Abstract
Background
Studies have suggested that HOXB9 expression in breast cancer cells promotes cellular invasiveness, metastatic ability, and tumor neovascularization in the surrounding tissue in in vitro and in vivo assays. These findings imply that HOXB9 overexpression may alter tumor-specific cell fates and the tumor stromal microenvironment, contributing to breast cancer progression. The objective of this study was to analyze whether these results could be applied to clinical practice.
Methods
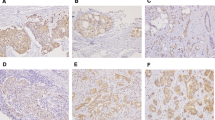
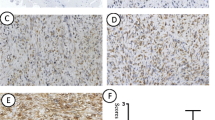
A total of 141 consecutive, invasive ductal carcinoma patients who underwent surgical treatment were examined. Immunohistochemical staining was performed to evaluate the expression of HOXB9, Ki-67, CD31, and CD34, and the association of tumor proliferation and angiogenesis with HOXB9 expression was analyzed.
Results
Of the 141 tumor specimens immunostained for HOXB9, 69 (48.9%) stained positive. Larger primary tumor size, hormone receptor negativity, HER2 positivity, higher nuclear grade, and number of pathologic nodal metastases were significant variables associated with HOXB9 expression. Notably, 12 (92.3%) of 13 triple-negative breast cancer cases showed HOXB9 expression. Disease-free survival and overall survival were significantly different between the HOXB9-positive and HOXB9-negative groups (hazard ratio 20.714, P = 0.001; and hazard ratio 9.206, P = 0.003, respectively). Multivariate analysis indicated that HOXB9 expression was the only independent prognostic factor for disease-free survival (hazard ratio 15.532, P = 0.009). HOXB9-positive tumors showed a significant increase in the number of vasculature and the Ki-67 ratio compared with HOXB9-negative tumors.
Conclusions
HOXB9 expression, which promotes tumor proliferation and angiogenesis, is a significant prognostic factor in breast cancer.



Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74–108.
Goldhirsch A, Ingle JN, Gelber RD, Coates AS, Thurlimann B, Senn HJ. Thresholds for therapies: highlights of the St Gallen international expert consensus on the primary therapy of early breast cancer 2009. Ann Oncol. 2009;20:1319–29.
Abate-Shen C. Deregulated homeobox gene expression in cancer: cause or consequence? Nat Rev Cancer. 2002;2:777–85.
Hatzis P, van der Flier LG, van Driel MA, et al. Genome-wide pattern of TCF7L2/TCF4 chromatin occupancy in colorectal cancer cells. Mol Cell Biol. 2008;28:2732–44.
Calvo R, West J, Franklin W, et al. Altered HOX and WNT7A expression in human lung cancer. Proc Natl Acad Sci USA. 2000;97:12776–81.
Nguyen DX, Chiang AC, Zhang XH, et al. WNT/TCF signaling through LEF1 and HOXB9 mediates lung adenocarcinoma metastasis. Cell. 2009;138:51–62.
Hayashida T, Takahashi F, Chiba N, et al. HOXB9, a gene overexpressed in breast cancer, promotes tumorigenicity and lung metastasis. Proc Natl Acad Sci USA. 2010;107:1100–5.
Folkman J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat Med. 1995;1:27–31.
Folkman J. Role of angiogenesis in tumor growth and metastasis. Semin Oncol. 2002;29(6 Suppl. 16):15–8.
Linderholm BK, Hellborg H, Johansson U, et al. Significantly higher levels of vascular endothelial growth factor (VEGF) and shorter survival times for patients with primary operable triple-negative breast cancer. Ann Oncol. 2009;20:1639–46.
Allred DC, Harvey JM, Berardo M, Clark GM. Prognostic and predictive factors in breast cancer by immunohistochemical analysis. Mod Pathol. 1998;11:155–68.
Harvey JM, Clark GM, Osborne CK, Allred DC. Estrogen receptor status by immunohistochemistry is superior to the ligand-binding assay for predicting response to adjuvant endocrine therapy in breast cancer. J Clin Oncol. 1999;17:1474–81.
Nielsen TO, Hsu FD, Jensen K, et al. Immunohistochemical and clinical characterization of the basal-like subtype of invasive breast carcinoma. Clin Cancer Res. 2004;10:5367–74.
al-Sader MH, Doyle E, Kay EW, et al. Proliferation indexes—a comparison between cutaneous basal and squamous cell carcinomas. J Clin Pathol. 1996;49:549–51.
Cattoretti G, Becker MH, Key G, et al. Monoclonal antibodies against recombinant parts of the Ki-67 antigen (MIB 1 and MIB 3) detect proliferating cells in microwave-processed formalin-fixed paraffin sections. J Pathol. 1992;168:357–63.
McCormick D, Chong H, Hobbs C, Datta C, Hall PA. Detection of the Ki-67 antigen in fixed and wax-embedded sections with the monoclonal antibody MIB1. Histopathology. 1993;22:355–60.
McCormick D, Yu C, Hobbs C, Hall PA. The relevance of antibody concentration to the immunohistological quantification of cell proliferation–associated antigens. Histopathology. 1993;22:543–7.
Bluff JE, Menakuru SR, Cross SS, et al. Angiogenesis is associated with the onset of hyperplasia in human ductal breast disease. Br J Cancer. 2009;101:666–72.
Davies G, Salter J, Hills M, Martin LA, Sacks N, Dowsett M. Correlation between cyclooxygenase-2 expression and angiogenesis in human breast cancer. Clin Cancer Res. 2003;9:2651–6.
Goodman WA, Levine AD, Massari JV, Sugiyama H, McCormick TS, Cooper KD. IL-6 signaling in psoriasis prevents immune suppression by regulatory T cells. J Immunol. 2009;183:3170–6.
Zhou D, Cheng SQ, Ji HF, et al. Evaluation of protein pigment epithelium–derived factor (PEDF) and microvessel density (MVD) as prognostic indicators in breast cancer. J Cancer Res Clin Oncol. 2010;136:1719–27.
Bettencourt MC, Bauer JJ, Sesterhenn IA, Connelly RR, Moul JW. CD34 immunohistochemical assessment of angiogenesis as a prognostic marker for prostate cancer recurrence after radical prostatectomy. J Urol. 1998;160:459–65.
Vartanian RK, Weidner N. Correlation of intratumoral endothelial cell proliferation with microvessel density (tumor angiogenesis) and tumor cell proliferation in breast carcinoma. Am J Pathol. 1994;144:1188–94.
Micalizzi DS, Christensen KL, Jedlicka P, et al. The Six1 homeoprotein induces human mammary carcinoma cells to undergo epithelial–mesenchymal transition and metastasis in mice through increasing TGF-beta signaling. J Clin Invest. 2009;119:2678–90.
Yu M, Smolen GA, Zhang J, et al. A developmentally regulated inducer of EMT, LBX1, contributes to breast cancer progression. Genes Dev. 2009;23:1737–42.
Sarrió D, Rodriguez-Pinilla SM, Hardisson D, Cano A, Moreno-Bueno G, Palacios J. Epithelial–mesenchymal transition in breast cancer relates to the basal-like phenotype. Cancer Res. 2008;68:989–97.
Ganapathy V, Ge R, Grazioli A, et al. Targeting the transforming growth factor-beta pathway inhibits human basal-like breast cancer metastasis. Mol Cancer. 2010;9:122.
Jain RK. Molecular regulation of vessel maturation. Nat Med. 2003;9:685–93.
Millanta F, Lazzeri G, Vannozzi I, Viacava P, Poli A. Correlation of vascular endothelial growth factor expression to overall survival in feline invasive mammary carcinomas. Vet Pathol. 2002;39:690–6.
Toi M, Kashitani J, Tominaga T. Tumor angiogenesis is an independent prognostic indicator in primary breast carcinoma. Int J Cancer. 1993;55:371–4.
Weidner N, Semple JP, Welch WR, Folkman J. Tumor angiogenesis and metastasis—correlation in invasive breast carcinoma. N Engl J Med. 1991;324:1–8.
Uzzan B, Nicolas P, Cucherat M, Perret GY. Microvessel density as a prognostic factor in women with breast cancer: a systematic review of the literature and meta-analysis. Cancer Res. 2004;64:2941–55.
Borg A, Tandon AK, Sigurdsson H, et al. HER-2/neu amplification predicts poor survival in node-positive breast cancer. Cancer Res. 1990;50:4332–7.
Carter CL, Allen C, Henson DE. Relation of tumor size, lymph node status, and survival in 24,740 breast cancer cases. Cancer. 1989;63:181–7.
Fisher B, Bauer M, Wickerham DL, et al. Relation of number of positive axillary nodes to the prognosis of patients with primary breast cancer. An NSABP update. Cancer. 1983;52:1551–7.
Ragaz J, Jackson SM, Le N, et al. Adjuvant radiotherapy and chemotherapy in node-positive premenopausal women with breast cancer. N Engl J Med. 1997;337:956–62.
Acknowledgment
The authors thank Chisato Saito, Hiroshi Okazaki, and Kazuhiro Miyao for their technical support and helpful discussions. This research was supported by the Ministry of Education, Culture, Sports, Science and Technology Grants-in-Aid for Scientific Research [B] (21791260 and 23791499 to T.H.) and [C] (22591442 to H.J., 21591677 to M.T.), and a Grant-in-Aid from the Global Center of Excellence Program entitled “Education and Research Center for Stem Cell Medicine” (Keio University).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Seki, H., Hayashida, T., Jinno, H. et al. HOXB9 Expression Promoting Tumor Cell Proliferation and Angiogenesis Is Associated with Clinical Outcomes in Breast Cancer Patients. Ann Surg Oncol 19, 1831–1840 (2012). https://doi.org/10.1245/s10434-012-2295-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-012-2295-5