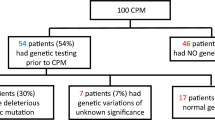
Abstract
The use of both bilateral prophylactic mastectomy and contralateral prophylactic mastectomy (CPM) has increased significantly during the last decade. Various risk models have been developed to identify patients at increased risk for breast cancer. The indications for bilateral prophylactic mastectomy for patients without a diagnosis of breast cancer include high risk from mutation in BRCA or other breast cancer predisposition gene, very strong family history with no identifiable mutation, and high risk based on breast histology. Additionally, the use of CPM has more than doubled in the last decade, and this increase is noted among all stages of breast cancer, even in patients with ductal carcinoma in situ (stage 0). The risk of contralateral breast cancer often is overestimated by both patients and physicians. Nevertheless, specific risk factors are associated with an increased risk of contralateral breast cancer, including BRCA or other genetic mutation, young age at diagnosis, lobular histology, family history, and prior chest wall irradiation. Although CPM reduces the incidence of contralateral breast cancer, the effect on disease-free survival and, more importantly, overall survival is questionable and underscored by the fact that the reason most patients choose CPM is to achieve “peace of mind.” Newer and effective reconstructive options have made the procedure more attractive. This panel addresses the indications and rationale for bilateral prophylactic mastectomy and CPM, the decision-making process by patients, and ethical considerations. Changes in the physician–patient relationship during the past few decades have altered the approach, and ethical considerations are paramount in addressing these issues.
Similar content being viewed by others
References
Reiner AS, John EM, Brooks JD, et al. Risk of asynchronous contralateral breast cancer in noncarriers of BRCA1 and BRCA2 mutations with a family history of breast cancer: a report from the Women’s Environmental Cancer and Radiation Epidemiology Study. J Clin Oncol. 2013;31:433–9.
Fisher B, Costantino J, Redmond C, Poisson R, et al. A randomized clinical trial evaluating tamoxifen in the treatment of patients with node-negative breast cancer who have estrogen-receptor-positive tumors. N Engl J Med. 1989;320:479–84.
Metcalfe K, Gershman S, Lynch, HT. Predictors of contralateral breast cancer in BRCA1 and BRCA2 mutation carriers. Br J Cancer. 2011;104:1384–92.
Forbes JF, Cuzick J, Buzdar A, Howell A, Tobias JS, Baum M. Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 100-month analysis of the ATAC trial. Lancet Oncol. 2008;9:45–53.
Nichols HB, Berrington de Gonzalez A, Lacey JV Jr, Rosenberg PS, Anderson WF. Declining incidence of contralateral breast cancer in the United States from 1975 to 2006. J Clin Oncol. 2011;29:1564–9.
Graeser MK, Engel C, Rhiem K. Contralateral breast cancer risk in BRCA1 and BRCA2 mutation carriers. J Clin Oncol. 2009;27:5887–92.
Bedrosian I, Hu CY, Chang GJ. Population-based study of contralateral prophylactic mastectomy and survival outcomes of breast cancer patients. J Natl Cancer Inst. 2010;102:401–9.
Brewster AM, Bedrosian I, Parker PA, et al. Association between contralateral prophylactic mastectomy and breast cancer outcomes by hormone receptor status. Cancer. 2012;118:5637–43.
Portschy PR, Kuntz KM, Tuttle TM. Survival outcomes after contralateral prophylactic mastectomy: a decision analysis. J Natl Cancer Inst. 2014;106:1–7.
Herrinton LJ, Barlow WE, Yu O, et al. Efficacy of prophylactic mastectomy in women with unilateral breast cancer: a cancer research network project. J Clin Oncol. 2005;23:4275–86.
Tuttle TM, Jarosek S, Habermann EB, et al. Increasing rates of contralateral prophylactic mastectomy among patients with ductal carcinoma in situ. J Clin Oncol. 2009;27:1362–7.
Hawley ST, Jagsi R, Morrow M, Janz NK, Hamilton A, Graff JJ, Katz SJ. Social and clinical determinants of contralateral prophylactic mastectomy. JAMA Surg. 2014;149:582–9.
Katz SJ, Morrow M. Contralateral prophylactic mastectomy for breast cancer: addressing peace of mind. JAMA. 2013;310:793–4.
Hawley ST, Fagerlin A, Janz NK, Katz SJ. Racial/ethnic differences in knowledge about the risks and benefits of breast cancer treatment: does it matter where you go? Health Serv Res. 2008;43:1366–87.
Katz SJ, Morrow M. Addressing overtreatment in breast cancer: the doctors’ dilemma. Cancer. 2013;119:3584–8.
Oken D. What to tell cancer patients: a study of medical attitudes. JAMA. 1961;175:1120–8.
Novack DH, Plumer R, Smith RL, Ochitill H, Morrow GR, Bennett JM. Changes in physicians’ attitudes toward telling the cancer patient. JAMA. 1979;241:897–900.
Tung N, Battelli C, Allen B, et al. Frequency of mutations in individuals with breast cancer referred for BRCA1 and BRCA2 testing using next-generation sequencing with a 25-gene panel. Cancer. 2015; 121:25–33. http://www.ncbi.nlm.nih.gov/pubmed/25186627.
LaDuca H, Stuenkel AJ, Dolinsky JS, et al. Utilization of multigene panels in hereditary cancer predisposition testing: analysis of more than 2000 patients. Genet Med. 2014;16:830–7. http://www.ncbi.nlm.nih.gov/pubmed/24763289.
National Comprehensive Cancer Network. Genetic/familial high-risk assessment: breast and ovarian. In: NCCN clinical practice guidelines in oncology v12015. vol. 1. Fort Washington; 2015.
Cybulski C, Wokolorczyk D, Jakubowska A, et al. Risk of breast cancer in women with a CHEK2 mutation with and without a family history of breast cancer. J Clin Oncol. 2011;29:3747–52.
Goldgar DE, Healey S, Dowty JG, et al. Rare variants in the ATM gene and risk of breast cancer. BCR Breast Cancer Res. 2011;13:R73.
Bogdanova N, Cybulski C, Bermisheva M, et al. A nonsense mutation (E1978X) in the ATM gene is associated with breast cancer. Breast Cancer Res Treat. 2009;118:207–11. http://www.ncbi.nlm.nih.gov/pubmed/18807267.
Hu ES, Pusic AL, Waljee JF, Kuhn L, Hawley ST, Wilkins E, Alderman AK. Patient-reported aesthetic satisfaction with breast reconstruction during the long-term survivorship period. Plast Reconstr Surg. 2009;124:1–8.
Atisha DM, Rushing CN, Samsa GP, Locklear TD, Cox CE, Shelley Hwang E, Zenn MR, Pusic AL, Abernethy AP. A national snapshot of satisfaction with breast cancer procedures. Ann Surg Oncol. 2015;22:361–9.
Albornoz CR, Bach PB, Mehrara BJ, Disa JJ, Pusic AL, McCarthy CM, Cordeiro PG, Matros E. A paradigm shift in U.S. breast reconstruction: increasing implant rates. Plast Reconstr Surg. 2013;131:15–23.
Albornoz CR, Bach PB, Pusic AL, McCarthy CM, Mehrara BJ, Disa JJ, Cordeiro PG, Matros E. The influence of sociodemographic factors and hospital characteristics on the method of breast reconstruction, including microsurgery: a U.S. population-based study. Plast Reconstr Surg. 2012;129:1071–9.
Han E, Johnson N, Glissmeyer M, Wagie T, Carey B, DelaMelena T, Nelson J. Increasing incidence of bilateral mastectomies: the patient perspective. Am J Surg. 2011;201:615–8.
Cemal Y, Albornoz CR, Disa JJ, McCarthy CM, Mehrara BJ, Pusic AL, Cordeiro PG, Matros E. A paradigm shift in U.S. breast reconstruction: part 2. The influence of changing mastectomy patterns on reconstructive rate and method. Plast Reconstr Surg. 2013;131:320e–6e.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Angelos, P., Bedrosian, I., Euhus, D.M. et al. Contralateral Prophylactic Mastectomy: Challenging Considerations for the Surgeon. Ann Surg Oncol 22, 3208–3212 (2015). https://doi.org/10.1245/s10434-015-4758-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4758-y