Abstract
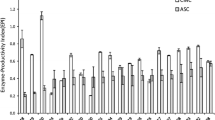
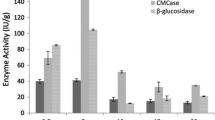
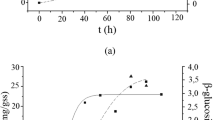
Currently, the high market price of cellulases prohibits commercialization of the lignocellulosics-to-fuel ethanol process, which utilizes enzymes for saccharification of cellulose. For this reason research aimed at understanding and improving cellulase production is still a hot topic in cellulase research. Trichoderma reesei RUT C30 is known to be one of the best hyper producing cellulolytic fungi, which makes it an ideal test organism for research. New findings could be adopted for industrial strains in the hope of improving enzyme yields, which in turn may result in lower market price of cellulases, thus making fuel ethanol more cost competitive with fossil fuels. Being one of the factors affecting the growth and cellulase production of T. reesei, the pH of cultivation is of major interest. In the present work, numerous pH-controlling strategies were compared both in shake-flask cultures and in a fermentor. Application of various buffer systems in shake-flask experiments was also tested. Although application of buffers resulted in slightly lower cellulase activity than that obtained in non-buffered medium, β-glucosidase production was increased greatly.
Similar content being viewed by others
References
Ryu, D. D. Y. and Mandels, M. (1980), Enzyme Microb. Technol. 2, 91–102.
Kanson, A. L., Essam, S. A., and Zeinat, A. N. (1999), Polym. Degrad. Stabil. 63, 273–278.
Mukhopadhyay, S. and Nandi, B. (1999), J. Sci. Ind. Res. 58, 107–111.
Wayman, M. and Chen, S. (1992), Enzyme Microb. Technol. 14, 825–831.
Kadam, K. L. and Keutzer, W. J. (1995), Biotechnol. Lett. 17, 1111–1114.
Tangnu, S. K., Blanch, H. W., and Wilke, C. R. (1981), Biotechnol. Bioeng. 23, 1837–1849.
Hendy, N. A., Wilke, C. R., and Blanch, H. W. (1984), Enzyme Microb. Technol. 6, 73–77.
Doppelbauer, R., Esterbauer, H., Steiner, W., Lafferty, R. M., and Steinmller, H. (1987), Appl. Microbiol. Biotechnol. 26, 485–494.
Mukhopadhyay, S. N. and Malik, R. K. (1980), Biotechnol. Bioeng. 22, 2237–2250.
Mandels, M. and Weber, J. (1969), Adv. Chem. Ser. 95, 391–414.
Sternberg, D. (1976), Biotechnol. Bioeng. Symp. 6(6), 35–53.
Chahal, D. S., McGuire, S., Pikor, H., and Noble, G. (1982), Biomass 2(2), 127–137.
Duff, S. J. B., Cooper, D. G., and Fuller, O. M. (1987), Enzyme Microb. Technol. 9, 47–51.
Yu, X.-B., Hyun S. Y., and Yoon-Mo, K. (1998), J. Microbiol. Biotechnol. 8, 208–213.
Mandels, M., Andreotti, R., and Roche, C. (1976), Biotechnol. Bioeng. Symp. 6(6), 21–33.
Berghem, L. E. E. and Petterson, L. G. (1976), Eur. J. Biochem. 46, 295–305.
Andreotti, R. E., Mandels, M., and Roche, C. (1977), in Bioconversion of Cellulosic Substrates into Energy, Chemicals and Microbial Protein: Proceedings of Bioconversion Symposium, Ghose, T. K., ed., Indian Institute of Technology, Delhi, pp. 249–267.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Juhász, T., Szengyel, Z., Szijártó, N. et al. Effect of pH on cellulase production of Trichoderma ressei RUT C30. Appl Biochem Biotechnol 113, 201–211 (2004). https://doi.org/10.1385/ABAB:113:1-3:201
Issue Date:
DOI: https://doi.org/10.1385/ABAB:113:1-3:201