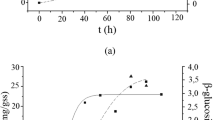
Abstract
The hydrolysis of cellulose to the water-soluble products cellobiose and glucose is achieved via synergistic action of cellulolytic proteins. The three types of enzymes involved in this process are endoglucanases, cellobiohydrolases, and β-glucosidases. One of the best fungal cellulase producers is Trichoderma reesei RUT C30. However, the amount of β-glucosidases secreted by this fungus is insufficient for effective cellulose conversion. We investigated the production of cellulases and β-glucosidases in shake-flask cultures by applying three pH-controlling strategies: (1) the pH of the production medium was adjusted to 5.8 after the addition of seed culture with no additional pH adjustment performed, (2) the pH was adjusted to 6.0 daily, and (3) the pH was maintained at 6.0 by the addition of Tris-maleate buffer to the growth medium. Different carbon sources—Solka Floc 200, glucose, lactose, and sorbitol—were added to standard Mandels nutrients. The lowest β-glucosidase activities were obtained when no pH adjustment was done regardless of the carbon source employed. Somewhat higher levels of β-glucosidase were measured in the culture filtrates when daily pH adjustment was carried out. The effect of buffering the culture medium on β-glucosidase liberation was most prominent when a carbon source inducing the production of other cellulases was applied.
Similar content being viewed by others
References
Goodwin, T. W., and Mercer, E. I. (1983), in Introduction to Plant Biochemistry, Pergamon, Exeter, UK, pp. 55–91.
Marsden, W. L. and Gray, P. P. (1986), CRC Crit. Rev. Biotechnol. 3, 235–276.
Hendy, N. A., Wilke, C. R., and Blanch, H. W. (1984), Enzyme Microb. Technol. 6, 73–77.
Velkovska, S., Marten, M. R., and Ollis, D. F. (1997), J. Biotechnol. 54, 83–94.
Chahal, D. S., McGuire, S., Pikor, H., and Noble, G. (1982), Biomass 2, 127–137.
Wyman, C. E. (1994), Bioresour. Technol. 50, 3–16.
Mandels, M. and Weber, J. (1969), Adv. Chem. Ser. 95, 391–414.
Persson, I., Tjerneld, F., and Hägerdal, B. H. (1991), Process. Biochem. 26, 65–74.
Montenecourt, B. S. and Eveleight, D. E. (1979), Adv. Chem. Ser. 181, 289–301.
Saddler, J. N. (1982), Enzyme Microb. Technol. 4, 414–418.
Wood, T. M. and Campayo, G. V. (1990), Biodegradation 1, 147–161.
Stockton, B. C., Mitchell, D. J., Grohmann, K., and Himmel, M. E. (1991), Biotechnol. Lett. 13, 57–62.
Sternberg, D. (1976), Biotechnol. Bioeng. Symp. 6, 35–53.
Hayward, T. K., Hamilton, J., Tholudur, A., and McMillan, J. D. (2000), Appl. Biochem. Biotechnol. 84–86, 859–874.
Tangnu, K. S., Blanch, H. W., and Charles, R. W. (1981), Biotechnol. Bioeng. 23, 1837–1849.
Juhasz, T., Szengyel, Z., Szijarto, N., and Reczey, K. (2003), Appl. Biochem. Biotechnol. 113–116, 201–211.
Mandels, M. and Weber, J. (1969), Adv. Chem. Ser. 95, 391–414.
Stoscheck, C. M. (1990), Methods Enzymol. 182, 50–69.
Mandels, M., Andreotti, R., and Roche, C. (1976), Biotechnol. Bioeng. Symp. 6, 21–33.
Bailey, M. J. (1981), Biotechnol. Lett. 3, 695–700.
Berghem, L. E. E. and Petterson, L. G. (1976), Eur. J. Biochem. 46, 295–305.
Ghose, T. K. (1987), Pure Appl. Chem. 59, 257–268.
Domingues, F. C., Querioz, J. A., Cabral, J. M. S., and Fonseca, L. P. (2001), Biotechnol. Lett. 23, 771–775.
Morikawa, Y., Ohashi, T., Mantani, O., and Okada, H. (1995), Appl. Microbiol. Biotechnol. 44, 106–111.
Kubicek, C. P. and Penttilä, M. E. (1998), in Trichoderma and Gliocladium, Enzymes, Biological Control and Commercial Applications, vol. 2, Harman, G. E. and Kubicek, C. P., eds., Taylor & Francis, Bristol, UK, pp. 49–67.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Juhász, T., Egyházi, A. & Réczey, K. β-Glucosidase production by Trichoderma reesei . Appl Biochem Biotechnol 121, 243–254 (2005). https://doi.org/10.1385/ABAB:121:1-3:0243
Issue Date:
DOI: https://doi.org/10.1385/ABAB:121:1-3:0243