Abstract
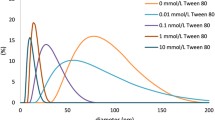
In this paper we report the use of size-exclusion chromatography (SEC) for rapid determination of the sizes and size distributions of Au nanoparticles (NPs) prepared by seed-assisted synthesis. Analytical separation of Au NPs was performed in a polymer-based column of pore size 400 nm. We characterized the sizes and size distributions of the Au NPs by using 10 mM sodium dodecyl sulfate (SDS) as mobile phase and obtained a linear relationship (R 2 = 0.986) between retention time and size of Au NPs within the range 9.8–79.1 nm; the relative standard deviations of these retention times were less than 0.3%. These separation conditions were used to characterize the sizes and size distributions of Au NPs prepared by seed-assisted synthesis. In addition to observing the elution times of the Au NPs we also simultaneously characterized their size-dependent optical properties by spectral measurement of the eluting peaks by use of an on-line diode-array detector (DAD), i.e., monitoring of the stability of the Au NP products. By using this approach we found the presence of SDS was beneficial in stabilizing the synthesized Au NPs. We also found that the volume of Au metal ions used affected the sizes of the final products. SEC seems an efficient tool for characterizing the sizes of NPs fabricated by seed-assisted synthesis.




Similar content being viewed by others
References
Mirkin CA, Letsinger RL, Mucic RC, Storhoff JJ (1996) Nature 382:607–609
Vo-Dinh T, Allain LR, Stokes DL (2002) J Raman Spectrosc 33:511–516
Jana NR (2003) Analyst 128:954–956
Lee CR, Kim SI, Yoon CJ, Gong MS, Choi BK, Kim K, Joo SW (2004) J Colloid Interface Sci 271:41–46
Watzky MA, Finke RG (1997) J Am Chem Soc 119:10382–10400
Jana NR, Gearheart L, Murphy CJ (2001) Chem Mater 13:2313–2322
Wilcoxon JP, Martin JE, Provencio P (2000) Langmuir 16:9912–9920
Liu FK, Wei GT (2004) Chromatographia 59:115–119
Gaikwad AV, Verschuren P, Eiser E, Rothenberg G (2006) J Phys Chem B 110:17437–17443
Shiundu PM, Munguti SM, Williams SKR (2003) J Chromatogr A 984:67–79
Shiundu PM, Munguti SM, Williams SKR (2003) J Chromatogr A 983:163–176
Wei GT, Liu FK (1999) J Chromatogr A 836:253–260
Choi MMF, Douglas AD, Murray RW (2006) Anal Chem 78:2779–2785
Grabar KC, Freeman RG, Hommer MB, Natan MJ (1995) Anal Chem 67:735–743
Harris DC (2007) Quantitative chemical analysis, 7th edn. WH Freeman, New York
Link S, El-Sayed MA (1999) J Phys Chem B 103:4212–4217
Toshima N, Takahashi T (1992) Bull Chem Soc Jpn 65:400–409
Yu YY, Chang SS, Lee CL, Wang CRC (1997) J Phys Chem B 101:6661–6664
Mafune F, Kohno J, Takeda Y, Kondow T (2002) J Phys Chem B 106:8555–8561
Acknowledgments
This study was supported financially by the National Science Council, Taiwan (NSC 95-2113-M-390-002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, FK. SEC Characterization of Au Nanoparticles Prepared through Seed-Assisted Synthesis. Chroma 66, 791–796 (2007). https://doi.org/10.1365/s10337-007-0412-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-007-0412-6