Abstract
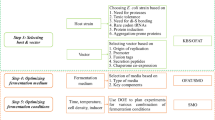
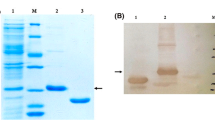
The medium formulation and robust process modeling for anti-HIV peptide (T-20) production by recombinant Escherichia coli overexpression were studied by employing a crossed experimental design. The crossed design, a mixture design combined with process factor (induction duration), was used to find the optimal medium formulation and process time. The optimal settings for three major components, (7.75 mL of NPK sources, 5.5 mL of glucose, and 11.75 mL of MgSO4) characterized by %T-20 (14.45%), the proportion of peptide to the total protein, were observed in a total of 100 mL of medium inducted at an optical density of 0.67 with 0.7 mM isopropyl-β-d-thiogalactopyranoside) for a 3-h induction duration at shake-flask scale. These conditions were further investigated to find robust, process conditions (8.2 mL of NPK sources, 5.6 mL of glucose, and 11.3 mL of MgSO4, and a 3.5-h induction duration time) for T-20 production (13.9%) by applying propagation of error.
Similar content being viewed by others
References
Richman, D. (1998), Nat. Med. 4(11), 1232–1233.
Rimsky, L. T., Shugars, D. C., and Matthews, T. (1998), J. Virol. 72, 986–992.
Wei, X., Ghosh, S. K., Taylor, M. E., et al. (1995), Nature 373, 117–122.
Chan, D. C., Fass, D., Berger, J. M., and Kim, P. S. (1997), Cell 89, 263–273.
Kilby, J. M., Hopkins, S., Venetta, T.M., et al. (1998), Nat. Med., 4(11), 1302–1307.
Rice, W. G., Supko, J. G., Malspeis, L., et al. (1995), Science 270, 1194–1197.
Vanot, G., Valerie, D., Guilhem, M. C., Phan-Tan-Luu, R., Comeau, L. C. (2002), Appl. Microbiol. Biotechnol. 60, 417–419.
www.fuzeon.com (accessed July 7, 2004).
Lee, K.-M. and Gilmore, D. F. (2005), Process, Biochem. 40, 226–249.
Hu, R. (1999), Flood Product Design, Technomic, Lancaster, PA.
Cornell, J. A. (2002) Experiments with Mixtures, 3rd ed., John Wiley & Sons, New York.
Myers, R. H. and Montgomery, D. C. (2002) Response Surface Methodology: Process and Product Optimization Using Designed Experiments, 2nd. ed., John Wiley & Sons, New York.
Montogmery, D. C. (2004), Design and Analysis of Experiments, 6th ed., John Wiley & Sons, New York.
Bollag, D. M., Rozycki, M. D., and Edelstein, S. J. (1996), Protein Methods, 2nd ed., John Wiley & Sons, New York.
Lee, K.-M. and Gilmore, D. F. (2005), Appl. Biochem. Biotechnol. 133, 113–148.
Montgomery, D. C. (1997), Introduction to Statistical Quality Control, 3rd ed., John Wiley & Sons, New York.
Mitra, A. (1997), Fundamentals of Quality Control and Improvement, 2nd ed., Prentice Hall, Upper Saddle River, NJ.
Author information
Authors and Affiliations
Additional information
Coauthors
Rights and permissions
About this article
Cite this article
Lee, KM., Rhee, CH., Kang, CK. et al. Statistical medium formulation and process modeling by mixture design of experiment for peptide overexpression in recombinant Escherichia coli . Appl Biochem Biotechnol 135, 81–100 (2006). https://doi.org/10.1385/ABAB:135:1:81
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/ABAB:135:1:81