Abstract
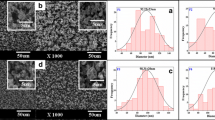
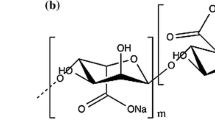
A simple composite electrospun nanofiber of cellulose acetate phthalate (CAP)-polyethylene glycol (PEG) loaded with tetrahydrocurcumin (THC) was developed in this study, and the in vitro diffusion of THC was evaluated. The nanofibers were characterized by scanning electron microscopy, powder X-ray diffraction (PXRD), Fourier-transform infrared spectroscopy (FT-IR), and differential scanning calorimetry (DSC). The formulated nanofiber (NF) with THC has smooth morphology with diameter of around 300–500 nm. The complete entrapment and dispersion of THC was observed from the results of PXRD and DSC due to the loss of THC crystalline property. Further, FT-IR demonstrated that the vibration bands for the polymers used were dominant over the THC, and the vibrational bands of THC were not observed from the final formulation. The drug entrapment by the final CAP + PEG NF was found to be 95.5% with the high swelling index. From the in vitro release study, it was found that the formulated THC-loaded CAP + PEG NF has followed anomalous mechanism, demonstrating both diffusion and swelling controlled modes. The drug release extended up to 12 h with a final cumulative release of 94.24%.









Similar content being viewed by others
References
Rashmi HP, Vandana BP, Medha DJ. Polymeric nanoparticles for targeted treatment in oncology: current insights. Int J Nanomedicine. 2015;10:1001–18. https://doi.org/10.2147/ijn.S56932.
Saravanakumar A, Ganesh M, Jun HJ, Je OC, Yun HC, Jung HL, et al. Preparation and characterization of gatifloxacin-loaded alginate/poly (vinyl alcohol) electrospun nanofibers. Artif Cells Nanomed Biotechnol. 2014;44:847–52. https://doi.org/10.3109/21691401.2014.986676.
Hu X, Liu S, Zhou G, Huang Y, Xie Z, Jing X. Electrospinning of polymeric nanofibers for drug delivery applications. J Control Release. 2014;185:12–21. https://doi.org/10.1016/j.jconrel.2014.04.018.
Wang B, Wang Y, Yin T, Yu Q. Applications of electrospinning technique in drug delivery. Chem Eng Commun. 2010;197:1315–38. https://doi.org/10.1080/00986441003625997.
Chakraborty S, Liao IC, Adler A, Leong KW. Electrohydrodynamics: a facile technique to fabricate drug delivery systems. Adv Drug Deliv Rev. 2009;61:1043–54. https://doi.org/10.1016/j.addr.2009.07.013.
Mendoza PC, Ferrand A, Facca S, Fioretti F, Ladam G, Kuchler BS, et al. Smart hybrid materials equipped by nanoreservoirs of therapeutics. ACS Nano. 2012;6:483–90. https://doi.org/10.1021/nn203817t.
Ignatious F, Sun L, Lee CP, Baldoni J. Electrospun nanofibers in oral drug delivery. J Pharm Res. 2010;27:576–88. https://doi.org/10.1007/s11095-010-0061-6.
Yan S, Xiaoqiang L, Shuiping L, Xiumei M, Ramakrishna S. Controlled release of dual drugs from emulsion electrospun nanofibrous mats. Colloids Surf B Biointerfaces. 2009;73:376–81. https://doi.org/10.1016/j.colsurfb.2009.06.009.
Shen X, Yu D, Zhu L, Branford WC, White K, Chatterton NP. Electrospun diclofenac sodium loaded Eudragit® L 100-55 nanofibers for colon-targeted drug delivery. Int J Pharm. 2011;408:200–7. https://doi.org/10.1016/j.ijpharm.2011.01.058.
Yoo JJ, Kim C, Chung CW, Jeong YI, Kang D. 5-aminolevulinic acid-incorporated poly(vinyl alcohol) nanofiber-coated metal stent for application in photodynamic therapy. Int J Nanomedicine. 2012;7:1997–2005. https://doi.org/10.2147/ijn.S30298.
Fathi AA, Qun L, Chan YW, Chan SY. Novel vitamin and gold-loaded nanofiber facial mask for topical delivery. AAPS PharmSciTech. 2010;11:1164–70. https://doi.org/10.1208/s12249-010-9475-z.
Opanasopit P, Ruktanonchai U, Suwantong O, Panomsuk S, Ngawhirunpat T, Sittisombut C, et al. Electrospun poly(vinyl alcohol) fiber mats as carriers for extracts from the fruit hull of mangosteen. J Cosmet Sci. 2008;59:233–42.
Chen DW, Liao JY, Liu SJ, Chan EC. Novel biodegradable sandwich structured nanofibrous drug-eluting membranes for repair of infected wounds: an in vitro and in vivo study. Int J Nanomedicine. 2012;7:763–71. https://doi.org/10.2147/IJN.S29119.
Wu XM, Branford-White CJ, Zhu LM, Chatterton NP, Yu DG. Ester prodrug-loaded electrospun cellulose acetate fiber mats as transdermal drug delivery systems. J Mater Sci Mater Med. 2010;21:2403–11. https://doi.org/10.1007/s10856-010-4100-y.
Janairo RR, Henry JJ, Lee BL, Hashi CK, Derugin N, Lee R, et al. Heparin-modified small-diameter nanofibrous vascular grafts. IEEE Trans Nanobioscience. 2012;11:22–7. https://doi.org/10.1109/tnb.2012.2188926.
Prabaharan M, Jayakumar R, Nair SV. Electrospun nanofibrous scaffolds-current status and prospects in drug delivery. In: Jayakumar R, Nair S, editors. Biomedical applications of polymeric nanofibers, Advances in polymer science. Berlin: Springer; 2011. p. 241–62.
Zhang Y, Lim CT, Ramakrishna S, Huang ZM. Recent development of polymer nanofibers for biomedical and biotechnological applications. J Mater Sci Mater Med. 2005;16:933–46. https://doi.org/10.1007/s10856-005-4428-x.
Sukhwinder KB, Harpal SB. Perspectives on nanofiber dressings for the localized delivery of botanical remedies in wound healing. AIMS Mater Sci. 2017;4:370–82. https://doi.org/10.3934/matersci.2017.2.370.
Abdelgawada AM, Hudsona SM, Rojasb OJ. Antimicrobial wound dressing nanofiber mats from multicomponent (chitosan/silver-NPs/polyvinyl alcohol) systems. Carbohydr Polym. 2014;100:166–78. https://doi.org/10.1016/j.carbpol.2012.12.043.
Kalaipriya M, Sridhar R, Sundarrajan S, Venugopal JR, Ramakrishna S. Vitamin B12 loaded poly caprolactone nanofibers: a novel transdermal route for the water soluble energy supplement delivery. Int J Pharm. 2013;444:70–6.
Olaru N, Olaru L, Tudorachi N, Simona D, Manuela P. Nanostructures of cellulose acetate phthalate obtained by electrospinning from 2-methoxyethanol-containingsolvent systems: morphological aspects thermal behavior, and antimicrobial activity. Ind Eng Chem Res. 2013;52:696–705. https://doi.org/10.1021/ie301299d.
Zhang YZ, Feng Y, Huang ZM, Ramakrishna S, Lim CT. Fabrication of porous electrospun nanofibers. Nanotechnology. 2006;17:901–8. https://doi.org/10.1088/0957-4484/17/3/047.
Sugiyama Y, Kawakishi S, Osawa T. Involvement of the β-diketone moiety in the antioidative mechanism of tetrahydrocurcumin. Biochem Pharmacol. 1996;52:519–25. https://doi.org/10.1016/0006-2952(96)00302-4.
Mahendra Kumar T, Mayank G, Sambhu CM, Snehasis J. Protective effects of tetrahydrocurcumin (THC) on fibroblast and melanoma cell lines in vitro: it’s implication for wound healing. J Food Sci Technol. 2017;54:1137–45. https://doi.org/10.1007/s13197-017-2525-8.
Saipin S, Wichan K, Duangkhae M, Ruedeekorn W. Controlled release of oral tetrahydrocurcumin from a novel self-emulsifying floating drug delivery system (SEFDDS). AAPS PharmSciTech. 2011;12:152–64. https://doi.org/10.1208/s12249-010-9568-8.
Wu JC, Tsai ML, Lai CS, Wang YJ, Ho CT, Pan MH. Chemopreventative effects of tetrahydrocurcumin on human diseases. Food Funct. 2014;5:12–27. https://doi.org/10.1039/c3fo60370a.
Shrestha R, Palat A, Punnoose AM, Joshi S, Ponraju D, Paul SFD. Electrospun cellulose acetate phthalate nanofibrous scaffolds fabricated using novel solvent combinations biocompatible for primary chondrocytes and neurons. Tissue Cell. 2016;18:634–43. https://doi.org/10.1016/j.tice.2016.07.007.
Xiaochen GU, Sreeneeranj K, Daryl JF, Frank JB. In-vitro permeation of the insect repellent N,N-diethyl-m-toluamide (DEET) and the sunscreen oxybenzone. J Pharm Pharmacol. 2004;56:621–8. https://doi.org/10.1211/0022357023402.
Prashant MS, Suniket VF, Avinash KD. Evaluation of polymerized rosin for the formulation and development of transdermal drug delivery system: a technical note. AAPS PharmSciTech. 2005;6:E649–54. https://doi.org/10.1208/pt060481.
Venkata RY, Satya LJ, Natarajan J, Senthil V, Gowthamarajan K, Sumeet S, et al. A novel vesicular transdermal delivery of nifedipine—preparation, characterization and in vitro/in-vivo evaluation. Drug Delivery. 2016;23:619–30. https://doi.org/10.3109/10717544.2014.931484.
Shiow-Fern Ng JJR, Francis DS, Victor M, Gillian ME. Validation of a static Franz diffusion cell system for in vitro permeation studies. AAPS PharmSciTech. 2010;11:1432–41. https://doi.org/10.1208/s12249-010-9522-9.
Rolland A, Demichelis G, Jamoulle JC, Shroot B. Influence of formulation, receptor fluid, and occlusion, on in vitro drug release from topical dosage forms, using an automated flow-through diffusion cell. Pharm Res. 1992;9:82–6. https://doi.org/10.1023/A:1018935912097.
Olaru N, Olaru L. Electrospinning of cellulose acetate phthalate from different solvent systems. Ind Eng Chem Res. 2010;49:1953–7. https://doi.org/10.1021/ie901427f.
Ravikumar R, Ganesh M, Ubaidulla U, Choi EY, Jang HT. Preparation, characterization, and in vitro diffusion study of nonwoven electrospun nanofiber of curcumin-loaded cellulose acetate phthalate polymer. Saudi Pharm J. 2017;25:921–6. https://doi.org/10.1016/j.jsps.2017.02.004.
Nezarati RM, Eifert MB, Cosgriff-Hernandez E. Effects of humidity and solution viscosity on electrospun Fiber morphology. Tissue Eng Part C Methods. 2013;19:810–9. https://doi.org/10.1089/ten.tec.2012.0671.
Manjunath L, Sailaja RRN. PMMA—cellulose acetate phthalate nano composites reinforced with silane-treated nanoclay. Cellulose. 2014;21:1793–802. https://doi.org/10.1007/s10570-014-0190-x.
Roxin P, Karlsson A, Singh SK. Characterization of cellulose acetate phthalate (CAP). Drug Dev Ind Pharm. 1998;24:1025–41. https://doi.org/10.3109/03639049809089946.
Rana IR, Ming MW, Amal HEK. Ketoprofen-loaded Eudragit electrospun nanofibers for the treatment of oral mucositis. Int J Nanomedicine. 2017;12:2335–51. https://doi.org/10.2147/IJN.S131253.
Zhaoyang X, Jianyu L, Huan Z, Xiangdong J, Chuang Y, Fei W, et al. Morphological and swelling behavior of cellulose nanofiber (CNF)/poly(vinyl alcohol) (PVA) hydrogels: poly(ethylene glycol) (PEG) as porogen. RSC Adv. 2016;6:43626–33. https://doi.org/10.1039/c6ra03620a.
Hayashi M, Nagai T, Nogami H. Factors affecting dissolution rate of cellulose acetate phthalate in aqueous solution. Chem Pharm Bull. 1970;18:2350–3.
Higuchi T. Rate of release of medicaments from ointment bases containing drugs in suspension. J Pharm Sci. 1961;50:874–5.
Ritger PL, Peppas NA. A simple equation for description of solute release. II. Fickian and anomalous release from swellable devices. J Control Release. 1987;5:37–42.
Acknowledgements
This work was supported by the Hanseo University Intramural Research Grant, South Korea (2017).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Electronic Supplementary Material
ESM 1
(DOC 221 kb)
Rights and permissions
About this article
Cite this article
Rramaswamy, R., Mani, G., Venkatachalam, S. et al. Preparation and Characterization of Tetrahydrocurcumin-Loaded Cellulose Acetate Phthalate/Polyethylene Glycol Electrospun Nanofibers. AAPS PharmSciTech 19, 3000–3008 (2018). https://doi.org/10.1208/s12249-018-1122-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-018-1122-0