Abstract
Background
Bone morphogenetic proteins (BMPs) are secreted signaling molecules belonging to the transforming growth factor (TGF)-β superfamily. Recent studies demonstrated that the expression patterns of BMPs are altered in several tumors. The purpose of the current study was to examine the expression of BMP7 in malignant and normal colorectal tissues, and to analyze whether BMP7 expression levels correlate with clinicopathological variables and prognosis in colorectal cancer.
Methods
Paired colorectal tissue samples from cancer and corresponding nonmalignant tissues were obtained from 65 patients who underwent surgical resection for colorectal cancer. The expression status of BMP7 mRNA was investigated by quantitative real-time reverse-transcription polymerase chain reaction (RT-PCR) and protein expression was analyzed by an immunohistochemical study.
Results
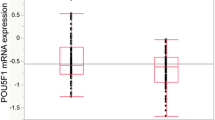
Quantitative real-time RT-PCR showed that BMP7 mRNA expression in cancerous tissue was significantly higher than that in normal tissue (p < 0.0001). An immunohistochemical study revealed that BMP7 was predominantly expressed in cancer cells. Elevated BMP7 expression was significantly correlated with depth of tumor invasion, liver metastasis, liver recurrence, advanced Dukes’ classification, and cancer-related death (p < 0.05, 0.001, 0.01, 0.05 and 0.01, respectively). Furthermore, patients with the highest levels of BMP7 expression showed the poorest prognosis (p < 0.01). A multivariate analysis showed that BMP7 expression status was an independent prognostic factor of overall survival (relative risk, 2.29; 95% confidence interval, 1.08–5.30; p < 0.05).
Conclusions
Expression of BMP7 in colorectal tumors correlates with parameters of pathological aggressiveness such as liver metastasis and poor prognosis. Thus, BMP7 could be a useful clinical marker for colorectal cancer patients.



Similar content being viewed by others
References
Messerini L, Cianchi F, Cortesini C, et al. Incidence and prognostic significance of occult tumor cells in lymph nodes from patients with stage IIA colorectal carcinoma. Hum Pathol 2006;37:1259–67.
Shih W, Chetty R, Tsao MS. Expression profiling by microarrays in colorectal cancer (Review). Oncol Rep 2005;13:517–24.
Tsukuma H, Ajiki W, Oshima A. [Cancer incidence in Japan]. Gan To Kagaku Ryoho 2004;31:840–6.
Weitz J, Koch M, Debus J, Hohler T, et al. Colorectal cancer. Lancet 2005;365:153–65.
Roberts RB, Arteaga CL, Threadgill DW. Modeling the cancer patient with genetically engineered mice: prediction of toxicity from molecule-targeted therapies. Cancer Cell 2004;5:115–20.
Ohmachi T, Tanaka F, Mimori K, et al. Clinical significance of TROP2 expression in colorectal cancer. Clin Cancer Res 2006;12:3057–63.
Williams NS, Gaynor RB, Scoggin S, et al. Identification and validation of genes involved in the pathogenesis of colorectal cancer using cDNA microarrays and RNA interference. Clin Cancer Res 2003;9:931–46.
Ichikawa Y, Ishikawa T, Takahashi S, et al. Identification of genes regulating colorectal carcinogenesis by using the algorithm for diagnosing malignant state method. Biochem Biophys Res Commun 2002;296:497–506.
Kawabata M, Imamura T, Miyazono K. Signal transduction by bone morphogenetic proteins. Cytokine Growth Factor Rev 1998;9:49–61.
Hogan BL. Bone morphogenetic proteins in development. Curr Opin Genet Dev 1996;6:432–8.
Urist MR. Bone: formation by autoinduction. Science 1965;150:893–9.
Celeste AJ, Iannazzi JA, Taylor RC, et al. Identification of transforming growth factor beta family members present in bone-inductive protein purified from bovine bone. Proc Natl Acad Sci USA 1990;87:9843–7.
Miyazono K, Kusanagi K, Inoue H. Divergence and convergence of TGF-beta/BMP signaling. J Cell Physiol 2001;187:265–76.
Ducy P, Karsenty G. The family of bone morphogenetic proteins. Kidney Int 2000;57:2207–14.
Graff JM. Embryonic patterning: to BMP or not to BMP, that is the question. Cell 1997;89:171–4.
Ebendal T, Bengtsson H, Soderstrom S. Bone morphogenetic proteins and their receptors: potential functions in the brain. J Neurosci Res 1998;51:139–46.
Wozney JM. The bone morphogenetic protein family: multifunctional cellular regulators in the embryo and adult. Eur J Oral Sci 1998;106:160–6.
Mimori K, Mori M, Shiraishi T, et al. Clinical significance of tissue inhibitor of metalloproteinase expression in gastric carcinoma. Br J Cancer 1997;76:531–6.
Mori M, Mimori K, Yoshikawa Y, et al. Analysis of the gene-expression profile regarding the progression of human gastric carcinoma. Surgery 2002;131:S39–47.
Ogawa K, Utsunomiya T, Mimori K, et al. Clinical significance of elongation factor-1 delta mRNA expression in oesophageal carcinoma. Br J Cancer 2004;91:282–6.
Mantel N. Evaluation of survival data and two new rank order statistics arising in its consideration. Cancer Chemother Rep 1966;50:163–70.
Gobbi G, Sangiorgi L, Lenzi L, et al. Seven BMPs and all their receptors are simultaneously expressed in osteosarcoma cells. Int J Oncol 2002;20:143–7.
Rothhammer T, Poser I, Soncin F, et al. Bone morphogenic proteins are overexpressed in malignant melanoma and promote cell invasion and migration. Cancer Res 2005;65:448–56.
Alarmo EL, Rauta J, Kauraniemi P, et al. Bone morphogenetic protein 7 is widely overexpressed in primary breast cancer. Genes Chromosomes Cancer 2006;45:411–9.
Alarmo EL, Kuukasjarvi T, Karhu R, et al. A comprehensive expression survey of bone morphogenetic proteins in breast cancer highlights the importance of BMP4 and BMP7. Breast Cancer Res Treat 2006.
Masuda H, Fukabori Y, Nakano K, et al. Increased expression of bone morphogenetic protein-7 in bone metastatic prostate cancer. Prostate 2003;54:268–74.
Hibbs K, Skubitz KM, Pambuccian SE, et al. Differential gene expression in ovarian carcinoma: identification of potential biomarkers. Am J Pathol 2004;165:397–414.
Sunde JS, Donninger H, Wu K, et al. Expression profiling identifies altered expression of genes that contribute to the inhibition of transforming growth factor-{beta} signaling in ovarian cancer. Cancer Res 2006;66:8404–12.
Bobinac D, Maric I, Zoricic S, et al. Expression of bone morphogenetic proteins in human metastatic prostate and breast cancer. Croat Med J 2005;46:389–96.
Sulzbacher I, Birner P, Trieb K, et al. The expression of bone morphogenetic proteins in osteosarcoma and its relevance as a prognostic parameter. J Clin Pathol 2002;55:381–5.
Yang S, Zhong C, Frenkel B, et al. Diverse biological effect and SMAD signaling of bone morphogenetic protein 7 in prostate tumor cells. Cancer Res 2005;65:5769–77.
Deng H, Makizumi R, Ravikumar TS, et al. Bone morphogenetic protein-4 is overexpressed in colonic adenocarcinomas and promotes migration and invasion of HCT116 cells. Exp Cell Res 2007.
Grijelmo C, Rodrigue C, Svrcek M, et al. Proinvasive activity of BMP-7 through SMAD4/src-independent and ERK/Rac/JNK-dependent signaling pathways in colon cancer cells. Cell Signal 2007;19:1722–32.
Pessaux P, Lermite E, Brehant O, et al. Repeat hepatectomy for recurrent colorectal liver metastases. J Surg Oncol 2006;93:1–7.
Talbot IC, Ritchie S, Leighton MH, et al. The clinical significance of invasion of veins by rectal cancer. Br J Surg 1980;67:439–42.
Minsky BD, Mies C, Rich TA, et al. Potentially curative surgery of colon cancer: the influence of blood vessel invasion. J Clin Oncol 1988;6:119–27.
Inoue T, Mori M, Shimono R, et al. Vascular invasion of colorectal carcinoma readily visible with certain stains. Dis Colon Rectum 1992;35:34–9.
Beck SE, Jung BH, Fiorino A, et al. Bone morphogenetic protein signaling and growth suppression in colon cancer. Am J Physiol Gastrointest Liver Physiol 2006;291:G135–45.
Acknowledgments
Presented in part at The Society of Surgical Oncology 60th Annual Cancer Symposium, March 15–18, 2007, Washington, DC. This work was supported in part by Core Research for Evolutional Science and Technology (CREST), Japan Science and Technology Agency (JST); Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (grant numbers 17109013, 18390367, 18590333, 18659384, 18890133, 18015039, 19390336, and 19591509). We thank T. Shimooka, K. Ogata, M. Oda, M. Kasagi, and Y. Nakagawa for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Motoyama, K., Tanaka, F., Kosaka, Y. et al. Clinical Significance of BMP7 in Human Colorectal Cancer. Ann Surg Oncol 15, 1530–1537 (2008). https://doi.org/10.1245/s10434-007-9746-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-007-9746-4