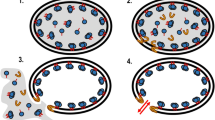
Abstract
Immobilized biocatalysts, including particulate enzymes, represent an attractive tool for research and industrial applications because they combine the specificity of native enzymes with the advantage that they can be readily separated from end product and reused. We demonstrated the use of the Caulobacter crescentus surface (S)-layer protein (RsaA) secretion apparatus for the generation of particulate enzymes. Specifically, a candidate protein made previously by fusion of the β-1,4-glycanase (Cex) from the cellulolytic bacterium Cellulomonas fimi with the C-terminus of RsaA was evaluated. Cex/RsaA cleaved the glycosidic linkage in the artificial substrate p-nitrophenyl-β-d-cellobioside with a K M similar to that of native Cex (1.1 mM for Cex/RsaA vs 0.60 mM for Cex), indicating that the particulate Cex enzyme was able to bind substrate with wild-type affinity. By contrast, the k cat value was significantly reduced (0.08 s−1 for Cex/RsaA vs 15.8 s−1 for Cex)cat, likely owing to the fact that the RsaA C-terminus induces spontaneous unstructured aggregation of the recombinant protein. Here, we demonstrated that not only can an RsaA fusion protein be cheaply produced and purified to a high yield (76 mg/L of dry wt for Cex/RsaA), but it can also be efficiently recycled. The Caulobacter S-layer secretion system therefore offers an attractive new model system for the production of particulate biocatalysts.
Similar content being viewed by others
References
Freeman, A., Abramov, S., and Georgiou, G. (1999), Biotechnol. Bioeng., 62, 155–159.
Strauss, A., and Gotz, F. (1996), Mol. Microbiol. 21, 491–500.
Lee, T. S., Turner, M. K., and Lye, G. J. (2002), Biotechnol. Prog. 18, 43–50.
Hoskin, F. C., Walker, J. E., and Stote, R. (1999), Chem. Biol. Interact 119–120, 439–444.
Haring, D., and Schreier, P. (1999), Curr. Opin. Chem. Biol. 3, 35–38.
Margolin, A. L. (1996), Trends Biotechnol. 14, 223–230.
Ellerby, L. M., Nishida, C. R., Nishida, F., Yamanaka, S. A., Dunn, B., Valentine, J. S., and Zink, J. I. (1992), Science 255, 1113–1115.
Shchipunov, Y. A., Karpenko, T. Y., Bakunina, I. Y., Burtseva, Y. V., and Zvyagintseva, T. N. (2004), J. Biochem. Biophys. Methods 58, 25–38.
Trivedi, B. (1999), Nat. Biotechnol. 17, 339–341.
Spagna, G., Barbagallo, R. N., Casarini, D., and Pifferi, P. G. (2001), Enzyme Microb. Technol. 28, 427–438.
Ichijo, H., Nagasawa, J., and Yamauchi, A. (1990), J. Biotechnol. 14, 169–178.
Pessela, B. C., Mateo, C., Fuentes, M., Vian, A., Garcia, J. L., Carrascosa, A. V., Guisan, J. M., and Fernandez-Lafuente, R. (2004), Biotechnol. Prog. 20, 388–392.
Min, D. J., Andrade, J. D., and Stewart, R. J. (1999), Anal. Biochem. 270, 133–139.
Alvaro, G., Fernandez-Lafuente, R., Blanco, R. M., and Guisan, J. M. (1990), Appl. Biochem. Biotechnol. 26, 181–195.
Cao, R., Gu, Z., Patterson, G. D., and Armitage, B. A. (2004), J. Am. Chem. Soc. 126, 726, 727.
Poindexter, J. S. (1981), Microbiol. Rev. 45, 123–179.
Shapiro, L. (1976), Annu. Rev. Microbiol. 30, 377–407.
Smit, J. (1986), in Bacterial Outer Membranes as Model Systems (Inouye, M., ed.), John Wiley & Sons, New York, pp. 343–376.
Sleytr, U. B. and Messner, P. (1983), Annu. Rev. Microbiol. 37, 311–339.
Sara, M. and Sleytr, U. B. (2000), J. Bacteriol. 182, 859–868.
Sara, M. and Sleytr, U. B. (1996), Prog. Biophys. Mol. Biol. 65, 83–111.
Messner, P. and Sleytr, U. B. (1992), Adv. Microb. Physiol. 33, 213–275.
Smit, J., Engelhardt, H., Volker, S., Smith, S. H., and Baumeister, W. (1992), J. Bacteriol. 174 6527–6538.
Awram, P. and Smit, J. (1998), J. Bacteriol. 180, 3062–3069.
Bingle, W. H., Nomellini, J. F., and Smit, J. (1997), J. Bacteriol. 179, 601–611.
Bingle, W. H., Nomellini, J. F., and Smit, J. (2000), J. Bacteriol. 182, 3298–3301.
Nomellini, J., Toporowski, M. C., and Smit, J. (2004), in Expression Technologies: Current Status and Future Trends (Baneyx, F., ed.), Horizon Scientific Press, Norfolk, UK, pp. 477–524.
Nierman, W. C., Feldblyum, T. V., Laub, M. T., et al. (2001), Proc. Natl. Acad. Sci. USA 98, 4136–4141.
Umelo-Njaka, E., Nomellini, J. F., Bingle, W. H., Glasier, L. G., Irvin, R. T., and Smit, J. (2001), Vaccine 19, 1406–1415.
Umelo-Njaka, E., Bingle, W. H., Borchani, F., Le, K. D., Awram, P., Blake, T., Nomellini, J. F., and Smit, J. (2002), J. Bacteriol. 184, 2709–2718.
Gilkes, N. R., Claeyssens, M., Aebersold, R., Henrissat, B., Meinke, A., Morrison, H. D., Kilburn, D. G., Warren, R. A., and Miller, R. C. Jr. (1991), Eur. J. Biochem. 202, 367–377.
White, A., Withers, S. G., Gilkes, N. R., and Rose, D. R. (1994), Biochemistry 33, 12,546–12,552.
Tull, D., Withers, S. G., Gilkes, N. R., Kilburn, D. G., Warren, R. A., and Aebersold, R. (1991), J. Biol. Chem. 266, 15,621–15,625.
Tull, D., and Withers, S. G. (1994), Biochemistry 33, 6363–6370.
Notenboom, V., Birsan, C., Nitz, M., Rose, D. R., Warren, R. A., and Withers, S. G. (1998), Nat. Struct. Biol. 5, 812–818.
Bingle, W. H., Le, K. D., and Smit, J. (1996), Can. J. Microbiol. 42, 672–684.
Langsford, M. L., Gilkes, N. R., Singh, B., Moser, B., Miller, R. C. Jr., Warren, R. A., and Kilburn, D. G. (1987), FEBS Lett. 225, 163–167.
Kempton, J. B., and Withers, S. G. (1992), Biochemistry 31, 9961–9969.
Leatherbarrow, R. J. (1990), Version 2.0 Ed., Erithacus Software, Staines, UK.
Kirk, O., Borchert, T. V., and Fuglsang, C. C. (2002), Curr. Opin. Biotechnol. 13, 345–351.
van Beilen, J. B., and Li, Z. (2002), Curr. Opin. Biotechnol. 13, 338–344.
Liang, J. F., Li, Y. T., and Yang, V. C. (2000), J. Pharm. Sci. 89, 979–990.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duncan, G., Tarling, C.A., Bingle, W.H. et al. Evaluation of a new system for developing particulate enzymes based on the surface (S)-layer protein (RsaA) of Caulobacter crescentus . Appl Biochem Biotechnol 127, 95–110 (2005). https://doi.org/10.1385/ABAB:127:2:095
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1385/ABAB:127:2:095