Abstract
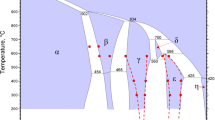
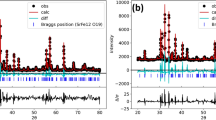
The phase relations involving various oxides in the system Sr-Cr-O at 1250 K have been determined by equilibrating samples representing 17 compositions inside the ternary and subsequent phase identification in quenched samples. Optical and scanning electron microscopes were used in conjunction with X-ray diffraction (XRD) to identify phases; the composition of each phase was verified using energy dispersive analysis of X-rays (EDX). Four stable ternary oxides, SrCrO4, Sr3Cr2O8, Sr2CrO4, and SrCr2O4, were identified. All the compounds except Sr3Cr2O8 were found stoichiometric within the accuracy of EDX. X-ray absorption near-edge structure (XANES) analysis showed that the average valence of Cr in Sr3Cr2O8 is +5.2, suggesting the presence of mixed valence states in this compound. Only one of the ternary oxides, SrCr2O4, was found to coexist with Cr metal. The compounds SrCrO4 and Sr3Cr2O8 were stable in pure oxygen. The standard Gibbs energy of formation of SrCrO4 was determined as a function of temperature in the range from 950 to 1280 K using a solidstate cell incorporating single-crystal SrF2 as the electrolyte. The oxygen potential corresponding to the three-phase equilibria involving SrCrO4, Sr3Cr2O8, and Cr2O3 was measured from 1080 to 1380 K using a cell with yttria-stabilized zirconia as the solid electrolyte and pure oxygen gas at a pressure of 0.1 MPa as the reference electrode. The standard Gibbs energy of formation of Sr3Cr2O8 was derived from the oxygen potential. The results can be represented by the following equations:
Similar content being viewed by others
References
K.T. Jacob, G.M. Kale, and K.P. Abraham: J. Electrochem. Soc., Vol 139, 1992, p 517–20.
C.W.F.T. Pistorius and M.C. Pistorius: Z. Krist., Vol 117, 1962, p 259–71.
T. Negas and R.S. Roth: J. Res. Nat. Bur. Stand. A, Vol 73A, 1969, p 431–42.
K.A. Wilhelmi: Arkiv Kemi, Vol 26, 1966, p 149–56.
V.B. Scholder and H. Schwarz: Z. Anorg. Allgem. Chem., Vol 326, 1963, p 11.
K.A. Wilhelmi and O. Jonsson: Acta Chem. Scand., Vol 19, 1965, p 177–84.
B.L. Chamberland: Solid State Commun., Vol 5, 1967, p 663–66.
K.A. Wilhelmi: Arkiv Kemi, Vol 26, 1966, p 157–65.
J.A. Kafalas and J.M. Longo: J. Solid State Chem., Vol 4, 1972, p 55–59.
Y.K. Kisil, N.G. Sharova, and B.V. Slobodin: Inorg. Mater., Vol 25, 1989, p 1490–91.
A.M. Azad, R. Sudha, and O.M. Sreedhran: Thermochim. Acta, Vol 194, 1992, p 129–36.
T. Maruyama, T. Inoue, and T. Akashi: Mater. Trans. JIM., Vol 39, 1998, p 1158–61.
A. Bianconi, J. Garcia, M. Benfatto, A. Marcelli, C.R. Natoli, and M.F. Ruiz-Lopez: Phys. Rev. B: Condens. Matter, Vol 43, 1991, p 6885–92.
K.T. Jacob and J.P. Hajra: Bull. Mater. Sci., Vol 9, 1987, p 37–46.
K.T. Jacob, T. Uda, T.H. Okabe, and Y. Waseda: J. Chem. Thermodyn., Vol 32, 2000, in press.
M.W. Chase, Jr.: NIST-JANAF Thermochemical Tables, 4th ed., Part II; J. Phys. Chem. Ref. Data, 1988, Monograph No. 9.
K.T. Jacob: J. Electrochem. Soc., Vol 124, 1977, p 1827–31.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jacob, K.T., Abraham, K.P. Phase relations in the system Sr-Cr-O and thermodynamic properties of SrCrO4 and Sr3Cr2O8 . JPE 21, 46–53 (2000). https://doi.org/10.1361/105497100770340417
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1361/105497100770340417