Abstract
Background
In gastrointestinal stromal tumors (GISTs), rupture is a high-risk feature and an indication for adjuvant treatment; however, the independent impact of rupture on prognosis is uncertain and the term is inconsistently defined. In the present study, a previously proposed definition of ‘tumor rupture’ was applied on a population-based cohort of gastric GISTs.
Methods
Patients undergoing surgery for non-metastatic gastric GISTs from 2000 to 2015 were identified in the regional sarcoma database of Oslo University Hospital. Tumor rupture included spillage or fracture, piecemeal resection, incisional biopsy, blood-tinged ascites, gastric perforation, and microscopic adjacent infiltration. Minor defects of tumor integrity were not considered rupture, i.e. core needle biopsy, peritoneal tumor penetration, superficial peritoneal rupture, and R1 resection. Risk was assessed according to the modified National Institutes of Health consensus criteria.
Results
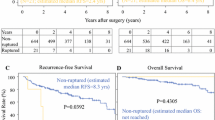
Among 242 patients, tumor rupture occurred in 22 patients and minor defects of tumor integrity occurred in 81 patients. Five-year recurrence-free survival (RFS) for patients with tumor rupture, minor defects of tumor integrity, and no defect was 37, 91, and 96%, respectively (p < 0.001). In the high-risk group, 5 year RFS for patients with rupture was 37%, versus 77% without rupture (hazard ratio 3.56, 95% confidence interval 1.57–8.08, p = 0.001). On multivariable analysis, tumor rupture and mitotic index were independently associated with recurrence. Of 13 patients who received adjuvant imatinib after tumor rupture, 11 relapsed.
Conclusions
Tumor rupture according to the present definition was independently associated with recurrence. With tumor rupture, patients relapsed despite adjuvant treatment. Without rupture, prognosis was good, even in the high-risk group.

Similar content being viewed by others
References
Hohenberger P, Ronellenfitsch U, Oladeji O, Pink D, Ströbel, Wardelmann E, Reichardt P. Pattern of recurrence in patients with ruptured primary gastrointestinal stromal tumour. Br J Surg 2010; 97(12):1854-1859.
Joensuu H, Eriksson M, Hall KS, et al. Risk factors for gastrointestinal stromal tumor recurrence in patients treated with adjuvant imatinib. Cancer 2014; 120(15):2325-2333.
Yanagimoto Y, Takahashi T, Muguruma K, et al. Re-appraisal of risk classifications for primary gastrointestinal stromal tumors (GISTs) after complete resection: indications for adjuvant therapy. Gastric Cancer 2014; 18(2):426-433.
Joensuu H. Risk stratification of patients diagnosed with gastrointestinal stromal tumor. Hum Pathol 2008; 39(10):1411-1419.
Fletcher CDM, Berman JJ, Corless C, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Hum Pathol 2002; 33(5):459-465.
Miettinen M, Lasota J. Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med 2006; 130(10):1466-1478.
Rutkowski P, Bylina E, Wozniak A, et al. Validation of the Joensuu risk criteria for primary resectable gastrointestinal stromal tumour: the impact of tumour rupture on patient outcomes. Eur J Surg Oncol 2011; 37(10):890-896.
Kim M-C, Yook J-H, Yang H-K, et al. Long-Term Surgical Outcome of 1057 Gastric GISTs According to 7th UICC/AJCC TNM System. Medicine 2015; 94(41):e1526-e1527.
Bischof DA, Kim Y, Dodson R, et al. Conditional disease-free survival after surgical resection of gastrointestinal stromal tumors. JAMA Surg 2015; 150(4):299–14.
Wozniak A, Rutkowski P, Schoffski P, et al. Tumor genotype is an independent prognostic factor in primary gastrointestinal stromal tumors of gastric origin: a european multicenter analysis based on ConticaGIST. Clin Cancer Res 2014; 20(23):6105-6116.
National Comprehensive Cancer Network guidelines. Version 2.2017. NCCN Clinical Practice Guidelines in Oncology: Soft Tissue Sarcoma. Available at: http://www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed 3 Mar 2017.
Hølmebakk T, Bjerkehagen B, Boye K, Bruland Ø, Stoldt S, Sundby Hall K. Definition and clinical significance of tumour rupture in gastrointestinal stromal tumours of the small intestine. Br J Surg 2016; 103:684–691.
13. Joensuu H, Vehtari A, Riihimäki J, et al. Risk of recurrence of gastrointestinal stromal tumour after surgery: an analysis of pooled population-based cohorts. Lancet Oncol 2012; 13(3):265-274.
Fletcher CDM, Bridge JA, Hogendoorn PCW, Mertens F (eds) (2013) WHO Classification of Tumours of Soft Tissue and Bone WHO/IARC Classification of tumours. 4th Edition, ARC, Lyon
Joensuu H, Eriksson M, Sundby Hall K, et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor: a randomized trial. JAMA 2012; 307(12):1265-1272.
Rutkowski P, Nowecki ZI, Michej W, et al. Risk criteria and prognostic factors for predicting recurrences after resection of primary gastrointestinal stromal tumor. Ann Surg Oncol 2007; 14(7):2018-2027.
The ESMO/European Sarcoma Network Working Group. Gastrointestinal stromal tumours: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2014; 25:iii21-iii26.
Acknowledgements
The authors are indebted to Ms. Trine Thoresen, administrator of the Sarcoma Database at OUH, for secretarial assistance, and Ms. Jeanne-Marie Berner, PhD, head of the Section for Molecular Pathology, for providing mutational data. In the present study, information from the Cancer Registry of Norway has been used. The interpretation and reporting of these data are the responsibility of the authors and no endorsement by the Cancer Registry of Norway should be inferred.
Funding
This work was supported by the Norwegian Cancer Society (Grant Number 5790283 to KB).
Disclosures
Toto Hølmebakk, Ivar Hompland, Bodil Bjerkehagen, Stephan Stoldt, Øyvind Sverre Bruland, Kirsten Sundby Hall, and Kjetil Boye declare no conflict of interests.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10434_2018_6353_MOESM1_ESM.tif
Recurrence-free survival after complete resection of gastric GISTs. Kaplan–Meier survival curves of recurrence-free survival. a For the complete cohort; and b by the modified NIH criteria. GISTs gastrointestinal stromal tumors, HR hazard ratio, CI confidence interval, NIH National Institutes of Health. aCalculated between intermediate- and high-risk tumors. Supplementary material 1 (TIFF 1746 kb)
Rights and permissions
About this article
Cite this article
Hølmebakk, T., Hompland, I., Bjerkehagen, B. et al. Recurrence-Free Survival After Resection of Gastric Gastrointestinal Stromal Tumors Classified According to a Strict Definition of Tumor Rupture: A Population-Based Study. Ann Surg Oncol 25, 1133–1139 (2018). https://doi.org/10.1245/s10434-018-6353-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-018-6353-5