Abstract
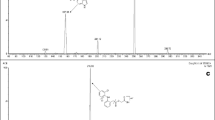
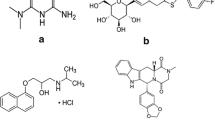
A sensitive and specific liquid chromatography–electrospray ionization-tandem mass spectrometry method has been developed and validated for the identification and quantification of ursolic acid in human plasma using glycyrrhetic acid as an internal standard. The method involves extraction with methyl tert-butyl ether. The analyte was separated on a C18 column and analyzed in multiple reaction monitoring mode with a negative electrospray ionization interface using the [M–H]− ions, m/z 455.4 for ursolic acid and m/z 469.5 → m/z 425.5 for glycyrrhetic acid. The method was validated over the concentration range of 0.86–110.0 μg L−1. The intra- and inter-day precisions were less than 13.53% relative standard deviation (RSD) and the accuracy was within −4.76% in terms of relative error (RE). The lower limit of quantification was 0.86 μg L−1 with acceptable precision and accuracy. There were almost no matrix effects. Recovery of ursolic acid from spiked drug-free plasma was higher than 68%. The method was used to study the pharmacokinetic profile of ursolic acid in human plasma after oral administration of Jieyu capsules.



Similar content being viewed by others
References
Liu J (1995) J Ethnopharmacol 49(2):57–68. doi:10.1016/0378-8741(95)90032-2
Liu J (2005) J Ethnopharmacol 100(1–2):92–94. doi:10.1016/j.jep.2005.05.024
Baricevic D, Sosa S, DellaLoggia R, Tubaro A, Simonovska B, Krasna A (2001) J Ethnopharmacol 75(2–3):125–132. doi:10.1016/S0378-8741(00)00396-2
Shishodia S, Majumdar S, Banerjee S, Aggarwal BB (2003) Cancer Res 63(15):4375–4383
Xie Y, Hang TJ, Cheng Z, Zhang ZX, An DK (2001) Zhongguo Zhong Yao Za Zhi 26(9):615–616
Lacikova L, Abdel-Hamid ME, Novotny L, Masterova I, Grancai D (2006) Chromatographia 63((3–4)):117–122. doi:1365/s10337-006-0724-y
Shen D, Pan MH, Wu QL, Park CH, Juliani HR, Ho CT (2010) J Agric Food Chem 58(12):7119–7125. doi:10.1021/jf100636h
Zhou XJ, Hu XM, Yi YM, Wan J (2009) Drug Dev Ind Pharm 35(3):305–310. doi:10.1080/03639040802302165
Liao Q, Yang W, Jia Y, Chen X, Gao Q, Bi K (2005) Yakugaku Zasshi 125(6):509–515. doi:10.1248/yakushi.125.509
Chen L, Qin F, Ma Y, Li F (2007) J Chromatogr B 855(2):255–261. doi:10.1016/j.jchromb.2007.05.016
Cai S, Huo T, Feng W, Chen L, Qin F, Li F (2008) J Chromatogr B 868(1–2):83–87
Shah VP, Midha KK, Findlay JWA, Hill HM, Hulse D, McGilveray J (2000) Pharm Res 17(12):1551–1557. doi:10.1023/A:1007669411738
Karnes HT, March C (1993) Pharm Res 10(10):1420–1426. doi:10.1023/A:1018958805795
Taylor PJ (2005) Clin Biochem 38(4):328–334. doi:10.1016/j.clinbiochem.2004.11.007
Acknowledgments
This work was supported by the National Scientific Foundation of China (No.30801421, 30873089, 81001471), Huge Project to Boost Chinese Drug Development (No.2009ZX09501-032, 2009ZX09304-003), 863 Project (No.2009AA022710, 2009AA022703, 2009AA022704), Foundation of Hunan Science Technology Department general project (2009FJ4008), and Young Teachers Boost Plans of Central South University (No.201012200133).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tan, ZR., Chen, Y., Zhou, G. et al. LC–MS–MS Quantitative Determination of Ursolic Acid in Human Plasma and Its Application to Pharmacokinetic Studies. Chroma 72, 1107–1113 (2010). https://doi.org/10.1365/s10337-010-1797-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-010-1797-1