Abstract
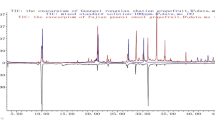
Chromatographic fingerprints of 46 Eucommia Bark samples were obtained by liquid chromatography-diode array detector (LC-DAD). These samples were collected from eight provinces in China, with different geographical locations, and climates. Seven common LC peaks that could be used for fingerprinting this common popular traditional Chinese medicine were found, and six were identified as substituted resinols (4 compounds), geniposidic acid and chlorogenic acid by LC-MS. Principal components analysis (PCA) indicated that samples from the Sichuan, Hubei, Shanxi and Anhui—the SHSA provinces, clustered together. The other objects from the four provinces, Guizhou, Jiangxi, Gansu and Henan, were discriminated and widely scattered on the biplot in four province clusters. The SHSA provinces are geographically close together while the others are spread out. Thus, such results suggested that the composition of the Eucommia Bark samples was dependent on their geographic location and environment. In general, the basis for discrimination on the PCA biplot from the original 46 objects× 7 variables data matrix was the same as that for the SHSA subset (36 × 7 matrix). The seven marker compound loading vectors grouped into three sets: (1) three closely correlating substituted resinol compounds and chlorogenic acid; (2) the fourth resinol compound identified by the OCH3 substituent in the R4 position, and an unknown compound; and (3) the geniposidic acid, which was independent of the set 1 variables, and which negatively correlated with the set 2 ones above. These observations from the PCA biplot were supported by hierarchical cluster analysis, and indicated that Eucommia Bark preparations may be successfully compared with the use of the HPLC responses from the seven marker compounds and chemometric methods such as PCA and the complementary hierarchical cluster analysis (HCA).




Similar content being viewed by others
References
Wen KC, Huang CY, Lu FL (1993) J Chromatogr A 631:241–250
Drasar P, Moravcova J (2004) J Chromatogr B 812:3–21
Normile D (2004) Am J Med 116:478–485
Stephen B, Richard K (2004) Am J Med 116:478–485
World Health Organization (1991) Guidelines for the Assessment of Herbal Medicines, Munich, 28.6.1991. WHO, Geneva
Cheng YY, Chen MJ, Tong WD (2003) J Chem Inf Comput Sci 43:1068–1076
Schoonjans V, Taylor N, Hudson BD, Massart DL (2002) J Pharmaceut Biomed Anal 28:537–548
Donohue MJ, Smallwood AW, Pfaller S, Rodgers M, Shoemaker JA, (2006) J Microbiol Meth 65:380–389
Liang YZ, Xie PS, Chan K (2007) J Chromatogr B 812:53–70
Ni YN, Zhang LS, Churchill J, Kokot S (2007) Talanta 72:1533–1539
Cheng YY, Chen MJ, Tong WD (2003) J Chem Inf Comput Sci 43:1959–1965
Shen N (2005) Shen Nong Ben Cao Jing, Gu GG (ed), Lanzhou University Press, Lanzhou
Li SZ (2005) Ben Cao Gang Mu: a translation version in modern Chinese (CD-ROM edition). Inner Mongolia Press, Holhot
Chinese Pharmacopoeia Committee (2005) Chinese Pharmacopoeia. Chemical Industry Press, Beijing
Kwan CY, Chen CX, Deyama T (2004) Vasc Pharmacol 11:229–235
Kwan CY, Zhang WB, Deyama T, Nishibe S (2004) Naunyn-Schmied Arch Pharmacol 369:206–211
Li YM, Metrori K (1999) Bio Pharm Bull 22:582–585
Yang J, Kato K, Noguchi K, Dairaku N, Koike T, Iijima K, Imatani A, Sekine H, Ohara S, Sasano H, Shimosegawa T (2003) Life Sci 73:3245–3256
Hsieh CL, Yen GC (2000) Life Sci 66:1387–1400
Hajimahmoodi M, Heyden YV, Sadeghi N, Jannat B, Oveisi MR, Shahbazian S (2005) Talanta 66:1108–1116
Gu M, Su ZG, Fan OY (2006) J Liq Chromatogr Related Technol 29:1503–1514
Zhou X, Peng JY, Fan GR, Wu YT (2005) J Chromatogr A 1092:216–221
Gu M, Zhang SF, Su ZG, Chen Y, Fan OY (2004) J Chromatogr A 1057:133–140
Gong F, Liang YZ, Xie PS, Chau FT (2004) J Chromatogr A 1029:173–183
Cheng YY, Chen MJ, Wu YJ (2002) Acta Chim Sin 60:2017–2021
Marbach R, Heise M (1990) Chemom Intell Lab Sys 9:45–63
Martens H, Naes T (1991) Multivariate Calibration. John Wiley and Sons, New York
Feng Y, Zhang L, Cai JN, Wang ZT, Xu L, Zhang ZX (2001) Talanta 53:1155–1162
Nie L, Luo GA, Cao J, Wang YM (2004) Acta Pharm Sin 39:136–139
Brereton RG (2003) Chemometrics: Data analysis for the laboratory and chemical plant. John Wiley and Sons, New York, pp 184
Gan F, Ye RY (2006) J Chromatog A 1104:100–105
Deyama T (1983) Chem Pharm Bull 31:2993–2997
Deyama T, Ikawa T, Nishibe S (1985) Chem Pharm Bull 33:3651–3657
Deyama T, Ikawa T, Kitagawa S, Nishibe S (1986) Chem Pharm Bull 34:523–527
Deyama T, Ikawa T, Kitagawa S, Nishibe S (1986) Chem Pharm Bull 34:4933–4938
Geladi P, Kowalski BR (1986) Anal Chim Acta 185:1–17
Acknowledgments
The authors gratefully acknowledge the financial support of this study by the National Natural Science Foundation of China (NSFC20365002 and NSFC20562009), the State Key Laboratories of Electroanalytical Chemistry of Changchun Applied Chemical Institute (SKLEAC2004-3) and the Chemo/Biosensing and Chemometrics of Hunan University (SKLCBC2005-22), the Jiangxi Provincial Natural Science Foundation (JXNSF062004), and the program for Changjiang Scholars and Innovative Research Team in Universities (IRT0540).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ni, Y., Peng, Y. & Kokot, S. Fingerprint Analysis of Eucommia Bark by LC-DAD and LC-MS with the Aid of Chemometrics. Chroma 67, 211–217 (2008). https://doi.org/10.1365/s10337-007-0500-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-007-0500-7