Abstract
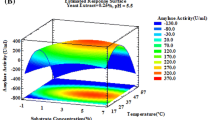
Glucoamylase production by Aspergillus niger in solid-state fermentation was optimized using factorial design and response surface techniques. The variables evaluated were pH and bed thickness in tray, having as response enzyme production and productivity. The bed thickness in tray was the most significant variable for both responses. The highest values for glucoamylase production occurred using pH 4.5 and bed thickness in the inferior limits at 2.0–4.2 cm. For productivity, the optimal conditions were at pH 4.5 as well and bed thickness from 4.4 to 7.5 cm. The optimal conditions for glucoamylase production while obtaining high activity without loss of productivity were pH 4.5 and bed thickness in tray from 4.0 to 4.5 cm, which resulted in an enzyme production of 695 U/g and productivity of 5791 U/h.
Similar content being viewed by others
References
Mase, T., Matsumiya, Y., Mori, S., and Matsuura, A. (1996), J. Ferment. Bioeng. 81, 347–350.
James, J. A. and Lee, B. H. (1997), J. Food Biochem. 21, 1–52.
Hasan, S. D. M., Costa, J. A. V., and Sanzo, A. V. (1998), Biotech. Tech. 12, 787–791.
Bertolin, T. E., Costa, J. A. V., and Pasquali, G. D. L. (2001), J. Microbiol. Biotechnol. 11, 13–16.
Pandey, A. (1991), Proc. Biochem. 26, 355–361.
Pandey, A., Selvakumar, P., and Ashkumary, L. (1996), Proc. Biochem. 31, 43–46.
Adinarayana, K., Prabhakar, T., Srinivasulu, V., Anitha Rao, M., Jhansi Lakshmi, P., and Ellaiah, P. (2003), Proc. Biochem. 39, 171–177.
Singh, H. and Soni, S. K. (2001), Proc. Biochem 31, 453–459.
Ellaiah, P., Adinarayana, K., Bhavani, Y., Padmaja, P., ADN Srinivasulu, B. (2002), Proc. Biochem. 38, 615–620.
Sanzo, A. V. L., Hansan, S. D. M., Costa, J. A. V., and Bertolin, T. E. (2001), Sci. Eng. J. 10, 59–62.
Kalil, S. J., Maugeri, F., and Rodrigues, M. I. (2000), Proc. Biochem. 35, 539–550.
Khuri, A. I. and Cornell, J. A. (1987), Response Surface Design and Analysis, Marcel Dekker, New York.
Miller, G. L. (1959), Anal. Chem. 31, 426–428.
Association of Official Analytical Chemists (AOAC). (2000), Official Methods of Analysis International, 17th ed., CD-ROM, William Horwitz, Washington, DC.
Haaland, P. D. (1989), Experimental Design in Biotechnology, Marcel Dekker, New York.
Burkert, J. F. M., Maugeri, F., and Rodrigues, M. I. (2004), Bioresour. Technol. 91, 77–84.
Balasubramaniem, A. K., Nagarajan, K. V., AND Paramasamy, G. (2001), Proc. Biochem. 36, 1241–1247.
Anto, H., Trivedi, U. B., and Patel, K. C. (2005), Bioresour. Technol., in press.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Silveira, S.T., Oliveira, M.S., Costa, J.A.V. et al. Optimization of glucoamylase production by Aspergillus niger in solid-state fermentation. Appl Biochem Biotechnol 128, 131–139 (2006). https://doi.org/10.1385/ABAB:128:2:131
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/ABAB:128:2:131