Abstract
Background
Studies to identify preoperative prognostic variables for pancreatic neuroendocrine tumor (PNET) have been inconclusive. Specifically, the prevalence and prognostic significance of radiographic calcifications in these tumors remains unclear.
Methods
From 1998 to 2009, a total of 110 patients with well-differentiated PNET underwent surgical resection at our institution. Synchronous liver metastases present in 31 patients (28%) were addressed surgically with curative intent. Patients with high-grade PNET were excluded. The presence of calcifications in the primary tumor on preoperative computed tomography was recorded and correlated with clinicopathologic variables and overall survival.
Results
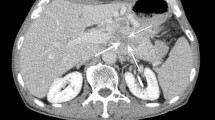
Calcifications were present in 16% of patients and were more common in gastrinomas and glucagonomas (50%), but never encountered in insulinomas. Calcified tumors were larger (median size 4.5 vs. 2.3 cm, P = 0.04) and more commonly associated with lymph node metastasis (75 vs. 35%, P = 0.01), synchronous liver metastasis (62 vs. 21%, P < 0.01), and intermediate tumor grade (80 vs. 31%, P < 0.01). On multivariate analysis of factors available preoperatively, calcifications (P = 0.01) and size (P < 0.01) remained independent predictors of lymph node metastasis. Overall survival after resection was significantly worse in the presence of synchronous liver metastasis (5-year, 64 vs. 86%, P = 0.04), but not in the presence of radiographic calcifications.
Conclusions
Calcifications on preoperative computed tomography correlate with intermediate grade and lymph node metastasis in well-differentiated PNET. This information is available preoperatively and supports the routine dissection of regional lymph nodes through formal pancreatectomy rather than enucleation in calcified PNET.


Similar content being viewed by others
References
Klimstra DS, Modlin IR, Coppola D, et al. The pathologic classification of neuroendocrine tumors: a review of nomenclature, grading, and staging systems. Pancreas. 2010;39:707–12.
Fitzgerald TL, Hickner ZJ, Schmitz M, et al. Changing incidence of pancreatic neoplasms: a 16-year review of statewide tumor registry. Pancreas. 2008;37:134–8.
Buchanan KD, Johnston CF, O’Hare MM, et al. Neuroendocrine tumors. A European view. Am J Med. 1986;81:14–22.
Eriksson B, Oberg K, Skogseid B. Neuroendocrine pancreatic tumors. Clinical findings in a prospective study of 84 patients. Acta Oncol. 1989;28:373–7.
Halfdanarson TR, McWilliams RR, Donohue JH, et al. A single-institution experience with 491 cases of small bowel adenocarcinoma. Am J Surg. 2010;199:797–803.
Halfdanarson TR, Rabe KG, Rubin J, et al. Pancreatic neuroendocrine tumors (PNETs): incidence, prognosis and recent trend toward improved survival. Ann Oncol. 2008;19:1727–33.
Lepage C, Bouvier AM, Phelip JM, et al. Incidence and management of malignant digestive endocrine tumours in a well defined French population. Gut. 2004;53:549–53.
Yao JC, Hassan M, Phan A, et al. One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. 2008;26:3063–72.
Norton JA. Neuroendocrine tumors of the pancreas and duodenum. Curr Probl Surg. 1994;31:77–156.
Norton JA, Warren RS, Kelly MG, et al. Aggressive surgery for metastatic liver neuroendocrine tumors. Surgery. 2003;134:1057–63.
Norton JA. Surgery for primary pancreatic neuroendocrine tumors. J Gastrointest Surg. 2006;10:327–31.
Schurr PG, Strate T, Rese K, et al. Aggressive surgery improves long-term survival in neuroendocrine pancreatic tumors: an institutional experience. Ann Surg. 2007;245:273–81.
Akerström G, Hellman P. Surgery on neuroendocrine tumours. Best Pract Res Clin Endocrinol Metab. 2007;21:87–109.
Pitt SC, Pitt HA, Baker MS, et al. Small pancreatic and periampullary neuroendocrine tumors: resect or enucleate? J Gastrointest Surg. 2009;13:1692–8.
Ballian N, Loeffler AG, Rajamanickam V, et al. A simplified prognostic system for resected pancreatic neuroendocrine neoplasms. HPB. 2009;11:422–8.
Bilimoria KY, Tomlinson JS, Merkow RP, et al. Clinicopathologic features and treatment trends of pancreatic neuroendocrine tumors: analysis of 9,821 patients. J Gastrointest Surg. 2007;11:1460–9.
Bloomston M, Muscarella P, Shah MH, et al. Cytoreduction results in high perioperative mortality and decreased survival in patients undergoing pancreatectomy for neuroendocrine tumors of the pancreas. J Gastrointest Surg. 2006;10:1361–70.
Ekeblad S, Skogseid B, Dunder K, et al. Prognostic factors and survival in 324 patients with pancreatic endocrine tumor treated at a single institution. Clin Cancer Res. 2008;14:7798–803.
Ferrone CR, Tang LH, Tomlinson J, et al. Determining prognosis in patients with pancreatic endocrine neoplasms: can the WHO classification system be simplified? J Clin Oncol. 2007;25:5609–15.
Fischer L, Kleeff J, Esposito I, et al. Clinical outcome and long-term survival in 118 consecutive patients with neuroendocrine tumours of the pancreas. Br J Surg. 2008;95:627–35.
Franko J, Feng W, Yip L, et al. Non-functional neuroendocrine carcinoma of the pancreas: incidence, tumor biology, and outcomes in 2,158 patients. J Gastrointest Surg. 2010;14:541–8.
Hochwald SN, Zee S, Conlon KC, et al. Prognostic factors in pancreatic endocrine neoplasms: an analysis of 136 cases with a proposal for low-grade and intermediate-grade groups. J Clin Oncol. 2002;20:2633–42.
Ito H, Abramson M, Ito K, et al. Surgery and staging of pancreatic neuroendocrine tumors: a 14-year experience. J Gastrointest Surg. 2010;14:891–8.
Martin RCG, Kooby DA, Weber SM, et al. Analysis of 6,747 pancreatic neuroendocrine tumors for a proposed staging system. J Gastrointest Surg. 2011;15:175–83.
Bilimoria KY, Talamonti MS, Tomlinson JS, et al. Prognostic score predicting survival after resection of pancreatic neuroendocrine tumors. Ann Surg. 2008;247:490–500.
Gullo L, Migliori M, Falconi M, et al. Nonfunctioning pancreatic endocrine tumors: a multicenter clinical study. Am J Gastroenterol. 2003;98:2435–9.
Phan GQ, Yeo CJ, Hruban RH, et al. Surgical experience with pancreatic and peripancreatic neuroendocrine tumors: review of 125 patients. J Gastrointest Surg. 1998;2:473–82.
Tomassetti P, Campana D, Piscitelli L, et al. Endocrine pancreatic tumors: factors correlated with survival. Ann Oncol. 2005;16:1806–10.
Imhof H, Frank P. Pancreatic calcifications in malignant islet cell tumors. Radiology. 1977;122:333–7.
Rodallec M, Vilgrain V, Couvelard A, et al. Endocrine pancreatic tumours and helical CT: contrast enhancement is correlated with microvascular density, histoprognostic factors and survival. Pancreatology. 2006;6:77–85.
Norton JA. Intraoperative methods to stage and localize pancreatic and duodenal tumors. Ann Oncol. 1999;10(Suppl 4):182–4.
Norton JA, Alexander HR, Fraker DL, et al. Does the use of routine duodenotomy (DUODX) affect rate of cure, development of liver metastases, or survival in patients with Zollinger-Ellison syndrome? Ann Surg. 2004;239:617–25.
Norton JA, Doppman JL, Jensen RT. Curative resection in Zollinger-Ellison syndrome. Results of a 10-year prospective study. Ann Surg. 1992;215:8–18.
Norton JA, Fraker DL, Alexander HR, et al. Surgery to cure the Zollinger-Ellison syndrome. N Engl J Med. 1999;341:635–44.
Sugg SL, Norton JA, Fraker DL, et al. A prospective study of intraoperative methods to diagnose and resect duodenal gastrinomas. Ann Surg. 1993;218:138–44.
Arnold WS, Fraker DL, Alexander HR, et al. Apparent lymph node primary gastrinoma. Surgery. 1994;116:1123–9.
Norton JA, Alexander HR, Fraker DL, et al. Possible primary lymph node gastrinoma: occurrence, natural history, and predictive factors: a prospective study. Ann Surg. 2003;237:650–7.
Norton JA, Doherty GM, Fraker DL, et al. Surgical treatment of localized gastrinoma within the liver: a prospective study. Surgery. 1998;124:1145–52.
Norton JA, Kivlen M, Li M, et al. Morbidity and mortality of aggressive resection in patients with advanced neuroendocrine tumors. Arch Surg. 2003;138:859–66.
Rindi G, Klöppel G, Alhman H, et al. TNM staging of foregut (neuro)endocrine tumors: a consensus proposal including a grading system. Virchows Archiv. 2006;449:395–401.
Hiramoto JS, Feldstein VA, LaBerge JM, et al. Intraoperative ultrasound and preoperative localization detects all occult insulinomas. Arch Surg. 2001;136:1020–5.
Fishbeyn VA, Norton JA, Benya RV, et al. Assessment and prediction of long-term cure in patients with the Zollinger-Ellison syndrome: the best approach. Ann Intern Med. 1993;119:199–206.
Roy PK, Venzon DJ, Feigenbaum KM, et al. Gastric secretion in Zollinger-Ellison syndrome. Correlation with clinical expression, tumor extent and role in diagnosis—a prospective NIH study of 235 patients and a review of 984 cases in the literature. Medicine (Baltimore). 2001;80:189–222.
Roy PK, Venzon DJ, Shojamanesh H, et al. Zollinger-Ellison syndrome. Clinical presentation in 261 patients. Medicine (Baltimore). 2000;79:379–411.
Norton JA, Kahn CR, Schiebinger R, et al. Amino acid deficiency and the skin rash associated with glucagonoma. Ann Intern Med. 1979;91:213–5.
Alexiev BA, Darwin PE, Goloubeva O, et al. Proliferative rate in endoscopic ultrasound fine-needle aspiration of pancreatic endocrine tumors: correlation with clinical behavior. Cancer. 2009;117:40–5.
Jarufe NP, Coldham C, Orug T, et al. Neuroendocrine tumours of the pancreas: predictors of survival after surgical treatment. Dig Surg. 2005;22:157–62.
Kazanjian KK, Reber HA, Hines OJ. Resection of pancreatic neuroendocrine tumors: results of 70 cases. Arch Surg. 2006;141:765–9.
Sarmiento JM, Farnell MB, Que FG, et al. Pancreaticoduodenectomy for islet cell tumors of the head of the pancreas: long-term survival analysis. World J Surg. 2002;26:1267–71.
Vagefi PA, Razo O, Deshpande V, et al. Evolving patterns in the detection and outcomes of pancreatic neuroendocrine neoplasms: the Massachusetts General Hospital experience from 1977 to 2005. Arch Surg. 2007;142:347–54.
Chu QD, Hill HC, Douglass HO, et al. Predictive factors associated with long-term survival in patients with neuroendocrine tumors of the pancreas. Ann Surg Oncol. 2002;9:855–62.
Panzuto F, Nasoni S, Falconi M, et al. Prognostic factors and survival in endocrine tumor patients: comparison between gastrointestinal and pancreatic localization. Endocr Relat Cancer. 2005;12:1083–92.
Solorzano CC, Lee JE, Pisters PW, et al. Nonfunctioning islet cell carcinoma of the pancreas: survival results in a contemporary series of 163 patients. Surgery. 2001;130:1078–85.
Edge SB, Byrd DR, Compton CC, Fritz AG, Green FL, Trotti A. American Joint Committee on Cancer (AJCC) staging manual. 7th ed. New York: Springer-Verlag, 2010.
Bosman F, Carneiro F, Hruban R, et al. WHO classification of tumours of the digestive system. Lyon, France: IARC Press, 2010.
Keiser HR, Beaven MA, Doppman J, et al. Sipple’s syndrome: medullary thyroid carcinoma, pheochromocytoma, and parathyroid disease. Studies in a large family. NIH conference. Ann Intern Med. 1973;78:561–79.
Lee S, Shin JH, Han BK, et al. Medullary thyroid carcinoma: comparison with papillary thyroid carcinoma and application of current sonographic criteria. Am J Roentgenol. 2010;194:1090–4.
Buckley JA, Fishman EK. CT evaluation of small bowel neoplasms: spectrum of disease. Radiographics. 1998;18:379–92.
Pantongrag-Brown L, Buetow PC, Carr NJ, et al. Calcification and fibrosis in mesenteric carcinoid tumor: CT findings and pathologic correlation. AJR Am J Roentgenol. 1995;164:387–91.
Magid D, Siegelman SS, Eggleston JC, et al. Pulmonary carcinoid tumors: CT assessment. J Comput Assist Tomogr. 1989;13:244–7.
Grewal RG, Austin JH. CT demonstration of calcification in carcinoma of the lung. J Comput Assist Tomogr. 1994;18:867–71.
Chong S, Lee KS, Chung MJ, et al. Neuroendocrine tumors of the lung: clinical, pathologic, and imaging findings. Radiographics. 2006;26:41–57.
Akata S, Okada S, Maeda J, et al. Computed tomographic findings of large cell neuroendocrine carcinoma of the lung. Clin Imaging. 2007;31:379–84.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Poultsides, G.A., Huang, L.C., Chen, Y. et al. Pancreatic Neuroendocrine Tumors: Radiographic Calcifications Correlate with Grade and Metastasis. Ann Surg Oncol 19, 2295–2303 (2012). https://doi.org/10.1245/s10434-012-2305-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2305-7