Abstract
Background
Curative treatment of pseudomyxoma peritonei (PMP) is complete cytoreductive surgery (CRS) with hyperthermic intraperitoneal chemotherapy (HIPEC).
Objective
The aim of this study was to build and evaluate a preoperative imaging score to predict resectability.
Patients and methods
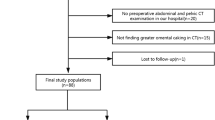
Between 2007 and 2014, all PMP patients in two tertiary reference centers who underwent laparotomy with intent to undergo CRS and HIPEC were included in this study retrospectively. Thickness of tumor burden was measured on preoperative multidetector-row computed tomography (MDCT) by two radiologists blinded to surgical results in five predetermined areas. Patients were divided into two cohorts with the same resectability rate (building and validation). The performances of the scores were assessed using receiver operating characteristic (ROC) curve analyses.
Results
Overall, 126 patients were included, with compete CRS being achieved in 91/126 patients (72.2%). Two cohorts of 63 patients matched by age, sex, burden of disease, resectability rate, and pathological grade were constituted. The MDCT score was the sum of the five measures, and was higher in unresectable disease [median 46.2 mm (range 27.9–74.6) vs. 0.0 mm (range 0.0–14.0), p < 0.001]. Area under the ROC curve was 0.863 (range 0.727–0.968) and 0.801 (range 0.676–0.914) in the building and validation cohorts, respectively. A threshold of 28 mm yielded a sensitivity, specificity, positive predictive and negative predictive value of 94, 81, 81 and 94% in the building cohort, and 80, 68, 59 and 85% in the validation cohort, respectively. Using our score, overall and disease-free survival were increased in the group classified as resectable.
Conclusion
A simple preoperative MDCT score measuring tumor burden in the perihepatic region is able to predict resectability and survival of PMP patients.




Similar content being viewed by others
References
Smeenk RM, Verwaal VJ, Zoetmulder FA. Pseudomyxoma peritonei. Cancer Treat Rev. 2007;33(2):138–45.
Ronnett BM, Shmookler BM, Sugarbaker PH, Kurman RJ. Pseudomyxoma peritonei: new concepts in diagnosis, origin, nomenclature, and relationship to mucinous borderline (low malignant potential) tumors of the ovary. Anat Pathol. 1997;2:197–26.
Slim K, Blay JY, Brouquet A, et al. Digestive oncology: surgical practices [in French]. J Chir (Paris). 2009;146(Suppl 2):S11–S80.
Moran BJ, Cecil TD. The etiology, clinical presentation, and management of pseudomyxoma peritonei. Surg Oncol Clin N Am. 2003;12(3):585–603.
Elias D, Gilly F, Quenet F, et al. Pseudomyxoma peritonei: a French multicentric study of 301 patients treated with cytoreductive surgery and intraperitoneal chemotherapy. Eur J Surg Oncol. 2010;36(5):456–62.
Passot G, Vaudoyer D, Villeneuve L, et al. What made hyperthermic intraperitoneal chemotherapy an effective curative treatment for peritoneal surface malignancy: a 25-year experience with 1,125 procedures. J Surg Oncol. 2016;113(7):796–803.
Chua TC, Moran BJ, Sugarbaker PH, et al. Early- and long-term outcome data of patients with pseudomyxoma peritonei from appendiceal origin treated by a strategy of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J Clin Oncol. 2012;30(20):2449–56.
Baratti D, Kusamura S, Milione M, et al. Pseudomyxoma peritonei of extra-appendiceal origin: a comparative study. Ann Surg Oncol. 2016;23(13):4222–30.
Ansari N, Chandrakumaran K, Dayal S, Mohamed F, Cecil TD, Moran BJ. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in 1000 patients with perforated appendiceal epithelial tumours. Eur J Surg Oncol. 2016;42(7):1035–41.
Loungnarath R, Causeret S, Brigand C, Gilly FN, Glehen O. Pseudomyxoma peritonei: new concept and new therapeutic approach [in French]. Ann Chir. 2005;130(2):63–69.
Delhorme JB, Elias D, Varatharajah S, et al. Can a Benefit be Expected from Surgical Debulking of Unresectable Pseudomyxoma Peritonei? Ann Surg Oncol. 2016;23(5):1618–24.
Sulkin TV, O’Neill H, Amin AI, Moran B. CT in pseudomyxoma peritonei: a review of 17 cases. Clin Radiol. 2002;57(7):608–13.
Diop AD, Fontarensky M, Montoriol PF, Da Ines D. CT imaging of peritoneal carcinomatosis and its mimics. Diagn Interv Imaging. 2014;95(9):861–72.
Menassel B, Duclos A, Passot G, et al. Preoperative CT and MRI prediction of non-resectability in patients treated for pseudomyxoma peritonei from mucinous appendiceal neoplasms. Eur J Surg Oncol. 2016;42(4):558–66.
Chua TC, Al-Zahrani A, Saxena A, et al. Determining the association between preoperative computed tomography findings and postoperative outcomes after cytoreductive surgery and perioperative intraperitoneal chemotherapy for pseudomyxoma peritonei. Ann Surg Oncol. 2011;18(6):1582–89.
Sugarbaker PH. Peritonectomy procedures. Ann Surg. 1995;221(1):29–42.
Sugarbaker PH. Pseudomyxoma peritonei. A cancer whose biology is characterized by a redistribution phenomenon. Ann Surg. 1994;219(2):109–11.
Jacquet P, Sugarbaker PH. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. Cancer Treat Res. 1996;82:359–74.
Benhaim L, Honore C, Goere D, Delhorme JB, Elias D. Huge pseudomyxoma peritonei: Surgical strategies and procedures to employ to optimize the rate of complete cytoreductive surgery. Eur J Surg Oncol. 2016;42(4):552–57.
Dohan A, Hoeffel C, Soyer P, et al. Evaluation of the peritoneal carcinomatosis index with CT and MRI. Br J Surg. 2017;104(9):1244–49.
Jacquet P, Jelinek JS, Chang D, Koslowe P, Sugarbaker PH. Abdominal computed tomographic scan in the selection of patients with mucinous peritoneal carcinomatosis for cytoreductive surgery. J Am Coll Surg. 1995;181(6):530–38.
Esquivel J, Sticca R, Sugarbaker P, et al. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in the management of peritoneal surface malignancies of colonic origin: a consensus statement. Society of Surgical Oncology. Ann Surg Oncol. 2007;14(1):128–33.
Low RN, Sebrechts CP, Barone RM, Muller W. Diffusion-weighted MRI of peritoneal tumors: comparison with conventional MRI and surgical and histopathologic findings–a feasibility study. AJR Am J Roentgenol. 2009;193(2):461–70.
Klumpp B, Aschoff P, Schwenzer N, et al. Correlation of preoperative magnetic resonance imaging of peritoneal carcinomatosis and clinical outcome after peritonectomy and HIPEC after 3 years of follow-up: preliminary results. Cancer Imaging. 2013;13(4):540–47.
Passot G, Glehen O, Pellet O, et al. Pseudomyxoma peritonei: role of 18F-FDG PET in preoperative evaluation of pathological grade and potential for complete cytoreduction. Eur J Surg Oncol. 2010;36(3):315–23.
Dubreuil J, Giammarile F, Rousset P, et al. FDG-PET/ceCT is useful to predict recurrence of Pseudomyxoma peritonei. Eur J Nucl Med Mol Imaging. 2016;43(9):1630–37.
Dohan A, Barral M, Eveno C, et al. Prediction of clinical outcome using blood flow volume in the superior mesenteric artery in patients with pseudomyxoma peritonei treated by cytoreductive surgery. Eur J Surg Oncol. 2017;43(10):1932–38.
Valle M, Garofalo A. Laparoscopic staging of peritoneal surface malignancies. Eur J Surg Oncol. 2006;32(6):625–27.
Pomel C, Appleyard TL, Gouy S, Rouzier R, Elias D. The role of laparoscopy to evaluate candidates for complete cytoreduction of peritoneal carcinomatosis and hyperthermic intraperitoneal chemotherapy. Eur J Surg Oncol. 2005;31(5):540–43.
Najah H, Lo Dico R, Grienay M, Dohan A, Dray X, Pocard M. Single-incision flexible endoscopy (SIFE) for detection and staging of peritoneal carcinomatosis. Surg Endosc. 2016;30(9):3808–815.
Ladjici Y, Dray X, Marteau P, Valleur P, Pocard M. Flexible versus rigid single-port peritoneoscopy: a randomized controlled trial in a live porcine model followed by initial experience in human cadavers. Surg Endosc. 2012;26(9):2651–57.
Najah H, Lo Dico R, Dohan A, Marry L, Eveno C, Pocard M. A feasibility study of the use of computed virtual chromoendoscopy for laparoscopic evaluation of peritoneal metastases. Surg Endosc. 2017;31(2):743–51.
Pocard M, Eveno C, Valleur P. Regarding “two steps procedure for pseudomyxoma”. Ann Surg Oncol. 2011;18(1):282–83; author reply 284–85.
Dayal S, Taflampas P, Riss S, et al. Complete cytoreduction for pseudomyxoma peritonei is optimal but maximal tumor debulking may be beneficial in patients in whom complete tumor removal cannot be achieved. Dis Colon Rectum. 2013;56(12):1366–72.
Disclosures
Morgane Bouquot, Anthony Dohan, Etienne Gayat, Maxime Barat, Olivier Glehen, Marc Pocard, Pascal Rousset, and Clarisse Eveno have no financial or other personal conflicts of interest to declare.
Author information
Authors and Affiliations
Contributions
Morgane Bouquot contributed to the acquisition of data, writing of the manuscript and analysis of data. Anthony Dohan, Maxime Barat and Clarisse Eveno contributed to development of the methodology, interpretation of data, and conception, design, and review of the manuscript. Etienne Gayat contributed to development of the methodology, analysis and interpretation of data, and writing and review of the manuscript. Olivier Glehen and Marc Pocard contributed to revision of the manuscript and study supervision. Pascal Rousset contributed to acquisition and analysis of data, and review of the manuscript.
Corresponding author
Rights and permissions
About this article
Cite this article
Bouquot, M., Dohan, A., Gayat, E. et al. Prediction of Resectability in Pseudomyxoma Peritonei with a New CT Score. Ann Surg Oncol 25, 694–701 (2018). https://doi.org/10.1245/s10434-017-6275-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-6275-7