. 8, 71–78.
Barman, E. T. (1969), Enzyme Handbook, Spring-Verlag, New York, p. 584.
Reddy, A. V., MacCall, R., and Maley, F. (1990), Biochemistry 29, 2482–2487.
Vitolo, M. and Carreira, T. C. M. S. (1992), Agro Food Industry Hi-Technol. 3, 24–25.
Somogyi, M. (1952), J. Biol. Chem. 1, 19–23.
Vitolo, M. and Borzani, W. (1983), Anal. Biochem. 130, 469–470.
Bradford, M. (1976), Anal. Biochem. 72, 248–254.
Owusu, R. K., Makhzoum, A., and Knapp, J. S. (1992), Food Chem. 44, 261–268.
Martinsen, A., Skjak-Braek, G., and Smidsrod, O. (1989), Biotechnol. Bioeng. 33, 79–89.
Abdellah, H. A., Abou, B., and Taisser, M. (1992), Food Chem. 43, 369–375.
Mansfeld, J., Schellenberger, A., and Roembach, J. (1992), Biotechnol. Bioeng. 40, 997–1003.
Leuba, J. L., Renken, A., and Flaschel, E. (1983), Eur. Pat. Appl. EP 79,595.
Maxim, S., Flondor, A., and Carpov, A. (1987), Biotechnol. Bioeng. 30, 593–597.
Yoshikuni, M., Tsukamoto, T., Takei, K., and Kojima, K. (1987), Konbushi Ronbunshu 44, 797–802.
Vitolo, M. and Almeida Cunha, B. C. (1984), Biotechnol. Bioeng. 26, 811–813.
Oshima, H., Sakimoto, M., and Harrano, Y. (1980), Biotechnol. Bioeng. 22, 2155–2167.
Reddy, A. V. and Maley, F. (1990), J. Biol. Chem. 265, 10,817–10,821.
De Queiroz, A. A. A., Vitolo, M., Oliveira, R. C., and Higa, Z. O. (1996), Rad. Phys. Chem. 47, 873–880.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arruda, L.M.O., Vitolo, M. Characterization of invertase entrapped into calcium alginate beads. Appl Biochem Biotechnol 81, 23–33 (1999). https://doi.org/10.1385/ABAB:81:1:23
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/ABAB:81:1:23