Abstract
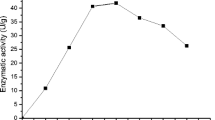
The effect of carbon source and its concentration, inoculum size, yeast extract concentration, nitrogen source, pH of the fermentation medium, and fermentation temperature on β-glucosidase production by Kluyveromyces marxianus in shake-flask culture was investigated. These were the independent variables that directly regulated the specific growth and β-glucosidase production rate. The highest product yield, specific product yield, and productivity of β-glucosidase occurred in the medium (pH 5.5) inoculated with 10% (v/v) inoculum of the culture. Cellobiose (20 g/L) significantly improved β-glucosidase production measured as product yield (Y P/S ) and volumetric productivity (Q P ) followed by sucrose, lactose, and xylose. The highest levels of productivity (144 IU/[L·h]) of β-glucosidase occurred on cellobiose in the presence of CSL at 35°C and are significantly higher than the values reported by other researchers on almost all other organisms. The thermodynamics and kinetics of β-glucosidase production and its deactivation are also reported. The enzyme was substantially stable at 60°C and may find application in some industrial processes.
Similar content being viewed by others
Abbreviations
- h :
-
Planck’s constant
- k B :
-
Boltzmann constant
- q p :
-
specific rate of enzyme production (IU/[g of cells·h])
- q S :
-
specific rate of substrate consumption (g of substrate consumed/[g of cells·h])
- Q IP :
-
rate of intracellular protein formation (mg/[L·h])
- Q P :
-
rate of β-glucosidase formation (IU/[L·h])
- Q S :
-
rate of substrate consumption (g of substrate consumed/[L·h])
- Q X :
-
rate of cell mass formation (g of cells/[L·h])
- R :
-
gas constant
- T :
-
absolute temperature
- Y P/S :
-
β-glucosidase yield (IU/g of substrate utilized)
- Y P/X :
-
specific yield of enzyme production (IU/g of cells)
- Y X/S :
-
cell yield coefficient (g of cells formed/g of substrate utilized)
- ΔH*:
-
enthalpy of activation
- ΔS*:
-
entropy of activation
- μ:
-
specific growth rate (h−1)
References
Woodward, J. (1991), Bioresour. Technol. 36, 67–75.
Rajoka, M. I., Bashir, A., Hussain, M.-R. A., Parvez, S., Ghauri, M. T., and Malik, K. A. (1998), Folia Microbiol. 43, 129–135.
Gadgil, N. J., Daginawala, H. F., Chakarabarti, T., and Khanna, P. (1995), Enzyme Microb. Technol. 17, 942–946.
Suto, M. and Tomita, F. J. (2001), Biosci. Bioeng. 92, 305–311.
Latif, F., Rajoka, M. I., and Malik, K. A. (1994), Bioresour. Technol. 50, 107–112.
Rajoka, M. I., Bashir, A., Hussain, M.-R. A., and Malik, K. A. (1998), Folia Microbiol. 43, 15–22.
Brady, D., Merchant, R., and McHale, A. P. (1994), Biotechnol. Lett. 16, 737–740.
Espinoza, P., Barzana, E., Garcia-Garibay, M., and Gomez-Ruiz, L. (1992), Biotechnol. Lett. 14, 1053–1058.
Belem, M. A. F. and Lee, B. H. (1998), Crit. Rev. Food Sci. Nutr. 38, 565–598.
Barron, N., Merchant, R., and McHale, A. P. (1994), World J. Microbiol. Biotechnol. 16, 625–630.
Banat, N., Merchant, R., and McHale, A. P. (1992), World J. Microbiol. Biotechnol. 16, 259–263.
Rajoka, M. I. and Malik, K. A. (1997), Folia Microbiol. 42, 59–64.
Saha, B. C. and Bothast, R. J. (1996), Appl. Environ. Microbiol. 62, 3165–3170.
Converti, A. and Del Borghi, M. (1997), Enzyme Microb. Technol. 21, 511–517.
Al-Asheh, S. and Duvniak, Z. (1994), Acta Biotechnol. 14, 223–237.
Converti, A. and Dominguez, J. M. (2001), Biotechnol. Bioeng. 75, 39–45
Aiba, S., Humphrey, A. E., and Millis, N. F., eds. (1973), in Biochemical Engineering, 2nd ed., Academic, New York, pp. 92–127.
Arni, S., Molinari, F., Del Borghi, M., and Converti, A. (1996), Starke/Starch 51, 218–224.
Lawford, H. G. and Rousseau, J. D. (1993), Biotechnol. Lett. 15, 615–620.
Pirt, S. J. (1975), Principles of Cell Cultivation, Blackwell Scientific, London.
Furlan, A. S., Schneider, A. S. L., Merckle, R., Carvalho-Johans, M. F., and Jonas, R. (2000), Biotechnol. Lett. 22, 1195–1198.
Inchaurrondo, V. A., Yantorno, O. M., and Voget, C. E. (1994), Process Biochem. 29, 47–53.
Singh, D., Nigam, P., Banat, I. M., Marchant, R., and McHale, A. P. (1998), World J. Microbiol. Biotechnol. 14, 823–834.
Spiridonov, N. A. and Wilson, D. B. (2001), Curr. Microbiol. 42, 295–302.
Skory, C. D., Freer, S. N., and Bothast, R. J. (1996), Curr. Genet. 30, 417–422.
Cummings, C. and Fowler, T. (1996), Curr. Genet. 29, 227–230.
Jäger, S., Brumbauer, A., Fehér, E., Réczey, K., and Kiss, L. (2001), World J. Microbiol. Biotechnol. 17, 455–461.
Gomes, I., Gomes, J., Gomes, D. J., and Steiner, W. (2000), Appl. Microbiol. Biotechnol. 53, 461–468.
Bollok, M. and Reczey, K. (2000), Acta Alimentaria 29, 155–168.
Shaukat, F., Ghauri, M. T., Shahid, R., Parvez, S., and Rajoka, M. I. (2002), Biotechnol. Lett. 24, 1803–1806.
Roels, J. A. (1983), Energetics and Kinetics in Biotechnology, Elsevier Biomedical, Amsterdam, The Netherlands.
Brady, D., Merchant, R., and McHale, A. P. (1995), Biotechnol. Lett. 17, 737–740.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rajoka, M.I., Khan, S., Latif, F. et al. Influence of carbon and nitrogen sources and temperature on hyperproduction of a thermotolerant β-glucosidase from synthetic medium by Kluyveromyces marxianus . Appl Biochem Biotechnol 117, 75–92 (2004). https://doi.org/10.1385/ABAB:117:2:075
Issue Date:
DOI: https://doi.org/10.1385/ABAB:117:2:075