Abstract
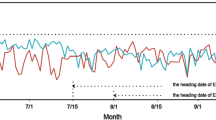
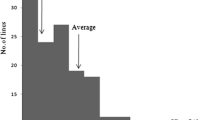
An advanced backcross population of rice was used to identify the quantitative trait locus (QTL) controlling the cold-tolerance at booting to flowering stages. The recipient, Guichao 2 (GC2), was a commercial Indica rice; the donor Dongxiang common wild rice, was an accession of common wild rice (DXCWR, Oryza rufipogon Griff.). Three QTLs for cold-tolerance were detected on chromosomes 1, 6 and 11. Two of them coming from DXCWR could enhance the cold-tolerance of the backcross progenies. Moreover, one sterility QTL that could reduce the seed set rate of the backcross progenies by 78% was mapped on chromosome 5.
Similar content being viewed by others
References
Toriyama, Y., Futsuhara, Y., Genetic studies on cold tolerance in rice, Jpn. J. Breed, 1960, 10: 143–152.
Toriyama, K., Futsuhara, Y., Genetic studies on cold tolerance in rice, V. Effectiveness of individual and line selection for cold tolerance, Jpn. J. Breed, 1971, 21: 181–188.
Futsuhara, Y., Toriyama, K., Genetic studies on cold tolerance in rice, III. Linkage relation between genes controlling cold tolerance and marker genes of Nagao and Takahashi, Jpn. J. Breed, 1966, 16: 231–242.
Futsuhara, Y., Toriyama, K., Genetic studies on cold tolerance in rice, IV. Direct and indirect effects of selection of cold tolerance, Jpn. J. Breed, 1969, 19: 286–292.
Takeuchi, Y., Hayasaka, H., Chiba, B. et al., Mapping quantitative trait loci controlling cool-temperature tolerance at the booting stage in temperate japonica rice, Breed Sci., 2001, 51: 191–197.
Andaya, V. C., Mackill, D. J., QTLs conferring cold tolerance at the booting stage of rice using recombinant inbred lines from a japonica×indica cross, Theor. Appl. Genet., 2003, 106: 1084–1090
Li, H. B., Wang, J., Liu, A. M. et al., Genetic basis of low-temperature-sensitive sterility in indica-japonica hybrids of rice as determined by RFLP analysis, Theor. Appl. Genet., 1997, 95: 1092–1097.
Saito, K., Miura, K., Nagano, K. et al., Chromosomal location of quantitative trait loci for cool tolerance at the booting stage in rice variety “Norin-PL8”, Breed Sci., 1995, 45: 337–340.
Zeng, Y. W., Ye, C. R., Shen, S. Q. et al., Chromosomal location of gene and development of near-isogenic lines for cold tolerance in rice, Sci. Agric. Sin. (in Chinese with English abstract), 2000, 33(4): 109.
Chen, D. Z., Xiao, Y.Q., Zhao, S. X. et al., Studies on cold tolerance of seedling and heading stage in Dongxiang wild rice, Acta Agric. Jiangxi (in Chinese), 1986, 8(1): 1–6.
Rogers, O. S., Bendich, A. J., Extraction of DNA from plant tissue, Plant Molecular Biology Manual, 1998, A6: 1–10.
Temnykh, S., Park, W. D., Ayres, N. et al., Mapping and genome organization of microsatellite sequences in rice (Oryza sativa L.), Theor. Appl. Genet., 2000, 100: 697–712.
Manly, K. F., Cudmore, Jr. R. H., Meer, J. M., Map Manager QTX, cross-platform software for genetic mapping, Mamm. Genome., 2001, 12: 930–932.
Sun, C. Q., Wang, X. K., Li, Z. C. et al., Comparison on the genetic diversity of common wild rice (Oryza rufipogon Griff.) and cultivated rice (O. sativa L.) using RFLP markers, Theor. Appl. Genet., 2001, 102: 157–162.
Sun, C. Q., Wang, X. K., Yoshimura, A. et al., Genetic differentiation for nuclear, mitochondrial and chloroplast genomes in common wild rice (O. rufipogon Griff.) and cultivated rice (O. sativa L.), Theor. Appl. Genet., 2002, 104: 1335–1345.
Li, D. J., Sun, C. Q., Fu, Y. C. et al., Identification and mapping of genes for improving yield from Chinese common wild rice (O. rufipogon Griff.) using advanced backcross QTL analysis, Chinese Science Bulletin, 2002, 47(18): 1533–1537.
Sasaki, T., Matsunaga, K., Inheritance and improvement of cold tolerance at booting stage in rice, I. Cold tolerance of rice cultivars: Yoneshiro, Todorokiwase and Koshihikari, Jpn. J. Breed, 1985, 35: 320–321.
Satake, T., Male sterility caused by cooling treatment at the young microspore stage in rice plants, XXIX. The mechanism of enhancement in cool tolerance by raising water temperature before the critical stage, Jpn. J. Crop. Sci., 1989, 8: 240–245.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Liu, F., Sun, C., Tan, L. et al. Identification and mapping of quantitative trait loci controlling cold-tolerance of Chinese common wild rice (O. rufipogon Griff.) at booting to flowering stages. Chin.Sci.Bull. 48, 2068–2071 (2003). https://doi.org/10.1360/03wc0287
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1360/03wc0287