Abstract
Purpose
Axl, which is in the TAM family of receptor tyrosine kinases, and its ligand, growth arrest-specific gene 6 (Gas6), have been associated with worse prognoses after the surgical treatment of some types of cancers. We herein investigated the biological significance of the protein expression of Axl and Gas6 on the outcomes of patients with upper tract urothelial carcinoma (UTUC).
Methods
The protein expression of Axl and Gas6 was evaluated by immunohistochemistry, and their relationships with clinicopathological features were investigated in surgical specimens obtained from 161 patients who had been surgically treated for UTUC.
Results
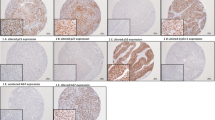
Axl labeling was strong in 67 of 161 (42 %) cases, while Gas6 labeling was strong in 72 of 161 (45 %) cases. The strong expression of Axl correlated with that of Gas6. A high pathological stage (p = 0.009), strong expression of Gas6 (p = 0.038), and strong expression of Axl (p = 0.016) were independent factors for predicting worse cancer-specific survival (CSS). In a subgroup analysis of patients with pT < 2 (N = 53), no significant difference in CSS was observed between patients weakly and strongly expressing Axl/Gas6. In contrast, a subgroup analysis of patients with pT ≥ 2 (N = 108) revealed that the expression levels of Axl and Gas6 correlated with CSS.
Conclusion
The protein expression of Axl and its ligand Gas6 may be a useful indicator for a worse clinical outcome in UTUC patients, especially patients with pT ≥ 2, who underwent radical nephroureterectomy.



Similar content being viewed by others
References
Oosterlinck W. Ureteral tumour: a specific upper urinary tract transitional cell carcinoma. Eur Urol. 2007;51(5):1164–5.
Oya M, Kikuchi E, Committee for Establishment of Clinical Practice Guideline for Management of Upper Tract Urothelial Carcinoma, Japanese Urological Association. Evidenced-based clinical practice guideline for upper tract urothelial carcinoma (summary–Japanese Urological Association, 2014 edition). Int J Urol. 2015;22(1):3–13.
Verma A, Warner SL, Vankayalapati H, Bearss DJ, Sharma S. Targeting Axl and Mer kinases in cancer. Mol Cancer Ther. 2011;10(10):1763–73.
Angelillo-Scherrer A, Burnier L, Flores N, et al. Role of Gas6 receptors in platelet signaling during thrombus stabilization and implications for antithrombotic therapy. J Clin Invest. 2005;115(2):237–46.
Angelillo-Scherrer A, Burnier L, Lambrechts D, et al. Role of Gas6 in erythropoiesis and anemia in mice. J Clin Invest. 2008;118(2):583–96.
Tjwa M, Bellido-Martin L, Lin Y, et al. Gas6 promotes inflammation by enhancing interactions between endothelial cells, platelets, and leukocytes. Blood. 2008;111(8):4096–105.
Bellosta P, Zhang Q, Goff SP, Basilico C. Signaling through the ARK tyrosine kinase receptor protects from apoptosis in the absence of growth stimulation. Oncogene. 1997;15(20):2387–97.
Loges S, Schmidt T, Tjwa M, et al. Malignant cells fuel tumor growth by educating infiltrating leukocytes to produce the mitogen Gas6. Blood. 2010;115(11):2264–73.
Sun W, Fujimoto J, Tamaya T. Coexpression of Gas6/Axl in human ovarian cancers. Oncology. 2004;66(6):450–57.
Green J, Ikram M, Vyas J, et al. Overexpression of the Axl tyrosine kinase receptor in cutaneous SCC-derived cell lines and tumours. Br J Cancer. 2006;94(10):1446–51.
Ou WB, Corson JM, Flynn DL, et al. AXL regulates mesothelioma proliferation and invasiveness. Oncogene. 2011;30(14):1643–52.
Gustafsson A, Martuszewska D, Johansson M, et al. Differential expression of Axl and Gas6 in renal cell carcinoma reflecting tumor advancement and survival. Clin Cancer Res. 2009;15(14):4742–9.
Hutterer M, Knyazev P, Abate A, et al. Axl and growth arrest-specific gene 6 are frequently overexpressed in human gliomas and predict poor prognosis in patients with glioblastoma multiforme. Clin Cancer Res. 2008;14(1):130–38.
Epstein JI, Amin MB, Reuter VR, Mostofi FK. The World Health Organization/International Society of Urological Pathology consensus classification of urothelial (transitional cell) neoplasms of the urinary bladder. Bladder Consensus Conference Committee. Am J Surg Pathol. 1998;22(12):1435–48.
Ishikawa M, Sonobe M, Nakayama E, et al. Higher expression of receptor tyrosine kinase Axl, and differential expression of its ligand, Gas6, predict poor survival in lung adenocarcinoma patients. Ann Surg Oncol. 2013;20(Suppl 3):S467–76.
Ye X, Li Y, Stawicki S, et al. An anti-Axl monoclonal antibody attenuates xenograft tumor growth and enhances the effect of multiple anticancer therapies. Oncogene. 2010;29(38):5254–64.
Linger RM, Keating AK, Earp HS, Graham DK. TAM receptor tyrosine kinases: biologic functions, signaling, and potential therapeutic targeting in human cancer. Adv Cancer Res. 2008;100:35–83.
Vajkoczy P, Knyazev P, Kunkel A, et al. Dominant-negative inhibition of the Axl receptor tyrosine kinase suppresses brain tumor cell growth and invasion and prolongs survival. Proc Natl Acad Sci USA. 2006;103(15):5799–804.
Wu CW, Li AF, Chi CW, et al. Clinical significance of AXL kinase family in gastric cancer. Anticancer Res. 2002;22(2B):1071–8.
Koorstra JB, Karikari CA, Feldmann G, et al. The Axl receptor tyrosine kinase confers an adverse prognostic influence in pancreatic cancer and represents a new therapeutic target. Cancer Biol Ther. 2009;8(7):618–26.
Yauch RL, Januario T, Eberhard DA, et al. Epithelial versus mesenchymal phenotype determines in vitro sensitivity and predicts clinical activity of erlotinib in lung cancer patients. Clin Cancer Res. 2005;11(24 Pt 1):8686–98.
Ben-Batalla I, Schultze A, Wroblewski M, et al. Axl, a prognostic and therapeutic target in acute myeloid leukemia mediates paracrine crosstalk of leukemia cells with bone marrow stroma. Blood. 2013;122(14):2443–52.
Yeh CY, Shin SM, Yeh HH, et al. Transcriptional activation of the Axl and PDGFR-alpha by c-Met through a ras- and Src-independent mechanism in human bladder cancer. BMC Cancer. 2011;11:139.
Holland SJ, Pan A, Franci C, et al. R428, a selective small molecule inhibitor of Axl kinase, blocks tumor spread and prolongs survival in models of metastatic breast cancer. Cancer Res. 2010;70(4):1544–54.
Wang X, Saso H, Iwamoto T, et al. TIG1 promotes the development and progression of inflammatory breast cancer through activation of Axl kinase. Cancer Res. 2013;73(21):6516–25.
Kurzrock R, Sherman SI, Ball DW, et al. Activity of XL184 (Cabozantinib), an oral tyrosine kinase inhibitor, in patients with medullary thyroid cancer. J Clin Oncol. 2011;29(19):2660–66.
Mayer EL, Krop IE. Advances in targeting SRC in the treatment of breast cancer and other solid malignancies. Clin Cancer Res. 2010;16(14):3526–32.
Mosse YP, Lim MS, Voss SD, et al. Safety and activity of crizotinib for paediatric patients with refractory solid tumours or anaplastic large-cell lymphoma: a Children’s Oncology Group phase 1 consortium study. Lancet Oncol. 2013;14(6):472–80.
Tibes R, Fine G, Choy G, et al. A phase I, first-in-human dose-escalation study of amuvatinib, a multi-targeted tyrosine kinase inhibitor, in patients with advanced solid tumors. Cancer Chemother Pharmacol. 2013;71(2):463–71.
Sayan AE, Stanford R, Vickery R, et al. Fra-1 controls motility of bladder cancer cells via transcriptional upregulation of the receptor tyrosine kinase AXL. Oncogene. 2012;31(12):1493–503.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hattori, S., Kikuchi, E., Kosaka, T. et al. Relationship Between Increased Expression of the Axl/Gas6 Signal Cascade and Prognosis of Patients with Upper Tract Urothelial Carcinoma. Ann Surg Oncol 23, 663–670 (2016). https://doi.org/10.1245/s10434-015-4848-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4848-x