Abstract
Background
The status of the sentinel lymph node in melanoma is an important prognostic factor. The clinical predictors and implications of false-negative (FN) biopsy remain debatable.
Methods
We compared patients with positive sentinel lymph node biopsy (SNB) [true positive (TP)] and negative SNB with and without regional recurrence [FN, true negative (TN)] from our prospective institutional database.
Results
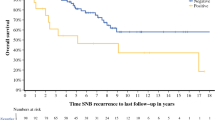
Among 2986 patients (84 FN, 494 TP, and 2408 TN; median follow-up 93 months), the incidence of FN-SNB was 2.8 %. While calculated FN rate was 14.5 % [84 FN/(494 TP + 84 FN) × 100], when we accounted for local/in-transit recurrence (LITR) this rate was 8.5 % [46 FN/(494 TP + 46 FN) × 100 %]. On multivariate analysis, male gender (OR 2.0, 95 % CI 1.1–3.6, p = 0.018), head/neck primaries (OR 2.5, 95 % CI 1.3–4.8, p < 0.006), and LITR (OR 3.5, 95 % CI 2.1–5.8, p < 0.001) were associated with FN-SNB. Melanoma-specific survival (MSS) for the FN group was similar to the TP group at 5 years (68 vs. 73 %, p = 0.539). However, MSS declined more for the FN group with a longer follow up and was significantly worse at 10 years (44 vs. 64 %, p < 0.001). On multivariate analysis, FN-SNB was a significant predictor of worse MSS in melanomas <4 mm in Breslow thickness (HR 1.6; 95 % CI 1.1–2.5, p = 0.021).
Conclusions
Male gender, LITR, and head and neck tumors were associated with FN-SNB. FN-SNB was an independent predictor of worse MSS in melanomas <4 mm in thickness, but this survival difference did not become apparent until after 5 years of follow-up.



Similar content being viewed by others
References
Morton DL, Thompson JF, Cochran AJ, et al. Final trial report of sentinel-node biopsy versus nodal observation in melanoma. N Engl J Med. 2014;370(7):599–609.
Wong SL, Balch CM, Hurley P, et al. Sentinel lymph node biopsy for melanoma: American Society of Clinical Oncology and Society of Surgical Oncology joint clinical practice guideline. J Clin Oncol. 2012;30(23):2912–8.
Morton DL, Cochran AJ, Thompson JF, et al. Sentinel node biopsy for early-stage melanoma: accuracy and morbidity in MSLT-I, an international multicenter trial. Ann Surg. 2005;242(3):302–11; discussion 311–3.
Gershenwald JE, Colome MI, Lee JE, et al. Patterns of recurrence following a negative sentinel lymph node biopsy in 243 patients with stage I or II melanoma. J Clin Oncol. 1998;16(6):2253–60.
Carlson GW, Page AJ, Cohen C, et al. Regional recurrence after negative sentinel lymph node biopsy for melanoma. Ann Surg. 2008;248(3):378–86.
Zogakis TG, Essner R, Wang HJ, et al. Melanoma recurrence patterns after negative sentinel lymphadenectomy. Arch Surg. 2005;140(9):865–71; discussion 871–2.
Chao C, Wong SL, Edwards MJ, et al. Sentinel lymph node biopsy for head and neck melanomas. Ann Surg Oncol. 2003;10(1):21–6.
Scoggins CR, Martin RC, Ross MI, et al. Factors associated with false-negative sentinel lymph node biopsy in melanoma patients. Ann Surg Oncol. 2010;17(3):709–17.
Yee VS, Thompson JF, McKinnon JG, et al. Outcome in 846 cutaneous melanoma patients from a single center after a negative sentinel node biopsy. Ann Surg Oncol. 2005;12(6):429–39.
Li LX, Scolyer RA, Ka VS, et al. Pathologic review of negative sentinel lymph nodes in melanoma patients with regional recurrence: a clinicopathologic study of 1152 patients undergoing sentinel lymph node biopsy. Am J Surg Pathol. 2003;27(9):1197–202.
McDonald K, Page AJ, Jordan SW, et al. Analysis of regional recurrence after negative sentinel lymph node biopsy for head and neck melanoma. Head Neck. 2013;35(5):667–71.
Karim RZ, Scolyer RA, Li W, et al. False negative sentinel lymph node biopsies in melanoma may result from deficiencies in nuclear medicine, surgery, or pathology. Ann Surg. 2008;247(6):1003–10.
Caraco C, Marone U, Celentano E, Botti G, Mozzillo N. Impact of false-negative sentinel lymph node biopsy on survival in patients with cutaneous melanoma. Ann Surg Oncol. 2007;14(9):2662–7.
Nowecki ZI, Rutkowski P, Nasierowska-Guttmejer A, Ruka W. Survival analysis and clinicopathological factors associated with false-negative sentinel lymph node biopsy findings in patients with cutaneous melanoma. Ann Surg Oncol. 2006;13(12):1655–63.
Jones EL, Jones TS, Pearlman NW, et al. Long-term follow-up and survival of patients following a recurrence of melanoma after a negative sentinel lymph node biopsy result. JAMA Surg. 2013;148(5):456–61.
Morton DL CL, Wong J. Intraoperative lymphatic mapping and selective lymphadenectomy: technical details of a new procedure for clinical stage I melanoma. In Paper presented at: 42nd Annual Meeting of the Society of Surgical Oncology, May 20–22, 1990, Washington, DC.
Essner R FL, Morton DL. A useful adjunct to intraoperative lymphatic mapping and selective lymphadenectomy in patients with clinical stage I melanoma. In Paper presented at: Annual Meeting of the Society of Surgical Oncology, March 17–20, 1994, Houston.
Bagaria SP, Faries MB, Morton DL. Sentinel node biopsy in melanoma: technical considerations of the procedure as performed at the John Wayne Cancer Institute. J Surg Oncol. 2010;101(8):669–76.
Morton DL, Thompson JF, Essner R, et al. Validation of the accuracy of intraoperative lymphatic mapping and sentinel lymphadenectomy for early-stage melanoma: a multicenter trial. Multicenter Selective Lymphadenectomy Trial Group. Ann Surg. 1999;230(4):453–63; discussion 463–5.
Dessureault S, Soong SJ, Ross MI, et al. Improved staging of node-negative patients with intermediate to thick melanomas (>1 mm) with the use of lymphatic mapping and sentinel lymph node biopsy. Ann Surg Oncol. 2001;8(10):766–70.
Yao KA, Hsueh EC, Essner R, Foshag LJ, Wanek LA, Morton DL. Is sentinel lymph node mapping indicated for isolated local and in-transit recurrent melanoma? Ann Surg. 2003;238(5):743–7.
Lin D, Franc BL, Kashani-Sabet M, Singer MI. Lymphatic drainage patterns of head and neck cutaneous melanoma observed on lymphoscintigraphy and sentinel lymph node biopsy. Head Neck. 2006;28(3):249–55.
Fincher TR, O’Brien JC, McCarty TM, et al. Patterns of drainage and recurrence following sentinel lymph node biopsy for cutaneous melanoma of the head and neck. Arch Otolaryngol Head Neck Surg. 2004;130(7):844–8.
Veenstra HJ, Klop WM, Speijers MJ, et al. Lymphatic drainage patterns from melanomas on the shoulder or upper trunk to cervical lymph nodes and implications for the extent of neck dissection. Ann Surg Oncol. 2012;19(12):3906–12.
Joosse A, Collette S, Suciu S, et al. Sex is an independent prognostic indicator for survival and relapse/progression-free survival in metastasized stage III to IV melanoma: a pooled analysis of five European organisation for research and treatment of cancer randomized controlled trials. J Clin Oncol. 2013;31(18):2337–46.
Joosse A, de Vries E, Eckel R, et al. Gender differences in melanoma survival: female patients have a decreased risk of metastasis. J Investig Dermatol. 2011;131(3):719–26.
Scoggins CR, Ross MI, Reintgen DS, et al. Gender-related differences in outcome for melanoma patients. Ann Surg. 2006;243(5):693–8; discussion 698–700.
Balch CM, Murad TM, Soong SJ, Ingalls AL, Richards PC, Maddox WA. Tumor thickness as a guide to surgical management of clinical stage I melanoma patients. Cancer. 1979;43(3):883–8.
Acknowledgments
This study was supported by the Dr. Miriam & Sheldon G Adelson Medical Research Foundation (Boston, MA), the Borstein Family Foundation (Los Angeles, CA), the John Wayne Cancer Institute Auxiliary (Santa Monica, CA), Mr. George W. Ogden, and Mr. John E. Connor. The authors are grateful to Gwen Berry for editorial assistance. This project was supported by grants R01 CA189163 from the National Cancer Institute. The content is solely the responsibility of the authors and does not necessarily represent the official view of the National Cancer Institute or the National Institutes of Health.
Disclosures
Dr. M. B. Faries has served as a consultant for Amgen Inc, Astellas Pharma Inc, and Genentech Inc. All other authors have no financial disclosure. Dr. D.Y. Lee is the Harold McAlister Charitable Foundation Fellow.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, D.Y., Huynh, K.T., Teng, A. et al. Predictors and Survival Impact of False-Negative Sentinel Nodes in Melanoma. Ann Surg Oncol 23, 1012–1018 (2016). https://doi.org/10.1245/s10434-015-4912-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4912-6