Abstract
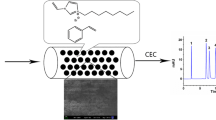
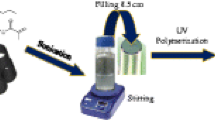
Capillary electrochromatography (CEC) has been performed with a series of C14, methlyacrylamide based monolithic columns. These monoliths with different porosities were prepared by in-situ copolymerization in fused-silica capillaries. The porous properties of monoliths were further observed using scanning electron micrographs (SEM) and measured using a mercury porosimeter. The effect of various alcohols as porogens on porous structural properties and chromatographic behaviors were also investigated. The effects of organic additive, pH value and ionic strength in mobile phase on electroosmosis flow (EOF) and separation were further discussed. Meanwhile, the baseline separation of 6 neutral compounds can be well obtained. In addition, the monolithic column demonstrates the high column efficiency and satisfactory reproducibility.
Similar content being viewed by others
References
Rebscher H, Pyell U (1996) Chromatographia 42:171–176
Behnke B, Jonansson J, Zhang S, Bayer E, Nilsson S (1998) J Chromatogr 818:257–259
Carney RA, Robson MM, Bartle KD, Myers P (1999) J High Resolut Chromatogr 22:29–32
Legido-Quigley C, Marlin ND, Melin V, Manz A, Smith NM (2003) Electrophoresis 24:917–944
Svec F, Peters EC, Sykora D, Fréchet JMJ J (2000) Chromatogr A 887:3–29
Colon LA, Burgos G, Maloney TD, Rodriguez RL (2000) Electrophoresis 21:3933–3965
Peters EC, Petro M, Svec F, Fréchet JMJ (1997) Anal Chem 69: 3646–3649
Grasselli M, Smolko E, Hargittai N, Safrany A (2001) Nuclear Instrument Methods Phys Res B 185:254–261
Moore RE, Licklider L, Schumann D, Lee TD (1998) Anal Chem 70:4879–4884
Gusev I, Huang X, Horváth C (1999) J Chromatogr A 855:273–290
Xie SF, Svec F, Fréchet JMJ, (1997) J Polym Sci A 35:1013–1021
Wu R, Zou H, Fu H, Jin W, Ye M (2002) Electrophoresis 23:1239–1245
Xie SF, Svec F, Frechet JMJ, (1997) J Chromatogr A 775:65–72.C
Zhang SH, Huang X, Zhang J, Horváth C (2000) J Chromatogr A 887:465–477
Peters EC, Petro M, Svec F, Fréchet JMJ (1998) Anal Chem 70:2288–2295
Hjertén S (1962) Arch Biochem Biophys Suppl 1:147–151
Hjertén S (1985) J Chromatogr 347:191–195
Fujimoto C, Fujise, Matsuzawa E (1996) Anal Chem 68:2753–2757
Liao JL, Li YM, Hjertén S (1996) Anal Biochem 234:27–30
Hoegger D, Freitag R (2001) J Chromatogr A 914:211–222
Palm A, Novotny MY (1997) Anal Chem 69:4499–4507
Keith D, Bartle PM (2001) J Chromatogr A 914:3–23
Author information
Authors and Affiliations
Corresponding author
Additional information
Acknowledgements We gratefully acknowledge the support of the National Natural Science Foundation of China.
Rights and permissions
About this article
Cite this article
Zhang, K., Gao, R., Yan, C. et al. Preparation and Porous Property of C14-Monolithic Column for Capillary Electrochromatography. Chromatographia 61, 55–60 (2005). https://doi.org/10.1365/s10337-004-0464-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-004-0464-9