Abstract
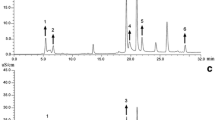
An accurate and simple HPLC method for the simultaneous determination of three protoberberine alkaloids (columbamine, jatrorrhizine, and palmatine) contained in Chinese medicine Jin-Guo-Lan (Tinospora sagittata Oliv. and Tinospora capillipes Gagnep) is presented in this study. The herb samples from six main origins and three herb markets were investigated. The separation was performed on a YMC-C18 ODS column at 30°C with a gradient elution program. Acetonitrile and phosphate buffer (0.02 mol L−1 sodium dihydrogen phosphate and 0.01 mol L−1 triethylamine, pH 3) were used as mobile phases and the flow rate was set at 1 mL min−1. The recovery of the method was in the range of 99.43–100.96%, and all the alkaloids showed good linearity (r 2 > 0.9997) in the relatively wide concentration ranges. The developed method was applied to the determination of these alkaloids in the collected herb samples, and the results showed that the contents of these components in Jin-Guo-Lan varied greatly from habitat to habitat. It was demonstrated that the proposed method was helpful for the quality evaluation of Chinese medicine Jin-Guo-Lan.





Similar content being viewed by others
References
National Commission of Chinese Pharmacopoeia (2005) Pharmacopoeia of the People’s Republic of China. Chemical Industry Press, Beijing, pp. 150–151
Vollekova A, Kostálova D, Kettmann V, Toth J (2003) Phytother Res 17:834–837
Shin JS, Kim EI, Kai M, Lee MK (2000) Neurochem Res 25:363–368
Orfila L, Rodriguez M, Colman T, Hasegawa M, Merentes E, Arvelo F (2000) J Ethnopharmacol 71:449–456
Sethi ML (1983) J Pharm Sci 72:538–541
Kobayashi Y, Yamashita Y, Fujii N, Takaboshi K, Kawakami T, Kawamura M, Mizukami T, Nakano H (1995) Planta Med 61:414–418
Chang HM, Elfishawy AM, Slatkin DJ, Schiff PL (1984) Planta Med 50:88–90
Lu GH, Wang LW, Chen JM, Xiao PG (2004) Yaowu Fenxi Zazhi 19:271–274 (in Chinese)
Zhou YY, Chen YX, Zhu B, Li F, Hao L, Li XJ (1988) Acta Pharm Sin 23:938–943 (in Chinese)
Luo X, Chen B, Yao S (2005) Talanta 66:103–110
Yang H, Wang TZ, Du LL, You YY (2004) West China J Pharm Sci 19:95–196 (in Chinese)
Wu W, Song F, Yan C, Liu Z, Liu S (2005) J Pharm Biomed Anal 37:437–446
Hussain RA, Kim JW, Beecher CWW, Kinghorn AD (1989) Heterocycles 29:2257–2260
Lu G, Wang L, Chen J, Xiao P (1999) Yaowu Fenxi Zazhi 19:271–274 (in Chinese)
Lu GH, Chen JM, Xiao PG (1995) Acta Pharm Sin 30:280–285 (in Chinese)
Sheu SJ, Li KL (1998) J High Resolut Chromatogr 21:569–573
Chuang WC, Young DS, Liu K, Li L, Sheu SJ (1996) J Chromatogr A 755:19–26
Acknowledgments
This work was supported by the National Basic Research Program (973) of Ministry of Science and Technology of the People’s Republic of China (No. 2005CB523402) and Shanghai Science and Technology Grant (Grant number: 04DZ19842, 03QMH1414, 03DZ19503, 04DZ19815, 04DZ19843, 04DZ19856, 04DZ19857 and 05DZ19733).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shi, P., Zhang, Y., Shi, Q. et al. Quantitative Determination of Three Protoberberine Alkaloids in Jin-Guo-Lan by HPLC-DAD. Chroma 64, 163–168 (2006). https://doi.org/10.1365/s10337-006-0006-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-006-0006-8