Abstract
Background
Individuals with a first-degree family history of colorectal cancer (CRC) are at increased risk of CRC. Study objectives were: (1) to estimate the proportion of first-degree relatives (FDR) of CRC patients being screened for CRC and (2) to identify predictors of screened behavior.
Methods
A questionnaire was mailed to 640 stage I–III CRC patients from a population-based registry to identify FDR. A survey was sent to 747 FDR, aged 40 or older, to assess CRC screening, knowledge, demographics, access, benefits, and barriers of CRC screening. Factor analysis was used to detect underlying constructs. Predictors of screening were explored by multivariate analysis (MVA).
Results
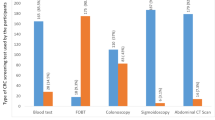
There was a 54% and 51% response for patients and FDR, respectively. Among FDR, 86% were born in Canada, 94% spoke English, 93.5% had a high school education, 73% were married, and 55% were employed. The age distribution was: 40–44 years (19.7%), 45–49 (19.1%), 50–54 (16%), 55–59 (15.2%), 60–64 (9.8%), and >65 (18%). Seventy percent had undergone CRC screening with 60% adherent to current guidelines. Of those screened, 33.7% had fecal occult blood testing, 19.4% had barium enema, 10.7% had sigmoidoscopy, and 58.7% had colonoscopy. Five constructs influencing CRC screening include: salience and coherence, perceived susceptibility, response efficacy, social influence, and cancer worries. MVA determined age >50 years as the most important predictor of screening.
Conclusion
In this survey, 70% of FDR of CRC patients had undergone screening; age was the most important predictor. Understanding underlying constructs influencing screening behavior may improve uptake of CRC screening in this population.
Similar content being viewed by others
References
National Cancer Institute of Canada. Canadian Cancer Statistics. Toronto, Ontario; 2001.
Vogelstein B, Fearon ER, Hamilton SR, Kern SE, Preisinger AC, Leppert M, et al. Genetic alterations during colorectal-tumor development. N Engl J Med. 1988;319:525–32.
Winawer SJ, Zauber AG, Ho MN, O’Brien MJ, Gottlieb LS, Sternberg SS, et al. Prevention of colorectal cancer by colonoscopic polypectomy. N Engl J Med. 1993;329:1977–81.
McLeod RS, Canadian task force on preventive health care. Screening strategies for colorectal cancer: a systematic review of the evidence. Can J Gastroenterol. 2001;15:647–60.
McGregor SE, Hilsden RJ, Murray A, Bryant HE. Colorectal cancer screening: practices and opinions of primary care physicians. Prev Med. 2004;39:279–85.
Bancej C, Nichol M, Jones-McLean E et al. Prevalence and correlates of colorectal cancer screening among adults aged 50 to 74 in four Canadian provinces. Canadian Cancer Society 2009; Available from: http://www.cancer.ca/canada-wide/about%20cancer/cancer%20statistics/canadian%20cancer%20statistics/special%20topics/progress%20in%20cancer%20control%20screening.aspx?sc_lang=en.
Fuchs CS, Giovannucci EL, Colditz GA, Stampfer MJ, Giovannucci EL, Hunter DJ, et al. A prospective study of family history and the risk of colorectal cancer. N Engl J Med. 1994;331:1669–74.
Johns LE, Houlston RS. A systematic review and meta-analysis of familial colorectal cancer risk. Am J Gastroenterol. 2001;96:2992–3003.
U.S. Preventive Services Task Force. Screening for colorectal cancer: recommendation and rationale. Ann Intern Med. 2002;133:573–84.
Simmang CL, Senatore P, Lowry A, Hicks T, Burnstein M, Dentsman F, et al. Practice parameters for detection of colorectal neoplasms. The Standards Committee, The American Society of Colon and Rectal Surgeons. Dis Colon Rectum. 1999;42:1123–9.
Smith RA, Cokkinides V, Eyre HJ. American Cancer Society guidelines for the early detection of cancer, 2004. CA Cancer J Clin. 2004;54:41–52.
Mack LA, Stuart H, Temple WJ. Survey of colorectal cancer screening practices in a large Canadian urban centre. Can J Surg. 2004;47:189–94.
McGregor SE, Hilsden RJ, Li FX, Bryant HE, Murray A. Low uptake of colorectal cancer screening 3 yr after release of national recommendations for screening. Am J Gastroenterol. 2007;102:1727–35.
Rawl S, Champion V, Menon U, Loehrer PJ Sr, Vance GH, Skinner CS. Validation of scales to measure benefits and barriers to colorectal cancer screening. J Psychosoc Oncol. 2001;19:63.
Tiro JA, Vernon SW, Hyslop T, Myers RE. Factorial validity and invariance of a survey measuring psychosocial correlates of colorectal cancer screening among African Americans and Caucasians. Cancer Epidemiol Biomarkers Prev. 2005;14:2855–61.
Dillman DA. Survey implementation. In: Dillman DA editors. Mail and internet surveys: the tailored design method. New York: John Wiley & Sons, Inc.; 2000. p. 149–93.
Kalantar JS, Talley NJ. The effects of lottery incentive and length of questionnaire on health survey response rates: a randomized study. J Clin Epidemiol. 1999;52:1117–22.
Roberts LM, Wilson S, Roalfe A, Bridge P. A randomised controlled trial to determine the effect on response of including a lottery incentive in health surveys. BMC Health Serv Res. 2004;4:30.
Asch DA, Jedrziewski MK, Christakis NA. Response rates to mail surveys published in medical journals. J Clin Epidemiol. 1997;50:1129–36.
Vernon SW, Myers RE, Tilley BC. Development and validation of an instrument to measure factors related to colorectal cancer screening adherence. Cancer Epidemiol Biomarkers Prev. 1997;6:825–32.
Codori AM, Petersen GM, Miglioretti DL, Boyd P. Health beliefs and endoscopic screening for colorectal cancer: potential for cancer prevention. Prev Med. 2001;33:128–36.
Coronado GD, Farias A, Thompson B, Godina R, Oderkirk W. Attitudes and beliefs about colorectal cancer among Mexican Americans in communities among the US-Mexican border. Ethn Dis. 2006;16:421–7.
Ramji F, Cotterchio M, Manno M, Rabeneck L, Gallinger S. Association between subject factors and colorectal cancer screening participation in Ontario, Canada. Cancer Detect Prev. 2005;29:221–6.
Center for Disease Control. Trends in screening for colorectal cancer—United States 1997 and 1999. MMWR CDC. 2001;50:162–6.
Etzioni DA, Ponce NA, Babey SH, Spencer BA, Brown ER, Ko CY, et al. A population-based study of colorectal cancer test use: results from the 2001 California Health Interview Survey. Cancer. 2004;101:2523–32.
Ioannou GN, Chapko MK, Dominitz JA. Predictors of colorectal cancer screening participation in the United States. Am J Gastroenterol. 2003;89:2082–91.
Cotterchio M, McKeown-Eyssen G, Sutherland H, Buchan G, Aronson M, Easson AM, et al. Ontario Familial Colon Cancer Registry: methods and first-year response rates. Chron Dis Can. 2000;21:81–6.
Madlensky L, Esplen MJ, Gallinger S, McLaughlin JR, Goel V. Relatives of colorectal cancer patients: factors associated with screening behavior. Am J Prev Med. 2003;25:187–94.
Manne S, Markowitz A, Winawer S, Meropol NJ, Haller D, Rakowski W, et al. Correlates of colorectal cancer screening compliance and stage of adoption among siblings of individuals with early onset colorectal cancer. Health Psychol. 2002;21:3–15.
Gordon NP, Hiatt RA, Lampert DI. Concordance of self-reported data and medical record audit for six cancer screening procedures. J Natl Cancer Inst. 1993;85:566–70.
Baier M, Calonge N, Cutter G, McClatchey M. Schoentgen S, Hines S, et al. Validity of self-reported colorectal cancer screening behavior. Cancer Epidemiol Biomarkers Prev. 2000;9:229–32.
Hawley ST, Levin B, Vernon SW. Colorectal cancer screening by primary care physicians in two medical care organizations. Cancer Detect Prev. 2001;25:309–18.
Read TE, Kodner IJ. Colorectal cancer: risk factors and recommendations for early detection. Am Fam Physician. 1999;59:3083–92.
Guidelines & Protocols Advisory Committee. Detection of colorectal neoplasms in asymptomatic patients. British Columbia, BC Health Services; 2004.
Maiman LA, Becker MH. The health belief model: origins and correlates in psychological theory. Health Educ Monogr. 1974;2:9–26.
Rawl SM, Menon U, Champion VL, Foster JL, Skinner CS. Colorectal cancer screening beliefs: focus groups with first-degree relatives. Cancer Pract. 2000;8:32–7.
Madlensky L, Esplen MJ, Goel V. Reasons given by relatives of colorectal cancer patients for not undergoing screening. Prev Med. 2004;39:643–8.
Montgomery GH, Erblich J, Dilorenzo T, Bovbjerg DH. Family and friends with disease: their impact on perceived risk. Prev Med. 2003;37:242–9.
Blalock SJ, Devellis B, Afifi RA, Sandler RS. Risk perceptions and participation in colorectal cancer screening. Health Psychol. 1990;9:792–806.
Stermer T, Hodgson S, Kavalier F, Watts S, Jones R. Patients’ and professionals’ opinions of services for people at an increased risk of colorectal cancer: an exploratory qualitative study. Fam Cancer. 2004;3:49–53.
Glanz K, Steffen AD, Taglialatela LA. Effects of colon cancer risk counseling for first-degree relatives. Cancer Epidemiol Biomarkers Prev. 2007;16:1485–91.
Alberta Cancer Board Cancer Screening Programs. Alberta colorectal cancer screening Program. Alberta, Alberta Cancer Board; 2008.
Taylor ML, Anderson R. Colorectal cancer screening: physician attitudes and practices. WMJ. 2002;101:39–43.
Mitchell RJ, Brewster D, Campbell H, Porteous ME, Wyllie AH, Bird CC, et al. Accuracy of reporting of family history of colorectal cancer. Gut. 2004;53:291–5.
Lynch KL, Ahnen DJ, Byers T, Weiss DG, Lieberman DA. First-degree relatives of patients with advanced colorectal adenomas have an increased prevalence of colorectal cancer. Clin Gastroenterol Hepatol. 2003;1:96–102.
St. John DJB, McDermott FT, Hopper JL, Debney EA, Johnson WR, Hughes ES. Cancer risk in relatives of patients with common colorectal cancer. Ann Intern Med. 1993;118:785–90.
Institute of Cancer Research. Colorectal cancer screening workshop report. CIHR 2006; Available from: http://www.cihr-irsc.gc.ca/e/31266.html. Accessed February 2, 2008.
Acknowledgment
The authors would like to acknowledge the helpful discussions regarding study design and analysis with Alberta Cancer Registry Staff as well as Dr. Elizabeth McGregor and Dr. Bejoy Thomas.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mack, L.A., Cook, L.S., Temple, W.J. et al. Colorectal Cancer Screening Among First-Degree Relatives of Colorectal Cancer Patients: Benefits and Barriers. Ann Surg Oncol 16, 2092–2100 (2009). https://doi.org/10.1245/s10434-009-0528-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-009-0528-z