Abstract
Background
Improved staging systems that better predict survival for breast cancer patients who receive neoadjuvant chemotherapy (NAC) by accounting for clinical pathological stage plus estrogen receptor (ER) and grade (CPS+EG) and ERBB2 status (Neo-Bioscore) have been proposed. We sought to evaluate the generalizability and performance of these staging systems in a national cohort.
Methods
The National Cancer Database (2006–2012) was reviewed for patients with breast cancer who received NAC and survived ≥90 days after surgery. Four systems were evaluated: clinical/pathologic American Joint Committee on Cancer (AJCC) 7th edition, CPS+EG, and Neo-Bioscore. Unadjusted Kaplan–Meier analysis and adjusted Cox proportional hazards models quantified overall survival (OS). Systems were compared using area under the curve (AUC) and integrated discrimination improvement (IDI).
Results
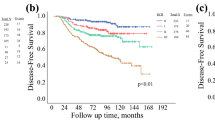
Overall, 43,320 patients (5-year OS 76.0, 95% confidence interval [CI] 75.4–76.5%) were included, 12,002 of whom had evaluable Neo-Bioscore. AUC at 5 years for CPS+EG (0.720, 95% CI 0.714–0.726) and Neo-Bioscore (0.729, 95% CI 0.716–0.742) were improved relative to AJCC clinical (0.650, 95% CI 0.643–0.656) and pathologic (0.683, 95% CI 0.676–0.689) staging. Both CPS+EG (IDI 7.2, 95% CI 6.6–7.7%) and Neo-Bioscore (IDI 9.8, 95% CI 8.0–11.6%) demonstrated superior discrimination when compared with AJCC clinical staging at 5 years. Comparison of CPS+EG with Neo-Bioscore yielded an IDI of 2.6% (95% CI 0.9–4.5%), indicating that Neo-Bioscore is the best staging system.
Conclusions
In a heterogenous national cohort of breast cancer patients treated with NAC and surgery, the incorporation of chemotherapy response, tumor grade, ER status, and ERBB2 status into the staging system substantially improved on the AJCC TNM staging system in discrimination of OS. Neo-Bioscore provided the best staging discrimination.


Similar content being viewed by others
References
Haddad TC, Goetz MP. Landscape of neoadjuvant therapy for breast cancer. Ann Surg Oncol. 2015;22(5):1408–15.
Fisher B, Brown A, Mamounas E, Wieand S, Robidoux A, Margolese RG, et al. Effect of preoperative chemotherapy on local-regional disease in women with operable breast cancer: findings from National Surgical Adjuvant Breast and Bowel Project B-18. J Clin Oncol. 1997;15(7):2483–93.
Untch M, Konecny GE, Paepke S, von Minckwitz G. Current and future role of neoadjuvant therapy for breast cancer. Breast. 2014;23(5):526–37.
Loibl S, Denkert C, von Minckwitz G. Neoadjuvant treatment of breast cancer: clinical and research perspective. Breast. 2015;24 Suppl 2:S73-7.
Boughey JC, McCall LMM, Ballman KV, Mittendorf EA, Ahrendt GM, Wilke LG, et al. Tumor biology correlates with rates of breast-conserving surgery and pathologic complete response after neoadjuvant chemotherapy for breast cancer: findings from the ACOSOG Z1071 (Alliance) Prospective Multicenter Clinical Trial. Ann Surg. 2014. 2014;260(4):608–16.
Cortazar P, Zhang L, Untch M, Mehta K, Costantino JP, Wolmark N, et al. Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet. 2014;384(9938):164–72.
Wu K, Yang Q, Liu Y, Wu A, Yang Z. Meta-analysis on the association between pathologic complete response and triple-negative breast cancer after neoadjuvant chemotherapy. World J Surg Oncol. 2014;12(1):95.
Symmans WF, Peintinger F, Hatzis C, Rajan R, Kuerer H, Valero V, et al. Measurement of residual breast cancer burden to predict survival after neoadjuvant chemotherapy. J Clin Oncol. 2007;25(28):4414–22.
Von Minckwitz G, Untch M, Blohmer JU, Costa SD, Eidtmann H, Fasching PA, et al. Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J Clin Oncol. 2012;30(15):1796–804.
Schwartz AM, Henson DE, Chen D, Rajamarthandan S. Histologic grade remains a prognostic factor for breast cancer regardless of the number of positive lymph nodes and tumor size: A study of 161 708 cases of breast cancer from the SEER program. Arch Pathol Lab Med. 2014;138(8):1048–52.
Edge SB, American Joint Committee on Cancer. AJCC cancer staging manual, 8th ed. American Joint Committee on Cancer; 2017. p. 1024.
Jeruss JS, Mittendorf EA, Tucker SL, Gonzalez-Angulo AM, Buchholz TA, Sahin AA, et al. Combined use of clinical and pathologic staging variables to define outcomes for breast cancer patients treated with neoadjuvant therapy. J Clin Oncol. 2008;26(2):246–52.
Mittendorf EA, Vila J, Tucker SL, Chavez-MacGregor M, Smith BD, Symmans WF, et al. The Neo-Bioscore update for staging breast cancer treated with neoadjuvant chemotherapy. JAMA Oncol. 2016;2(7):929–936.
Yi M, Mittendorf EA, Cormier JN, Buchholz TA, Bilimoria K, Sahin AA, et al. Novel staging system for predicting disease-specific survival in patients with breast cancer treated with surgery as the first intervention: time to modify the current American Joint Committee on Cancer staging system. J Clin Oncol. 2011;29(35):4654–61.
Abdelsattar JM, Al-Hilli Z, Hoskin TL, Heins CN, Boughey JC. Validation of the CPS + EG staging system for disease-specific survival in breast cancer patients treated with neoadjuvant chemotherapy. Ann Surg Oncol. 2016;23(10):3206–11.
Mittendorf EA, Jeruss JS, Tucker SL, Kolli A, Newman LA, Gonzalez-Angulo AM, et al. Validation of a novel staging system for disease-specific survival in patients with breast cancer treated with neoadjuvant chemotherapy. J Clin Oncol. 2011;29(15):1956–62.
Mittendorf EA, Jeruss JS, Tucker SL, Kolli A, Newman LA, Hortobagyi GN, et al. Validation of a novel staging system for disease-specific survival in patients with breast cancer treated with neoadjuvant chemotherapy. J Clin Oncol. 2011;29(15):1956–62.
Bilimoria KY, Stewart AK, Winchester DP, Ko CY. The National Cancer Data Base: a powerful initiative to improve cancer care in the United States. Ann Surg Oncol. 2008;15(3):683–90.
Raval MV, Bilimoria KY, Stewart AK, Bentrem DJ, Ko CY. Using the NCDB for cancer care improvement: an introduction to available quality assessment tools. J Surg Oncol. 2009;99(8):488–90.
Deyo RA, Cherkin DC, Ciol MA. Adapting a clinical comorbidity index for use with ICD-9-CM administrative databases. J Clin Epidemiol. 1992;45(6):613–9.
Hanley JA, McNeil BJ. The meaning and use of the area under a receiver operating (ROC) curve characteristic. Radiology. 1982;143(1):29–36.
Chambless LE, Diao G. Estimation of time-dependent area under the ROC curve for long-term risk prediction. Stat Med. 2006;25(20):3474–86.
Pencina MJ, D’Agostino RB, Song L. Quantifying discrimination of Framingham risk functions with different survival C statistics. Stat Med. 2012;31(15):1543–53.
Pencina MJ, D’Agostino RB, D’Agostino RB, Vasan RS. Evaluating the added predictive ability of a new marker: From area under the ROC curve to reclassification and beyond. Stat Med. 2008;27(2):157–72.
Pencina MJ, D’Agostino RB, Vasan RS. Statistical methods for assessment of added usefulness of new biomarkers. Vol. 48, Clin Chem Lab Med. 2010;48(12):1703–11.
Leening MJG, Steyerberg EW, Van Calster B, D’Agostino RB, Pencina MJ. Net reclassification improvement and integrated discrimination improvement require calibrated models: relevance from a marker and model perspective. Stat Med. 2014;33(19):3415–8.
Storlie CB, Swiler LP, Helton JC, Sallaberry CJ. Implementation and evaluation of nonparametric regression procedures for sensitivity analysis of computationally demanding models. Reliab Eng Syst Saf. 2009;94(11):1735–63.
Storlie CB, Reich BJ, Helton JC, Swiler LP, Sallaberry CJ. Analysis of computationally demanding models with continuous and categorical inputs. Reliab Eng Syst Saf. 2013;113(1):30–41.
Schafer JL, Graham JW. Missing data: our view of the state of the art. Psychol Methods. 2002;7(2):147–77.
R Core Team. R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2016. https://www.r-project.org.
Swisher SK, Vila J, Tucker SL, Bedrosian I, Shaitelman SF, Litton JK, et al. Locoregional control according to breast cancer subtype and response to neoadjuvant chemotherapy in breast cancer patients undergoing breast-conserving therapy. Ann Surg Oncol. 2016;23(3):749–56.
Elizalde PV, Cordo Russo RI, Chervo MF, Schillaci R. ErbB-2 nuclear function in breast cancer growth, metastasis and resistance to therapy. Endocr Relat Cancer. 2016;23(12):T243–57.
Mayer EL, Gropper AB, Harris L, Gold JM, Parker L, Kuter I, et al. Long-term follow-up after preoperative trastuzumab and chemotherapy for HER2-overexpressing breast cancer. Clin Breast Cancer. 2015;15(1):24–30.
Slamon DJ, Godolphin W, Jones LA, Holt JA, Steven G, Keith DE, et al. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science. 1989;244(4905):707–12.
Romond EH, Perez EA, Bryant J, Suman VJ, Geyer CE, Davidson NE, et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med. 2005;353(16):1673–84.
Slamon D, Eiermann W, Robert N, Pienkowski T, Martin M, Press M, et al. Adjuvant trastuzumab in HER2-positive breast cancer. N Engl J Med. 2011;365(14):1273–83.
Swain SM, Kim SB, Cortés J, Ro J, Semiglazov V, Campone M, et al. Pertuzumab, trastuzumab, and docetaxel for HER2-positive metastatic breast cancer (CLEOPATRA study): overall survival results from a randomised, double-blind, placebo-controlled, phase 3 study. Lancet Oncol. 2013;14(6):461–71.
Metzger-Filho O, Winer EP, Krop I. Pertuzumab: optimizing HER2 blockade. Clin Cancer Res. 2013;19(20):5552–6.
Gleason DF. Classification of prostatic carcinomas. Cancer Chemother Rep. 1966;50(3):125–8.
Delahunt B, Miller RJ, Srigley JR, Evans AJ, Samaratunga H. Gleason grading: past, present and future. Histopathology. 2012;60(1):75–86.
Marisa L, de Reyniès A, Duval A, Selves J, Gaub MP, Vescovo L, et al. Gene expression classification of colon cancer into molecular subtypes: characterization, validation, and prognostic value. PLoS Med. 2013;10(5):e1001453.
Marisa L, de Reyniès A, Duval A, Selves J, Gaub MP, Vescovo L, et al. Gene expression classification of colon cancer into molecular subtypes: characterization, validation, and prognostic value. PLoS Med. 2013;10(5):e1001453.
Caritg O, Navarro A, Moreno I, Martínez-Rodenas F, Cordeiro A, Muñoz C, et al. Identifying high-risk stage II colon cancer patients: a three-microRNA-based score as a prognostic biomarker. Clin Colorectal Cancer. 2016;15(4):e175–82.
Zhou X, Huang Z, Xu L, Zhu M, Zhang L, Zhang H, et al. A panel of 13-miRNA signature as a potential biomarker for predicting survival in pancreatic cancer. Oncotarget. 2016;7(43):69616–69624.
Yao Y-F, Du C-Z, Chen N, Chen P, Gu J. Expression of HER-2 in rectal cancers treated with preoperative radiotherapy. Dis Colon Rectum. 2014;57(5):602–7.
Acknowledgment
The NCDB is a joint project of the CoC of the American College of Surgeons and the American Cancer Society. The data used are derived from a de-identified NCDB PUF. The American College of Surgeons and the CoC have not verified, and are not responsible for, the analytic or statistical methods or the conclusions drawn from these data by the investigators. The authors gratefully acknowledge the support of the Mayo Clinic Department of Surgery and the Mayo Clinic Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery as substantial contributors of resources to the project. Additionally, Dr. Bergquist acknowledges the support of the Mayo Clinic Clinician Investigator Training Program for salary support. Finally, we would like to thank the Society of Surgical Oncology for affording us the opportunity to present this work at their annual Cancer Symposium in March 2017. Additionally, Dr. Bergquist acknowledges the Susan G Komen for the Cure foundation for their support of his attendance at the SSO meeting with their annual Breast Cancer research award.
Conflicts of interest
John R. Bergquist, Brittany L. Murphy, Curtis B. Storlie, Elizabeth B. Habermann, and Judy C. Boughey disclose no conflicts of interest.
Funding/Support
The Mayo Clinic Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery provides salary support for Dr. Habermann and Dr. Murphy. Dr. Bergquist receives salary support from the Mayo Clinic Clinician Investigator Training program. The conduct and presentation of this research was independent of the above funding sources. This work has not previously, or concurrently, been submitted for publication.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bergquist, J.R., Murphy, B.L., Storlie, C.B. et al. Incorporation of Treatment Response, Tumor Grade and Receptor Status Improves Staging Quality in Breast Cancer Patients Treated with Neoadjuvant Chemotherapy. Ann Surg Oncol 24, 3510–3517 (2017). https://doi.org/10.1245/s10434-017-6010-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-6010-4