Abstract
Objective
This is a phase II study, the aim of which is to determine if a laparoscopic approach can be used in pelvic autonomic nerve-preserving surgery for patients with lower rectal cancer following chemoradiation therapy.
Methods
Patients with T3 lower rectal cancer treated by preoperative chemoradiation were recruited and subjected to laparoscopic pelvic autonomic nerve-preserving surgery with total mesorectal excision and a sphincter-saving procedure. This study was performed with the approval of the ethics committee of National Taiwan University Hospital. Because the quality of a surgical trial is highly dependent on the skill of the surgeon with respect to the technique under study, it is imperative that a surgical trial only be implemented after the surgical technique has been judged to be mature. Before the start of this clinical trial, we gained a sound knowledge of surgical anatomy through conventional open surgery for rectal cancer and mastered the related laparoscopic skills from other sound and proven laparoscopic approaches, including right hemicolectomy, left hemicolectomy, among others. We determined that the learning curve for this surgical technique necessitated that colorectal surgeons carry out at least 20 such procedures. At this point we conducted this clinical trial.
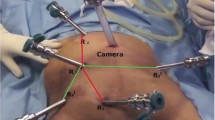
The details of the surgical procedures have been shown in the attached video. Briefly, the dissection commences at the pelvic promontory with exposure and preservation of the superior hypogastric plexus. The pre-aortic plexus and inferior mesenteric plexus are preserved by sparing the pre-aortic connective tissue and leaving a 1– to 2-cm-long stump of the inferior mesenteric artery in situ. Subsequently, the “holy plane” at the transition of the mesosigmoid to the mesorectum is meticulously dissected to progressively displace the hypogastric nerves dorsally and laterally and, therefore, preserving them. Following adequate dorsal and lateral dissection down to the floor of the pelvis, the so-called lateral ligament is reached at which the mesorectum appears to be adherent, anteriorly and laterally, to the inferior hypogastric plexus (at roughly 10:00–2:00 O’clock or within an angle of 60° about symphysis on both sides). The ligaments are divided immediately at the endopelvic fascia of the mesorectum to avoid damage to the inferior hypogastric plexus (pelvic plexus). Finally, great care was taken to dissect the lateral border of Denonvilliers’ fascia where the inferior hypogastric plexus joins the neurovascular bundle described by Walsh.
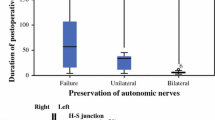
Postoperatively, only patients successfully operated on by total pelvic autonomic nerve-preserving surgery were included in the statistical analysis of surgical outcomes. Preoperatively, all patients were screened for their genitourinary function by a questionnaire-based interview. Patients with abnormal preoperative baseline functional data were excluded from further postoperative assessment of sexual or urinary function. The male sexual function was evaluated by potency and ejaculation. In female patients, the sexual function was assessed by vaginal lubrication, dyspareunia, sexual arousal, and orgasm. The reason for these four parameters is because the influence of pelvic autonomic nerve damage on female sexual function has been ambiguous but would most likely result in impairment of vaginal lubrication and congestion of the genitals. We evaluated sexual function at 6 months postoperatively, when the temporary colostomy had been closed and the patients were completely recovered from surgical disability. In evaluating urinary function, the duration between initial voiding trial and spontaneous voiding was recorded. The questionnaire used for the assessment of urinary dysfunction was based on the International Prostate Symptom Score and the following parameters from this Score were used: incomplete emptying, frequency, intermittency, urgency, week stream, straining, and nocturia. Any voiding problems recovered within 3 months after the operation were considered to be transient bladder voiding dysfunction; all other voiding problems were deemed persistent. The interview and scoring of the questionnaire were done by the research assistant blinded to operation procedures. The genitourinary function was ranked as good, fair (decreased), and poor (impaired).
Results
Between June 2003 and December 2005, a total of 98 patients (stage II: n = 44; stage III: n = 54; male: n = 50; female: n = 48) were enrolled in this study. Technically, although the dissection plane is a little blurred by preoperative chemoradiation therapy, the laparoscopic pelvic nerve-preserving procedure with total mesorectal excision was successfully performed in 89 (90.8%) patients with an acceptable operation time (284.4 ± 44.8 minutes; mean ± standard deviation) and little blood loss (114.5 ± 24 ml). The number of dissected lymph nodes was 16.4 ± 4.0. With respect to scrutiny of surgical specimens, the distal safety margin was adequate (mean: 2.4 cm; range: 1.2–5.6 cm), and the circumferential resection margins were free of tumor invasion (mean: 8.6 mm; range: 2–18 mm). A total of 74 patients completed the evaluation of urinary function. For these 74 patients, the median duration of training for the Foley catheter was 7 days (range: 4–64 days). The voiding function after removal of the Foley catheter were good in 53 (71.6%) patients, fair in 17 (23.0%), and poor in four (5.4%). Of the 17 patients with fair bladder function, eight were transient dysfunction and recovered thereafter. Thirty-two male and 28 female patients who were sexually active before the operation responded to the assessment of sexual function. In male patients, the ejaculation was good in 18 (56.3%) patients, fair (decrease in ejaculatory amounts) in six (18.7%), and poor (retrograde ejaculation, failure to ejaculate) in eight (25%). The potency was good in 20 (62.5%) patients, fair in five (15.6%), and poor in seven (21.9%). In female patients, the sexual function was good in 15 (53.6%) patients, fair in four (14.3%), and poor in nine (32.1%). Specific sexual problems in women included lubrication (46.6%, n = 13), dyspareunia (39.2%, n = 11), sexual arousal (28.6%, n = 8), and orgasm in (32.1%, n = 9).
Conclusions
By the laparoscopic approach, total preservation of pelvic autonomic nerves without compromise of the radical extirpation of tumor is technically feasible in the vast majority of patients with lower rectal cancer who have undergone concurrent chemoradiation therapy, thus facilitating the retention of genitourinary function in a significant proportion of such patients.
Similar content being viewed by others
References
Moriya Y. Pelvic node dissection with autonomic nerve sparing for invasive lower rectal cancer: Japanese experience. In: Wanebo HJ, ed. Colorectal Cancer, 1st edn. St. Louis: Mosby-Year Book; 1993, pp 274–89
Milsom JW, Böhm B. Laparoscopic Colorectal Surgery. New York: Springer-Verlag, 1996
Yildirim C, Geers P, Köckerling F. Disorders of bladder and sexual function following total mesorectal excision (TME). In: Köckerling F, ed. Total Mesorectal Excision with the Water-Jet, 1st edn. Hanover: Science Med Dr. Sperber, 2002; 59–66
Heald RJ. The ‘Holy Plane’ of rectal surgery. J R Soc Med 1988; 81:503–8
Heald RJ, Moran BJ, Brown G, Daniels IR. Optimal total mesorectal excision for rectal cancer is by dissection in front of Denonvilliers’ fascia. Br J Surg 2004; 91:121–3
Enker WE. Total mesorectal excision–the new golden standard of surgery for rectal cancer. Ann Med 1997; 29:127–33
Lepor H, Gregerman M, Crosby R, Mostofi FK, Walsh PC. Precise localization of the autonomic nerves from the pelvic plexus to the corpora cavernosa: a detailed anatomical study of the adult male pelvis. J Urol 1985; 133:207–12
Takahashi T, Ueno M, Azekura K, Ohta H. Lateral ligament: its anatomy and clinical importance. Semin Surg Oncol 2000; 19:386–95
Liang JT, Chien CT, Chang KJ, et al. Neurophysiological basis of sympathetic nerve-preserving surgery for lower rectal cancer–a canine model. Hepatogastroenterology 1998; 45:2206–14
Liang JT, Chang KJ, Wang SM. Anatomical basis of autonomic nerve-preserving total mesorectal excision for rectal cancer. Br J Surg 1997; 84:586–7
Liang JT, Chang KJ, Wang SM. Lateral rectal ligaments contain important nerves. Br J Surg 1998; 85:1162
Walsh PC, Schlegel PN. Radical pelvic surgery with preservation of sexual function. Ann Surg 1988; 208:391–400
Havenga K, Enker WE, McDermott K, Cohen AM, Minsky BD, Guillem J. Male and female sexual and urinary function after total mesorectal excision with autonomic nerve preservation for carcinoma of the rectum. J Am Coll Surg 1996; 182:495–502
Sugihara K, Moriya Y, Akasu T, Fujita S. Pelvic autonomic nerve preservation for patients with rectal carcinoma. Oncologic and functional outcome. Cancer 1996; 78:1871–80
Enker WE. Potency, cure, and local control in the operative treatment of rectal cancer. Arch Surg 1992; 127:1396–401
Leveckis J, Boucher NR, Parys BT, et al. Bladder and erectile dysfunction before and after rectal surgery for cancer. Br J Urol 1995; 76:752–6
Kim NK, Aahn TW, Park JK, et al. Assessment of sexual and voiding function after total mesorectal excision with pelvic autonomic nerve preservation in males with rectal cancer. Dis Colon Rectum 2002; 45:1178–85
Havenga K, Maas CP, DeRuiter MC, et al. Avoiding long-term disturbance to bladder and sexual function in pelvic surgery, particularly with rectal cancer. Semin Surg Oncol 2000; 18:235–43
Nesbakken A, Nygaard K, Bull-Njaa T, et al. Bladder and sexual dysfunction after mesorectal excision for rectal cancer. Br J Surg 2000; 87:206–10
Mannaerts GH, Schijven MP, Hendrikx A, et al. Urologic and sexual morbidity following multimodality treatment for locally advanced primary and locally recurrent rectal cancer. Eur J Surg Oncol 2001; 27:265–72
Maurer CA, Z’Graggen K, Renzulli P, et al. Total mesorectal excision preserves male genital function compared with conventional rectal cancer surgery. Br J Surg 2001; 88:1501–5
Platell CF, Thompson PJ, Makin GB. Sexual health in women following pelvic surgery for rectal cancer. Br J Surg 2004; 91:465–8
Jayne DG, Brown JM, Thorpe H, et al. Bladder and sexual function following resection for rectal cancer in a randomized clinical trial of laparoscopic versus open technique. Br J Surg 2005; 92:1124–32
Hendren SK, O’Connor BI, Liu M, et al. Prevalence of male and female sexual dysfunction is high following surgery for rectal cancer. Ann Surg 2005; 242:212–23
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Video File (MPG 102032 kb)
Rights and permissions
About this article
Cite this article
Liang, JT., Lai, HS. & Lee, PH. Laparoscopic Pelvic Autonomic Nerve-Preserving Surgery for Patients with Lower Rectal Cancer after Chemoradiation Therapy. Ann Surg Oncol 14, 1285–1287 (2007). https://doi.org/10.1245/s10434-006-9052-6
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-006-9052-6