Abstract
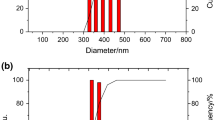
In the present study, stable chitosan nanoparticles (Ch-NPs) were developed using the ionotropic gelation method, where poly(sodium 4-styrenesulfonate) (PSS) was used as a cross-linking agent while polyglutamic acid (PGA) for functionalization to improve the oral uptake through calcium-sensing receptors and amino acid transporters present in intestinal epithelium. Formulation was optimized by the design of experiments (DoE) approach using a three-level central composite design and characterized for in vitro parameters such as morphology, particle size, polydispersity index (PDI), entrapment efficiency and zeta potential. Morphological analysis demonstrated the formation of spherical NPs with particle size, zeta potential, and entrapment efficiency in the range of 210 nm ± 2.8 nm, 18.1 mV ± 0.14 mV, and 85.9% ± 0.28%, respectively. The developed NPs exhibited sustained release at different pH conditions and almost threefold higher uptake in comparison with non-functionalized NPs in Caco-2 cell uptake studies. In vivo studies in diabetic animals demonstrated low levels of plasma glucose for almost 24 h. Pharmacological availability (PA) of insulin administered through Ch-PSS-PGA NPs (17.28 ± 0.9) was significantly higher as compared to that of insulin administered through control NPs, i.e., Ch-PGA NPs (10.9 ± 1.5) and Ch-PSS NPs (12.9 ± 1.8). Data on hand suggest the ability of the developed NPs in overcoming the poor stability and, thus, poor therapeutic efficacy following oral administration.










Similar content being viewed by others
References
Mukhopadhyay P, Mishra R, Rana D, Kundu PP. Strategies for effective oral insulin delivery with modified chitosan nanoparticles: a review. Prog Polym Sci. 2012;37(11):1457–75.
Sung H-W, Sonaje K, Liao Z-X, Hsu L-W, Chuang E-Y. pH-responsive nanoparticles shelled with chitosan for oral delivery of insulin: from mechanism to therapeutic applications. Acc Chem Res. 2012;45(4):619–29.
Khafagy E-S, Morishita M, Onuki Y, Takayama K. Current challenges in non-invasive insulin delivery systems: a comparative review. Adv Drug Deliv Rev. 2007;59(15):1521–46.
TenHoor C, Dressman J. Oral absorption of peptides and proteins. STP Pharm Sci. 1992;2(4):301–12.
Goldberg M, Gomez-Orellana I. Challenges for the oral delivery of macromolecules. Nat Rev Drug Discov. 2003;2(4):289–95.
Woodley JF. Enzymatic barriers for GI peptide and protein delivery. Crit Rev Ther Drug Carrier Syst. 1994;11(2–3):61–95.
Aoki Y, Morishita M, Takayama K. Role of the mucous/glycocalyx layers in insulin permeation across the rat ileal membrane. Int J Pharm. 2005;297(1):98–109.
Yin L, Ding J, He C, Cui L, Tang C, Yin C. Drug permeability and mucoadhesion properties of thiolated trimethyl chitosan nanoparticles in oral insulin delivery. Biomaterials. 2009;30(29):5691–700.
Fonte P, Nogueira T, Gehm C, Ferreira D, Sarmento B. Chitosan-coated solid lipid nanoparticles enhance the oral absorption of insulin. Drug Deliv Transl Res. 2011;1(4):299–308.
Sharma G, Sharma AR, Nam J-S, Doss GPC, Lee S-S, Chakraborty C. Nanoparticle based insulin delivery system: the next generation efficient therapy for type 1 diabetes. J Nanobiotechnol. 2015;13(1):74.
Zhang X, Qi J, Lu Y, He W, Li X, Wu W. Biotinylated liposomes as potential carriers for the oral delivery of insulin. Nanomedicine. 2014;10(1):167–76.
Rekha M, Sharma CP. Synthesis and evaluation of lauryl succinyl chitosan particles towards oral insulin delivery and absorption. J Control Release. 2009;135(2):144–51.
Sarmento B, Ribeiro A, Veiga F, Sampaio P, Neufeld R, Ferreira D. Alginate/chitosan nanoparticles are effective for oral insulin delivery. Pharm Res. 2007;24(12):2198–206.
Wedmore I, McManus JG, Pusateri AE, Holcomb JB. A special report on the chitosan-based hemostatic dressing: experience in current combat operations. J Trauma Acute Care Surg. 2006;60(3):655–8.
Pangburn S, Trescony P, Heller J. Lysozyme degradation of partially deacetylated chitin, its films and hydrogels. Biomaterials. 1982;3(2):105–8.
Dhawan S, Singla AK, Sinha VR. Evaluation of mucoadhesive properties of chitosan microspheres prepared by different methods. AAPS PharmSciTech. 2004;5(4):122–8.
Szczepanowicz K, Hoel H, Szyk-Warszynska L, Bielanska E, Bouzga A, Gaudernack G, et al. Formation of biocompatible nanocapsules with emulsion core and pegylated shell by polyelectrolyte multilayer adsorption. Langmuir. 2010;26(15):12592–7.
Yazdimamaghani M, Razavi M, Mozafari M, Vashaee D, Kotturi H, Tayebi L. Biomineralization and biocompatibility studies of bone conductive scaffolds containing poly (3,4-ethylenedioxythiophene):poly(4-styrene sulfonate) (PEDOT:PSS). J Mater Sci Mater Med. 2015;26(12):274.
Desai MP, Labhasetwar V, Amidon GL, Levy RJ. Gastrointestinal uptake of biodegradable microparticles: effect of particle size. Pharm Res. 1996;13(12):1838–45.
Agrawal AK, Kumar K, Swarnakar NK, Kushwah V, Jain S. “Liquid crystalline nanoparticles”: rationally designed vehicle to improve stability and therapeutic efficacy of insulin following oral administration. Mol Pharm. 2017;14(6):1874–82.
Agrawal AK, Urimi D, Harde H, Kushwah V, Jain S. Folate appended chitosan nanoparticles augment the stability, bioavailability and efficacy of insulin in diabetic rats following oral administration. RSC Adv. 2015;5(127):105179–93.
Agrawal AK, Aqil F, Jeyabalan J, Spencer WA, Beck J, Gachuki BW, et al. Milk-derived exosomes for oral delivery of paclitaxel. Nanomedicine. 2017;13(5):1627–36.
Jung T, Kamm W, Breitenbach A, Kaiserling E, Xiao J, Kissel T. Biodegradable nanoparticles for oral delivery of peptides: is there a role for polymers to affect mucosal uptake? Eur J Pharm Biopharm. 2000;50(1):147–60.
Bernkop-Schnurch A, Walker G. Multifunctional matrices for oral peptide delivery. Crit Rev Ther Drug Carrier Syst. 2001;18(5):43.
Makhlof A, Tozuka Y, Takeuchi H. Design and evaluation of novel pH-sensitive chitosan nanoparticles for oral insulin delivery. Eur J Pharm Sci. 2011;42(5):445–51.
Lin Y-H, Mi F-L, Chen C-T, Chang W-C, Peng S-F, Liang H-F, et al. Preparation and characterization of nanoparticles shelled with chitosan for oral insulin delivery. Biomacromolecules. 2007;8(1):146–52.
Chen M-C, Mi F-L, Liao Z-X, Hsiao C-W, Sonaje K, Chung M-F, et al. Recent advances in chitosan-based nanoparticles for oral delivery of macromolecules. Adv Drug Deliv Rev. 2013;65(6):865–79.
Conigrave AD, Brown EM. Taste receptors in the gastrointestinal tract II. L-amino acid sensing by calcium-sensing receptors: implications for GI physiology. Am J Physiol Gastrointest Liver Physiol. 2006;291(5):G753–61.
Conigrave AD, Quinn SJ, Brown EM. L-amino acid sensing by the extracellular Ca2+-sensing receptor. Proc Natl Acad Sci. 2000;97(9):4814–9.
Agrawal AK, Harde H, Thanki K, Jain S. Improved stability and antidiabetic potential of insulin containing folic acid functionalized polymer stabilized multilayered liposomes following oral administration. Biomacromolecules. 2013;15(1):350–60.
Greenfield NJ. Using circular dichroism spectra to estimate protein secondary structure. Nat Protoc. 2006;1(6):2876.
D’Souza S. A review of in vitro drug release test methods for nano-sized dosage forms. Adv Pharm. 2014.
Acknowledgments
The authors are thankful to the Director of the National Institute of Pharmaceutical Education and Research (NIPER) for providing the infrastructure facilities and to Mr. Rahul Mahajan for the technical assistance in the SEM analysis.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Animal Studies
All institutional and national guidelines for the care and use of laboratory animals were followed.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Urimi, D., Agrawal, A.K., Kushwah, V. et al. Polyglutamic Acid Functionalization of Chitosan Nanoparticles Enhances the Therapeutic Efficacy of Insulin Following Oral Administration. AAPS PharmSciTech 20, 131 (2019). https://doi.org/10.1208/s12249-019-1330-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-019-1330-2