Abstract
Background
We conducted the current study to investigate the clinical outcomes of metastatic gastric carcinoma (MGC) patients who experienced gastrointestinal (GI) perforation during palliative chemotherapy and to examine the prognostic factors associated with survival after perforation.
Methods
We reviewed the medical records of patients at the Center for Gastric Cancer of the National Cancer Center, Korea who developed GI perforation during palliative chemotherapy between January 2001 and December 2008.
Results
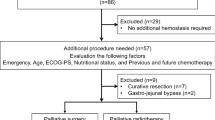
Of the 1,856 patients who received palliative chemotherapy for MGC, 32 patients (1.7%) developed GI perforation during chemotherapy. Patients with perforation at the primary gastric site were more likely to have ulcerative gastric cancer lesion (90.5 vs. 40.0%, P = 0.034) or gastric tumor bleeding (28.6 vs. 0%, P = 0.298), and less likely to have Bormann type IV (14.3 vs. 60.0%, P = 0.062), than patients with perforation at nongastric sites. In 14 patients (43.8%) who resumed chemotherapy after perforation, the disease control rate was 57.1%, and median overall survival (OS) after perforation was 7.5 months [95% confidence interval (CI), 6.0–9.0 months]. In all patients, median OS following perforation was 4.0 months (95% CI, 1.5–6.6 months), and multivariate analysis revealed that differentiated tumor histology, response to chemotherapy before perforation, and absence of septic shock at time of perforation were significantly associated with favorable OS after perforation.
Conclusions
As patients experiencing GI perforation during palliative chemotherapy have heterogeneous clinical presentation, we need to adopt different approaches in the management of the patients that are compatible with the favorable prognostic factors.


Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71–96.
Shin HR, Jung KW, Won YJ, Kong HJ, Yim SH, Sung J, et al. National cancer incidence for the year 2002 in Korea. Cancer Res Treat. 2007;39:139–49.
D'Angelica M, Gonen M, Brennan MF, Turnbull AD, Bains M, Karpeh MS. Patterns of initial recurrence in completely resected gastric adenocarcinoma. Ann Surg. 2004;240:808–16.
Yoo CH, Noh SH, Shin DW, Choi SH, Min JS. Recurrence following curative resection for gastric carcinoma. Br J Surg. 2000;87:236–42.
Glimelius B, Ekstrom K, Hoffman K, Graf W, Sjoden PO, Haglund U, et al. Randomized comparison between chemotherapy plus best supportive care with best supportive care in advanced gastric cancer. Ann Oncol. 1997;8:163–8.
Casaretto L, Sousa PL, Mari JJ. Chemotherapy versus support cancer treatment in advanced gastric cancer: a meta-analysis. Braz J Med Biol Res. 2006;39:431–40.
Asmis TR, Capanu M, Kelsen DP, Shah MA. Systemic chemotherapy does not increase the risk of gastrointestinal perforation. Ann Oncol. 2007;18:2006–8.
Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, et al. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350:2335–42.
Sandler A, Gray R, Perry MC, Brahmer J, Schiller JH, Dowlati A, et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med. 2006;355:2542–50.
Miller K, Wang M, Gralow J, Dickler M, Cobleigh M, Perez EA, et al. Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic breast cancer. N Engl J Med. 2007;357:2666–76.
Hapani S, Chu D, Wu S. Risk of gastrointestinal perforation in patients with cancer treated with bevacizumab: a meta-analysis. Lancet Oncol. 2009;10:559–68.
Han ES, Monk BJ. What is the risk of bowel perforation associated with bevacizumab therapy in ovarian cancer? Gynecol Oncol. 2007;105:3–6.
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;92:205–16.
Roviello F, Rossi S, Marrelli D, De Manzoni G, Pedrazzani C, Morgagni P, et al. Perforated gastric carcinoma: a report of 10 cases and review of the literature. World J Surg Oncol. 2006;4:19.
Adachi Y, Mori M, Maehara Y, Matsumata T, Okudaira Y, Sugimachi K. Surgical results of perforated gastric carcinoma: an analysis of 155 Japanese patients. Am J Gastroenterol. 1997;92:516–8.
Kasakura Y, Ajani JA, Fujii M, Mochizuki F, Takayama T. Management of perforated gastric carcinoma: a report of 16 cases and review of world literature. Am Surg. 2002;68:434–40.
Lehnert T, Buhl K, Dueck M, Hinz U, Herfarth C. Two-stage radical gastrectomy for perforated gastric cancer. Eur J Surg Oncol. 2000;26:780–4.
Gertsch P, Yip SK, Chow LW, Lauder IJ. Free perforation of gastric carcinoma. Results of surgical treatment. Arch Surg. 1995;130:177–81.
Shah MA, Ramanathan RK, Ilson DH, Levnor A, D'Adamo D, O'Reilly E, et al. Multicenter phase II study of irinotecan, cisplatin, and bevacizumab in patients with metastatic gastric or gastroesophageal junction adenocarcinoma. J Clin Oncol. 2006;24:5201–6.
Sugrue M, Kozloff M, Hainsworth J, Badarinath S, Cohn A, Flynn P, et al. Risk factors for gastrointestinal perforations in patients with metastatic colorectal cancer receiving bevacizumab plus chemotherapy. J Clin Oncol. 2006;24(suppl 18):abstr 3535.
Acknowledgment
This study was supported by grants 0710650 and 1010180 from Research Institute and Hospital, National Cancer Center, Republic of Korea.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, M.H., Kim, S.N., Kim, N.K. et al. Clinical Outcomes and Prognostic Factors of Metastatic Gastric Carcinoma Patients Who Experience Gastrointestinal Perforation During Palliative Chemotherapy. Ann Surg Oncol 17, 3163–3172 (2010). https://doi.org/10.1245/s10434-010-1164-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-010-1164-3