Abstract
Background
Following complete resection of pN2 non-small cell lung cancer (NSCLC), national guidelines recommend either sequential (sCRT) or concurrent chemoradiotherapy (cCRT). This is the largest study to date evaluating survival between both approaches. In sCRT patients, sequencing ‘chemotherapy first’ versus ‘radiotherapy first’ was also addressed.
Methods
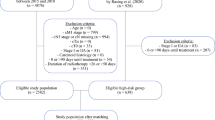
The National Cancer Data Base (NCDB) was queried for patients with primary NSCLC undergoing surgery (without neoadjuvant radiotherapy or chemotherapy), pN2 disease with negative surgical margins, and receiving postoperative CRT. Multivariable logistic regression ascertained factors associated with cCRT administration. Kaplan–Meier analysis evaluated overall survival (OS), and Cox proportional hazards modeling determined variables associated with OS. Propensity matching was performed to address group imbalances and indication biases.
Results
Of 1924 total patients, 1115 (58%) received sCRT and 809 (42%) underwent cCRT. Median OS in the sCRT and cCRT cohorts was 53 months versus 37 months (p < 0.001); differences persisted following propensity matching (p = 0.002). In the sCRT population, there was a trend for higher OS in the ‘chemotherapy first’ group, relative to ‘radiotherapy first’ (55 vs. 44 months, p = 0.079), but there were no statistically apparent differences following propensity matching (p = 0.302).
Conclusions
For completely resected pN2 NSCLC, delivering adjuvant sCRT was associated with improved survival over cCRT. Toxicity-related factors may help to explain these results but need to be better addressed in further investigations. Differential sequencing of sCRT did not appear to affect survival.



Similar content being viewed by others
References
National Comprehensive Cancer Network. Non-Small Cell Lung Cancer. Version 8.2017. https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf Accessed 21 Sep 2017.
Burdett S et al (2005) PORT Meta-analysis Trialists Group. Postoperative radiotherapy for non-small cell lung cancer. Cochrane Database Syst Rev. (18):CD002142.
Douillard JY, Rosell R, De Lena M, et al. Impact of postoperative radiation therapy on survival in patients with complete resection and stage I, II, or IIIA non-small-cell lung cancer treated with adjuvant chemotherapy: the adjuvant Navelbine International Trialist Association (ANITA) Randomized Trial. Int J Radiat Oncol Biol Phys. 2008;72:695-701.
Lally BE, Zelterman D, Colasanto JM, et al. Postoperative radiotherapy for stage ii or iii non–small-cell lung cancer using the Surveillance, Epidemiology, and End Results Database. J Clin Oncol. 2006;24:2998–3006.
Robinson CG, Patel AP, Bradley JD, et al. Postoperative Radiotherapy for Pathologic N2 Non–Small-Cell Lung Cancer Treated With Adjuvant Chemotherapy: A Review of the National Cancer Data Base. J Clin Oncol. 2015;33:870–876.
Curran WJ, Paulus R, Langer CJ et al. (2011) Sequential vs. concurrent chemoradiation for stage III non-small cell lung cancer: randomized phase III trial RTOG 9410. J Natl Cancer Inst. 103:1452–1460.
Auperin A, Le Pechoux C, Rolland E, et al. Meta-analysis of concomitant versus sequential radiochemotherapy in locally advanced non-small-cell lung cancer. J Clin Oncol. 2010;28:2181–2190.
Bradley JD, Paulus R, Graham MV, et al. Phase II trial of postoperative adjuvant paclitaxel/carboplatin and thoracic radiotherapy in resected stage II and IIIA non-small-cell lung cancer: promising long-term results of the Radiation Therapy Oncology Group–RTOG 9705. J Clin Oncol. 2005;23:3480–3487.
Feigenberg SJ, Hanlon AL, Langer C, et al. A phase II study of concurrent carboplatin and paclitaxel and thoracic radiotherapy for completely resected stage II and IIIA non-small cell lung cancer. J Thorac Oncol. 2007;2:287–292.
Kepka L, Socha J, Rucinska M, Wasilewska-Tesluk E, Komosinska K. Sequencing postoperative radiotherapy and adjuvant chemotherapy in non-small cell lung cancer: unanswered questions on the not evidence-based approach. J Thorac Dis. 2016;8:1381–1385.
Lee HW, Noh OK, Oh YT, et al. Radiation Therapy–First Strategy After Surgery With or Without Adjuvant Chemotherapy in Stage IIIA-N2 Non-Small Cell Lung Cancer. Int J Radiat Oncol Biol Phys. 2016;94:621–627.
Bilimoria KY, Stewart AK, Winchester DP, et al. The National Cancer Data Base: A Powerful Initiative to Improve Cancer Care in the United States. Ann Surg Oncol. 2008;15:683–690.
Wang EH, Corso CD, Rutter CE, et al. Postoperative Radiation Therapy Is Associated With Improved Overall Survival in Incompletely Resected Stage II and III Non–Small-Cell Lung Cancer. J Clin Oncol. 2015;33:2727–2734.
Amini A, Jones BL, McDermott JD, et al. Survival outcomes with concurrent chemoradiation for elderly patients with locally advanced head and neck cancer according to the National Cancer Data Base. Cancer. 2016;122:1533–1543.
Haque W, Verma V, Butler EB, Teh BS. Definitive chemoradiation at high volume facilities is associated with improved survival in glioblastoma. J Neurooncol. 2017;135(1):173–181.
Austin PC. Statistical criteria for selecting the optimal number of untreated subjects matched to each treated subject when using many-to- one matching on the propensity score. Am J Epidemiol. 2010; 172:1092–1097.
Rosenbaum PR, Rubin DB. The central role of the propensity score in observational studies for causal effects. Biometrika. 1983;70:15.
Austin PC, Gootendorst P, Anderson GM. A comparison of the ability of different propensity score models to balance measured variables between treated and untreated subjects: a Monte Carlo study. Stat Med. 2007;26:734–753.
Austin PC. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat Med 2009;28:3083–3107.
El-Sherif A, Fernando HC, Santos R, et al. Margin and local recurrence after sublobar resection of non-small cell lung cancer. Ann Surg Oncol. 2007;14:2400-2405.
Gomez DR, Komaki R. Postoperative Radiation Therapy for Non-Small Cell Lung Cancer and Thymic Malignancies. Cancers. 2012;4:307–322.
Kim HI, Noh OK, Oh YT, et al. Comparison of concurrent chemoradiotherapy versus sequential radiochemotherapy in patients with completely resected non-small cell lung cancer. Radiat Oncol J. 2016;34:202–208.
ClinicalTrials.gov. NCT00410683. Radiation therapy in treating patients with non-small cell lung cancer that has been completely removed by surgery (LUNG ART). Available at: https://clinicaltrials.gov/ct2/show/NCT00410683. Accessed 22 Sep 2017.
Acknowledgments
Disclosure
Steven H. Lin has received research funding from Elekta, STCube Pharmaceuticals, Peregrine, Bayer, and Roche/Genentech, and has served as a consultant for AstraZeneca and received honorarium from US Oncology and ProCure. Amy C. Moreno, Waqar Haque, Vivek Verma, and Penny Fang declare no conflicts of interest.
Funding
No research support was received for this study.
Disclaimers
None. This work has not been previously presented or published in any form.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10434_2018_6399_MOESM2_ESM.tiff
Supplemental Figure 1. Effect of clinical nodal status (cN0-1 versus cN2) on overall survival in both the sCRT (A) and cCRT (B) patients. Supplementary material 2 (TIFF 52 kb)
Rights and permissions
About this article
Cite this article
Moreno, A.C., Haque, W., Verma, V. et al. Concurrent Versus Sequential Chemoradiation Therapy in Completely Resected Pathologic N2 Non-Small Cell Lung Cancer: Propensity-Matched Analysis of the National Cancer Data Base. Ann Surg Oncol 25, 1245–1253 (2018). https://doi.org/10.1245/s10434-018-6399-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-018-6399-4