Abstract
Purpose
The circulating cell-free DNA (cfDNA) in plasma has been reported to be a marker of cancer detection. The aim of this study was to investigate whether the cfDNA has a role as response biomarker in patients receiving preoperative chemoradiotherapy (CRT) for rectal cancer.
Methods
Sixty-seven patients (median age 61 years; male/female 42/25) who underwent CRT for rectal cancer were evaluated. After tumor regression grade (TRG) classification was made, the patients were classified as having disease that responded (TRG 1–2) and that did not respond (TRG 3–5) to therapy. Plasma samples were obtained from patients before and after CRT. The cfDNA levels were analyzed by quantitative real-time polymerase chain reaction of β-globin. On the basis of the Alu repeats, the cfDNA was considered as either total (fragments of 115 bp, Alu 115) or tumoral (fragments of 247 bp, Alu 247). The association between the pre- or post-CRT levels and between variations during CRT of the Alu 247, Alu 115 repeat, and Alu 247/115 ratio (cfDNA integrity index) and the pathologic tumor response was analyzed.
Results
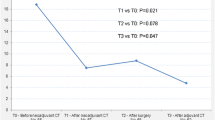
The baseline levels of cfDNA were not associated with tumor response. The post-CRT levels of the cfDNA integrity index were significantly lower in responsive compared to nonresponsive disease (P = 0.0009). Both the median value of the Alu 247 repeat and the cfDNA integrity index decreased after CRT in disease that responded to therapy (P < 0.005 and P < 0.005, respectively) compared to disease that did not respond to therapy (P = 0.83 and P = 0.726, respectively). The results of the multivariable logistic regression analysis showed that only the cfDNA integrity index was significantly and independently associated with tumor response to treatment.
Conclusions
The plasma levels of the longer fragments (Alu 247) of cfDNA and the cfDNA integrity index are promising markers to predict tumor response after preoperative CRT for rectal cancer.


Similar content being viewed by others
References
Bosset JF, Collette L, Calais G, et al. Chemotherapy with preoperative radiotherapy in rectal cancer. N Engl J Med. 2006;355:1114–23.
Sauer R, Becker H, Hohenberger W, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med. 2004;351:1731–40.
Gerard JP, Conroy T, Bonnetain F, et al. Preoperative radiotherapy with or without concurrent fluorouracil and leucovorin in T3–4 rectal cancers: results of FFCD 9203. J Clin Oncol. 2006;24:4620–5.
Minsky BD, Cohen AM, Kemeny N, et al. Enhancement of radiation-induced downstaging of rectal cancer by fluorouracil and high-dose leucovorin chemotherapy. J Clin Oncol. 1992;10:79–84.
Mohiuddin M, Hayne M, Regine WF, et al. M. Prognostic significance of postchemoradiation stage following preoperative chemotherapy and radiation for advanced/recurrent rectal cancers. Int J Radiat Oncol Biol Phys. 2000;48:1075–80.
Habr-Gama A, Perez RO, Nadalin W, et al. Operative versus nonoperative treatment for stage 0 distal rectal cancer following chemoradiation therapy: long-term results. Ann Surg. 2004;240:711–7.
Taback B, Hoon DS. Circulating nucleic acids and proteomics of plasma/serum: clinical utility. Ann N Y Acad Sci. 2004;1022:1–8.
Taback B, Hoon DS. Circulating nucleic acids in plasma and serum: past, present and future. Curr Opin Mol Ther. 2004;6:273–8.
Fujimoto A, O’Day SJ, Taback B, et al. Allelic imbalance on 12q22–23 in serum circulating DNA of melanoma patients predicts disease outcome. Cancer Res. 2004;64:4085–8.
Gormally E, Caboux E, Vineis P, et al. Circulating free DNA in plasma or serum as biomarker of carcinogenesis: practical aspects and biological significance. Mutat Res.Mutat Res. 2007;635:105–17.
Jahr S, Hentze H, Englisch S, et al. DNA fragments in the blood plasma of cancer patients: quantitations and evidence for their origin from apoptotic and necrotic cells. Cancer Res. 2001;61:1659–65.
Giacona MB, Ruben GC, Iczkowski KA, et al. Cell-free DNA in human blood plasma: length measurements in patients with pancreatic cancer and healthy controls. Pancreas. 1998;17:89–97.
Wang BG, Huang HY, Chen Y, et al. Increased plasma DNA integrity in cancer patients. Cancer Res. 2003;63:3966–8.
Umetani N, Kim J, Hiramatsu S, et al. Increased integrity of free circulating DNA in sera of patients with colorectal or periampullary cancer: direct quantitative PCR for repeats. Clin Chem. 2006;52:1062–9.
Umetani N, Giuliano AE, Hiramatsu, et al. Prediction of breast tumor progression by integrity of free circulating DNA in serum. J Clin Oncol. 2006;24:4270–6.
Jiang WW, Zahurak M, Goldenberg D, et al. Increased plasma DNA integrity index in head and neck cancer patients. Int J Cancer. 2006;119:2673–6.
Mandard AM, Dalibard F, Mandard JC, et al. Pathologic assessment of tumor regression after preoperative chemoradiotherapy of esophageal carcinoma. Clinicopathologic correlations. Cancer. 1994;73:2680–6.
Beddy D, Hyland JM, Winter DC, et al. A simplified tumor regression grade correlates with survival in locally advanced rectal carcinoma treated with neoadjuvant chemoradiotherapy. Ann Surg Oncol. 2008;15:3471–7.
Ryan R, Gibbons D, Hyland JMT, et al. Pathological response following long-course neoadjuvant chemoradiotherapy for locally advanced rectal cancer. Histopathology. 2005;47:141–6.
Huang ZH, Li LH, Hua D. Quantitative analysis of plasma circulating DNA at diagnosis and during follow-up of breast cancer patients. Cancer Lett. 2006;243:64–70.
Habr-Gama A. Colon and rectal surgery without mechanical bowel preparation: a randomized prospective trial. Ann Surg. 2003; 237:363–7. Tech Coloproctol. 2004;8:128.
Maretto I, Pomerri F, Pucciarelli S, et al. The potential of restaging in the prediction of pathologic response after preoperative chemoradiotherapy for rectal cancer. Ann Surg Oncol. 2007;14:455–61.
Goebel G, Zitt M, Zitt M, et al. Circulating nucleic acids in plasma or serum (CNAPS) as prognostic and predictive markers in patients with solid neoplasias. Dis Markers. 2005;21:105–20.
Locker GY, Hamilton S, Harris JJ, et al. ASCO. 2006 update of recommendations for the use of tumor markers in gastrointestinal cancer. J Clin Oncol. 2006;24:5313–27.
Tsang JC, Lo YM. Circulating nucleic acids in plasma/serum. Pathology. 2007;39:197–207.
Kamat AA, Sood AK, Dang D, et al. Quantification of total plasma cell-free DNA in ovarian cancer using real-time PCR. Ann N Y Acad Sci. 2006;1075:230–4.
Stroun M, Lyautey J, Lederrey C, et al. About the possible origin and mechanism of circulating DNA apoptosis and active DNA release. Clin Chim Acta. 2001;313:139–42.
Stroun M, Lyautey J, Lederrey C, et al. Alu repeat sequences are present in increased proportions compared to a unique gene in plasma/serum DNA: evidence for a preferential release from viable cells? Ann N Y Acad Sci. 2001;945:258–64.
Deligezer U, Eralp Y, Akisik EE, et al. Size distribution of circulating cell-free DNA in sera of breast cancer patients in the course of adjuvant chemotherapy. Clin Chem Lab Med. 2008;46:311–7.
Deligezer U, Eralp Y, Akisik EZ, et al. Effect of adjuvant chemotherapy on integrity of free serum DNA in patients with breast cancer. Ann N Y Acad Sci. 2008;1137:175–9.
Zitt M, Muller HM, Rochel M, et al. Circulating cell-free DNA in plasma of locally advanced rectal cancer patients undergoing preoperative chemoradiation: a potential diagnostic tool for therapy monitoring. Dis Markers. 2008;25:159–65.
Lindsay KJ, Coates PJ, Lorimore, et al. The genetic basis of tissue responses to ionizing radiation. Br J Radiol. 2007;80(Spec No 1):S2–6.
Acknowledgment
This study was supported in part by grants from the Banca AntonVeneta, CARIPARO, and the AIRC Foundation. Biological samples were provided by 2nd Surgical Clinic, Tumor Tissue Biobank. The article was reviewed and edited for English-language usage by American Journal Experts.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agostini, M., Pucciarelli, S., Enzo, M.V. et al. Circulating Cell-Free DNA: A Promising Marker of Pathologic Tumor Response in Rectal Cancer Patients Receiving Preoperative Chemoradiotherapy. Ann Surg Oncol 18, 2461–2468 (2011). https://doi.org/10.1245/s10434-011-1638-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-011-1638-y