Abstract
Background
No guidelines exist regarding physicians’ duty to inform former patients about novel genetic tests that may be medically beneficial. Research on the feasibility and efficacy of disseminating information and patient opinions on this topic is limited.
Methods
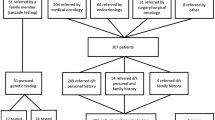
Adult patients treated at our institution from 1950 to 2010 for medullary thyroid cancer, pheochromocytoma, or paraganglioma were included if their history suggested being at-risk for a hereditary syndrome but genetic risk assessment would be incomplete by current standards. A questionnaire assessing behaviors and attitudes was mailed 6 weeks after an information letter describing new genetic tests, benefits, and risks was mailed.
Results
Ninety-seven of 312 (31.1%) eligible patients with an identified mailing address returned the questionnaire. After receiving the letter, 29.2% patients discussed genetic testing with their doctor, 39.3% considered pursuing genetic testing, and 8.5% underwent testing. Nearly all respondents (97%) indicated that physicians should inform patients about new developments that may improve their or their family’s health, and 71% thought patients shared this responsibility. Most patients understood the letter (84%) and were pleased it was sent (84%), although 11% found it upsetting.
Conclusions
Patients believe it is important for physicians to inform them of potentially beneficial developments in genetic testing. However, physician-initiated letters to introduce new information appear inadequate alone in motivating patients to seek additional genetic counseling and testing. Further research is needed regarding optimal methods to notify former patients about new genetic tests and corresponding clinical and ethical implications.
Similar content being viewed by others
References
Jimenez C, Cote G, Arnold A, Gagel RF. Review: should patients with apparently sporadic pheochromocytomas or paragangliomas be screened for hereditary syndromes?. J Clin Endocrinol Metab. 2006;91:2851–58.
Qin Y, Yao L, King EE, et al. Germline mutations in TMEM127 confer susceptibility to pheochromocytoma. Nat Genet. 2010;42(3):229–33.
Hao HX, Khalimonchuk O, Schraders M, et al. SDH5, a gene required for flavination of succinate dehydrogenase, is mutated in paraganglioma. Science. 2009;325:1139–42.
Burnichon N, Brière JJ, Libé R, et al. SDHA is a tumor suppressor gene causing paraganglioma. Hum Mol Genet. 2010;19(15):3011–20.
Yeh IT, Lenci RE, Qin Y, et al. A germline mutation of the KIF1B beta gene on 1p36 in a family with neural and nonneural tumors. Hum Genet. 2008;124(3):279–85.
Comino-Méndez I, Gracia-Aznárez FJ, Schiavi F, et al. Exome sequencing identifies MAX mutations as a cause of hereditary pheochromocytoma. Nat Genet. 2011;43(7):663–67.
Marquard J, Eng C. Multiple Endocrine Neoplasia Type 2. 1999 Sep 27 [Updated 2015 Jun 25]. In: Pagon RA, Adam MP, Ardinger HH, et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2017. https://www.ncbi.nlm.nih.gov/books/NBK1257/.
NGS in PPGL (NGSnPPGL) Study Group, Toledo RA, Burnichon N, Cascon A, et al. Consensus statement statement on next-generation-sequencing-based diagnostic testing of hereditary phaeochromocytomas and paragangliomas. Nat Rev Endocrinol. 2017;13:233–47 https://doi.org/10.1038/nrendo.2016.185 Accessed 18 Nov 2016.
Wells SA, Asa SL, Dralle H, et al. Revised American Thyroid Association guidelines for the management of medullary thyroid carcinoma. Thyroid. 2015;25(6):567–610.
Welander J, Söderkvist P, Gimm O. Genetics and clinical characteristics of hereditary pheochromocytomas and paragangliomas. Endocr Relat Cancer. 2011;18(6):R253–76.
Waguespack SG, Rich T, Grubbs E, Ying AK, Perrier ND, Ayala-Ramirez M, Jimenez C. A current review of the etiology, diagnosis, and treatment of pediatric pheochromocytoma and paraganglioma. J Clin Endocrinol Metab. 2010;95(5):2023–37.
Plouin PF, Amar L, Dekkers OM, et al. Guideline Working Group. European Society of Endocrinology clinical practice guideline for long-term follow-up of patients operated on for a phaeochromocytoma or a paraganglioma. Eur J Endocrinol. 2016;174(5):G1–10.
Lenders JW, Duh QY, Eisenhofer G, et al. Endocrine Society. Pheochromocytoma and paraganglioma: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2014;99(6):1915–42.
Tercyak KP, Hensley Alford S, Emmons KM, Lipkus IM, Wilfond BS, McBride CM. Parents’ attitudes toward pediatric genetic testing for common disease risk. Pediatrics. 2011;127(5):e1288–95.
Vernon SW, Gritz ER, Peterson SK, Perz CA, Marani S, Amos CI, Baile WF. Intention to learn results of genetic testing for hereditary colon cancer. Cancer Epidemiol Biomarkers Prev 1999;8:353–60.
Weiss, DS. The impact of event scale: revised. In: Wilson JP, Tang CS. editors Cross-cultural assessment of psychological trauma and PTSD. New York: Springer. 2007. p. 219–38.
Wolff K, Nordin K, Brun W, Berglund G, Kvale G. Affective and cognitive attitudes, uncertainty avoidance and intention to obtain genetic testing: an extension of the theory of planned behaviour. Psychol Health. 2011;26(9):1143-55.
Florida. Supreme Court. Pate v. Threlkel. Wests South Report. 1995;661:278–82.
Pentz RD, Peterson SK, Watts B, Vernon SW, Lynch PM, Koehly LM, Gritz ER. Hereditary nonpolyposis colorectal cancer family members’ perceptions about the duty to inform and health professionals’ role in disseminating genetic information. Genet Test. 2005;9:261–8.
Kohut K, Manno M, Gallinger S, Esplen MJ. Should healthcare providers have a duty to warn family members of individuals with an HNPCC-causing mutation? A survey of patients from the ontario familial colon cancer registry. J Med Genet. 2007;44:404–7.
Griffin CA, Axilbund JE, Codori AM, et al. Patient preferences regarding recontact by cancer genetics clinicians. Fam Cancer. 2007;6:265–73.
Phelps C, Platt K, France L, Gray J, Iredale R. Delivering information about cancer genetics via letter to patients at low and moderate risk of familial cancer: a pilot study in Wales. Fam Cancer. 2004;3:55–9.
Lerman C, Narod S, Schulman K, et al. BRCA1 testing in families with hereditary breast-ovarian cancer. A prospective study of patient decision making and outcomes. JAMA. 1996;275(24):1885–92.
Chaliki H, Loader S, Levenkron JC, Logan-Young W, Hall WJ, Rowley PT. Women’s receptivity to testing for a genetic susceptibility to breast cancer. Am J Public Health. 1995;85(8 Pt 1):1133–5.
Lerman C, Daly M, Masny A, Balshem A. Attitudes about genetic testing for breast-ovarian cancer susceptibility. J Clin Oncol. 1994;12(4):843–50.
Lerman C, Seay J, Balshem A, Audrain J. Interest in genetic testing among first-degree relatives of breast cancer patients. Am J Med Genet. 1995;57:385–92.
Struewing JP1, Lerman C, Kase RG, Giambarresi TR, Tucker MA. Anticipated uptake and impact of genetic testing in hereditary breast and ovarian cancer families. Cancer Epidemiol Biomarkers Prev.1995;4:169–73.
Andrykowski MA, Munn RK, Studts JL. Interest in learning of personal genetic risk for cancer: a general population survey. Prev Med. 1996;25:527–36.
Tambor ES, Rimer BK, Strigo TS. Genetic testing for breast cancer susceptibility: awareness and interest among women in the general population. Am J Med Genet. 1997;68(1):43–9.
Julian-Reynier C, Eisinger F, Vennin P, et al. Attitudes towards cancer predictive testing and transmission of information to the family. J Med Genet. 1996;33(9):731–6.
Lerman C, Biesecker B, Benkendorf JL, Kerner J, Gomez-Caminero A, Hughes C, Reed MM. Controlled trial of pretest education approaches to enhance informed decision-making for BRCA1 gene testing. J Natl Cancer Inst. 1997;89(2):148–57.
Lerman C, Schwartz MD, Lin TH, Hughes C, Narod S, Lynch HT. The influence of psychological distress on use of genetic testing for cancer risk. J Consult Clin Psychol. 1997;65(3):414–20.
Alford RL, Margolis RL, Ross CA, Richards CS. Screening for 185delAG in the Ashkenazim. Am J Hum Genet. 1997;60(5):1085–98.
Gritz ER, Peterson SK, Vernon SW, et al. Psychological impact of genetic testing for hereditary nonpolyposis colorectal cancer. J Clin Oncol. 2005;23(9):1902–10.
Lynch HT, Lemon SJ, Durham C, et al. A descriptive study of BRCA1 testing and reactions to disclosure of test results. Cancer. 1997;79(11):2219–28.
Croyle RT, Lerman C. Interest in genetic testing for colon cancer susceptibility: cognitive and emotional correlates. Prev Med. 1993;22(2):284–92.
Vernon SW, Gritz ER, Peterson SK, Amos CI, Perz CA, Baile WF, Lynch PM. Correlates of psychologic distress in colorectal cancer patients undergoing genetic testing for hereditary colon cancer. Health Psychol. 1997;16(1):73–86.
Ayala-Ramirez M, Feng L, Johnson MM, et al. Clinical risk factors for malignancy and overall survival in patients with pheochromocytomas and sympathetic paragangliomas: primary tumor size and primary tumor location as prognostic indicators. J Clin Endocrinol Metab. 2011;96(3):717–25.
Acknowledgment
The authors acknowledge the National Society of Genetic Counselors Cancer Special Interest Group for their generous funding for this project. This work was supported in part by the American Cancer Society Mentored Grant “Identifying key factors affecting the clinical course of MEN2-related MTC.”
Disclosures
Steven I. Sherman is a consultant for LOXO, Novartis, Veracyte, NovoNordisk, Genzyme, Bristol-Myers Squibb, Eisai Medical Research. The remaining authors have no financial disclosures to make.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Romero Arenas, M.A., Rich, T.A., Hyde, S.M. et al. Recontacting Patients with Updated Genetic Testing Recommendations for Medullary Thyroid Carcinoma and Pheochromocytoma or Paraganglioma. Ann Surg Oncol 25, 1395–1402 (2018). https://doi.org/10.1245/s10434-018-6366-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-018-6366-0