Abstract
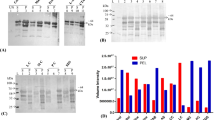
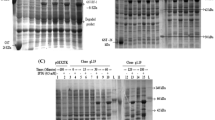
Interferon-α2a (IFN-α2a) has been used for the treatment of various viral infections and cancers for many years. However some untolerable side effects have limited its application in some aspects. To evaluate whether or not an oligopeptide containing GFE motif can home human IFN-α2a to specific tissues, a fusion gene was constructed by fusing the coding sequence of GFE peptide (CGFECVRQCPERC), which was screened from phage display peptide library, to the 3′ end of human IFN-α2a gene by recombinant DNA technique. Fusion protein rhIFN-α2a-GFE was expressed in Escherichia coli as inclusion bodies using a T7 RNA polymerase expression system, pET-22b, refolded through dialysis and purified to homogeneity to >95% of purity by affinity chromatography. Characterization by sodium dodecyl sulfate polyacrylamide gel electrophoresis and immunoblotting demonstrated the authenticity of the fusion protein. Purified rhIFN-α2a-GFE was found to be functionally active in terms of its antiviral activity for about 2.5×108 IU/mg in vitro. Yields of the purified fusion protein were about 200 mg/L of culture medium. Tissue distribution assay in mouse showed that at 30 min IFN-α2a could be enriched sevenfold higher in lung in the targeted IFN group of mice than in the standard IFN group of mice, and last for a long time. At 1 h, IFN-α2a in the targeted IFN group was still 4.02-fold higher than that in the standard group. This confirmed that GFE peptide has the ability to selectively deliver its fusion partner IFN-α2a to lungs. The results also showed that the IFN-α2a-GFE could be specifically enriched in kidney and liver. Its distribution in kidney was concordant with the finding of GFE receptor, MDP, in kidney. However, the IFN-α2a-GFE in liver may imply some significance in pharmacology and toxicology.
Similar content being viewed by others
References
Isaacs, A. and Lindenmann, J. (1957), Proc. R. Soc. London Ser. B 147, 258–267.
Aguet, M., Grobke, M., and Dreiding, P. (1984), Virology 132, 211–216
Meager, A. (2002), J. Immunol. Methods 261, 21–36.
Pfeffer, L. M., Dinarello, C. A., Herberman, R. B., Williams, B. R., Borden, E. C., Bordens, R., Walter, M. R., et al. (1998), Cancer Res. 58, 2489–2499.
Sen, G. C. and Lengyel, P. (1992), J. Biol. Chem. 267, 5017–5020.
Samuel, C. E. (2001), Clin. Microbiol. Rev. 14, 778–809.
Lauer, G. M. and Walker, B. D. (2001), N. Engl. J. Med. 345, 41–52.
Samuel, C.E. (1991), Virology 18, 31–11.
Jonasch, E. and Haluska, F. G. (2001), Oncologist 6, 34–55.
Vilcek, J. and Sen, G. C. (1996), in Fundamental Virology, 3rd ed (Fields, B. N., Knipe, D. M., and Howley, P. M., eds.), Lippincot-Raven, Philadelphia, pp. 341–365.
Motzer, R. J., Bacik, J., Murphy, B. A., Russo, P., and Mazumdar, M. (2002), J. Clin. Oncol. 20, 289–296.
Mahon, F. X., Delbrel, X., Cony-Makhoul, P., Faberes, C., Boiron, J. M., Barthe, C., Bilhou-Nabera, C., et al. (2002), J. Clin. Oncol., 20, 214–220.
Stark, J. J., Dillman, R. O., Schulof, R., Wiemann, M. C., Barth, N. M., Honeycutt, P. J., and Soori, G. (1998), Cancer 82, 1677–1681.
Rajotte, D., Arap, W., Hagedorn, M., Koivunen, E., Pasqualini, R., and Ruoslahti, E. (1998), J. Clin. Invest. 102, 430–437.
Weissmann, C. and Weber, H. (1986), Prog. Nucleic Acid Res. Mol. Biol. 33, 251–300.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., and Randall, R. J. (1951), J. Biol. Chem. 193, 265–275.
Familletti, P. C., Rubinstein, S., and Pestka, S. (1981), Methods Enzymol. 78, 387–394.
Standardization Committee of Department of Health in China. (2000), in Chinese Regulation for Biological Products (Pan, Z. A., Zhang, X. H., and Duan, Z. B., eds.), Chinese Chemical Industry Press, Beijing, China, pp. 373–375.
Rajotte, D. and Ruoslahti, E. (1999), J. Biol. Chem. 274, 11,593–11,598.
Thatcher, D. R. and Panayotatos, N. (1996), Methods Enzymol. 119, 166–177.
Swaminathan, S. and Khanna, N. (1999), Protein Express. Purif. 15, 236–242.
Neves, F. O., Ho, P. L., Raw, I., Pereira, C. A., Moreira, C., and Nascimento, A. L. (2004), Protein Express. Purif. 35, 353–359.
Keynan, S., Hooper, N. M., and Turner, A. J. (1996), in Zinc Metalloproteases in Health and Disease, Hooper, N. M., ed., Taylor & Francis, London, pp. 285–309.
Yamaya, M., Sekizawa, K., Terajima, M., Okinaga, S., Ohrui, T., and Sasaki, H. (1998), Respir. Physiol. 111, 101–109.
Hirota, T., Nishikawa, Y., Tanaka, M., Igarashi, T., and Kitagawa, H. (1986), Eur. J. Biochem. 160, 521–525.
Pytela, R., Pierschbacher, M. D., and Ruoslahti, E. (1985), Cell 40, 191–198.
Habib, G. M., Shi, Z. Z., Cuevas, A. A., Guo, Q., Matzuk, M. M., and Lieberman, M. W. (1998), Proc. Natl. Acad. Sci. USA 95, 4859–4863.
Habib, G. M., Barrios, R., Shi, Z. Z., and Lieberman, M. W. (1996), J. Biol. Chem. 271, 16,273–16,280.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yan, Z., Lu, L., Shi, J. et al. Expression, refolding, and characterization of GFE peptide-fused human interferon-α2a in Escherichia coli . Appl Biochem Biotechnol 133, 149–162 (2006). https://doi.org/10.1385/ABAB:133:2:149
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/ABAB:133:2:149