Summary
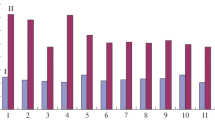
A strong tendency is currently emerging to remove not only serum but also any product of animal origin from animal cell culture media during production of recombinant proteins. This should facilitate downstream processing and improve biosafety. One way consists in the fortification of protein-free nutritive media with plant protein hydrolysates. To investigate the effects of plant peptones on mammalian cell cultivation and productivity, CHO 320 cells, a clone of CHO K1 cells genetically modified to secrete human interferon-γ (IFN-γ), were first adapted to cultivation in suspension in a protein-free medium. Both cell growth and IFN-γ secretion were found to be equivalent to those reached in serum-containing medium. Eight plant peptones, selected on the basis of their content in free amino acids and oligopeptides, as well as molecular weight distribution of oligopeptides, were tested for their ability to improve culture parameters. These were improved in the presence of three peptones, all having an important fraction of oligopeptides ranging from 1 to 10 kDa and a small proportion of peptides higher than 10 kDa. These peptones do not seem to add significantly to the nutritive potential to basal protein-free nutritive medium. Nevertheless, supplementation of an oligopeptide-enriched wheat peptone improved cell growth by up to 30% and IFN-γ production by up to 60% in shake-flask experiments. These results suggest that the use of plant peptones with potential growth factor-like or antiapoptotic bioactivities could improve mammalian cell cultivation in protein-free media while increasing the product biosafety.
Similar content being viewed by others
References
Andersen, D. C.; Krummen, L. Recombinant protein expression for therapeutic applications. Curr. Opin. Biotechnol. 13:117–123; 2002.
Castro, P. M. L.; Ison, A. P.; Hayter, P. M.; Bull, A. T The macroheterogeneity of recombinant human interferon-ψ produced by Chinese hamster ovary cells is affected by the protein and lipid content of the culture medium. Biotechnol. Appl. Biochem. 21:87–100; 1995.
Castro, P. M. L.; Ison, A. P.; Hayter, P. M.; Bull, A. T. CHO cell growth and recombinant interferon-ψ production: effects of BSA, Pluronic and lipids. Cytotechnology 19:27–36; 1996.
Chan, R. Y. Y.; Ponka, P; Sculman, H. M. Transferrin-receptor-independent but iron-dependent proliferation of variant Chinese hamster ovary cells. Exp. Cell Res. 202:326–336; 1992.
Coppen, S. R.; Newsam, R.; Bull, A. T.; Baines, A. J. Heterogeneity within populations of recombinant Chinese hamster ovary cells expressing human interferon-ψ Biotechnol. Bioeng. 46:147–158; 1995.
Curling, E.; Hayter, P. M.; Baines, A. J.; Bull, A. T.; Gull, K.; Strange, P. G.; Jenkins, N. Recombinant human interferon-ψ. Differences in glycosylation and proteolytic processing lead to heterogeneity in batch culture. Biochem. J. 272:333–337; 1990.
Dijkmans, R.; Heremans, H.; Billiau, A. Heterogeneity of Chinese hamster ovary cell produced recombinant murine interferon-ψ. J. Biol. Chem. 262(6):2528–2535; 1987
Fekkes, D.; Van Dalen, A.; Edelman, M.; Voskuilen, A. Validation of the determination of amino acids in plasma by high-performance liquid chromatography using automated pre-colum derivatization with Ophthaldialdehyde. J. Chromatogr. B 669:177–186; 1995.
Franek, F.; Eckschlager, T; Katinger, H. Enhancement of monoclonal antibody production by lysine-containing peptides. Biotechnol. Prog. 19:169–174; 2003.
Franek, F.; Hohenwarter, O.; Katinger, H. Plant protein hydrolysates: preparation of defined peptide fractions promoting growth and production in animal cells culture. Biotechnol. Prog. 16:688–692; 2000.
Franek, F.; Katinger, H. Specific effects of synthetic oligopeptides on cultured animal cells. Biotechnol. Prog. 18:155–158; 2002.
Fussenegger, M.; Betenbaugh, M. J. Metabolic Engineering II. Eukaryotic Systems Biotechnol. Bioeng. 79(5):509–31; 2002.
Gu, X.; Harmon, B.; Wang, D. I. C. Site- and branch-specific sialylation of recombinant human interferon-ψ in Chinese hamster ovary cell culture. Biotechnol. Bioeng. 55:390–398; 1997.
Hasegawa, A.; Yamashita, H.; Kondo, S., et al. Proteose peptone enhances production of tissue-type plasminogen activator from human diploid fibroblasts. Biochem. Biophys. Res. Commun. 150(3):1230–1236; 1988.
Heidemann, R.; Zhang, C.; Qi, H., et al. The use of peptones as medium additives for the production of a recombinant therapeutic protein in high density perfusion cultures of mammalian cells. Cytotechnology 32:157–167; 2000.
Hooker, A.; James, D. The glycosylation heterogeneity of recombinant human IFN-Gamma. J. Interferon Cytokine Res. 18(5):287–95; 1998.
Hooker, A.; Goldman, M. H.; Markham, H. et al. N-Glycans of recombinant human interferon-ψ change during batch culture of Chinese hamster ovary cell. Biotechnol. Bioeng. 48:639–648; 1995.
Ikonomou, L.; Bastin, G.; Schneider, Y.-J.; Agathos, S. N. Design of an efficient medium for insect cell culture and recombinant protein production. In Vitro Cell. Dev. Biol. 37A:549–559; 2001.
James, D.; Goldman, M. H.; Hoare, M.; Jenkins, N.; Oliver, R. W. A.; Green, B. N.; Freedman, R. B. Post-translational processing of recombinant human interferon-ψ in animal expression systems. Protein Sci. 5:331–340; 1996.
Jan, D. C.-H.; Jones, S. J.; Emery, A. N.; Al-Rubeai, M. Peptone, a low-cost growth-promoting nutrient for intensive animal cell culture. Cytotechnology 16:17–26; 1994.
Jayme, D.; Smith, S. R. Media formulation options and manufacturing process controls to safeguard agains introduction of animal origin contaminants in animal cell culture. Cytotechnology 33:27–36; 2000.
Mizrahi, A.: Primatone RL in mammalinan cell culure mediai. Biotechnol. Bioeng. 19:1557–1561; 1977.
Mols, J.; Burteau, C.; Verhoeyer, F.; Peeters-Joris, C.; Bastin, G; Agathos, S. N.; Schneider, Y. J. Proteolytic potential during batch cultivation in serum-free media of an IFN-ψ producing CHO cell line. In: Lindner-Olsson, E., ed. Animal cell technology: from target to market, Dordrecht, The Netherlands: Kluwer Academic Publishers; 2001:227–229.
Rasmussen, B.; Davis, R.; Thomas, J.; Reddy, P. Isolation, characterization and recombinant protein expression in Veggie-CHO: a serum-free CHO host cell line. Cytotechnology 28:31–42; 1998.
Sakai, K.; Matsunaga, T.; Hayashi, C.; Yamaji, H.; Fukuda, H. Effects of phosphatidic acid on recombinant protein production by Chinese hamster ovary cells in serum-free culture. Biochem. Eng. J. 10:85–92; 2002.
Schlaeger, E.-J. The protein hydrolysate, Primatone RL, is a cost-effective multiple growth promoter of mammanlian cell culture in serum-containing and serum-free media and displays anti-apoptosis properties. J. Immunol. Methods 194:191–199; 1996.
Schneider, Y.-J. Optimization of hybridoma cell growth and monoclonal antibody secretion in chemically defined serum- and protein-free medium. J. Immunol. Methods 116:67–77; 1989.
Sunstrom, N.-A.; Hunt, S.; Bailey, C.; Baig, M.; Sleigh, M.; Gray, P. Hegulated autocrine growth of CHO cells. Cytotechnology 34:39–46; 2000.
Towbin, H.; Staehelin, T.; Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc. Natl. Acad. Scil. USA 76:4350–4354; 1979.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Burteau, C.C., Verhoeye, F.R., Molsl, J.F. et al. Fortification of a protein-free cell culture medium with plant peptones improves cultivation and productivity of an interferon-γ-producing CHO cell line. In Vitro Cell.Dev.Biol.-Animal 39, 291–296 (2003). https://doi.org/10.1290/1543-706X(2003)039<0291:FOAPCC>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1543-706X(2003)039<0291:FOAPCC>2.0.CO;2