Abstract
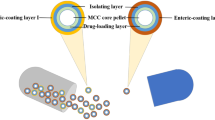
The objective of this study was to prepare ibuprofen enteric-coated sustained-release pellets (IB-SRPs) and codeine phosphate immediate-release pellets (CP-IRPs) to play a synergistic role in analgesia. The pellets were developed by extrusion-spheronization and fluidized bed coating technology. The single-factor investigation was used to determine the optimal prescription and process. The sustained-release membrane of IB-SRPs was water-insoluble ethyl cellulose (EC), triethyl citrate (TEC) was used as plasticizer, and hydroxypropyl methylcellulose (HPMCP) was chose as porogen. Besides, the immediate-release layer of CP-IRPs was gastric-soluble coating film. The ibuprofen and codeine phosphate compound capsules (IB-CP SRCs) were prepared by IB-SRPs and CP-IRPs packed together in capsules with the optimum doses of 200 and 13 mg, respectively. The prepared pellets were evaluated by scanning electron microscopy and dissolution test. Pharmacokinetic studies in beagle dogs indicated that the optimized IB-CP SRCs had smaller individual differences and better reproducibility comparing with commercial available tablets. Additionally, IB-CP SRCs achieved consistency with in vivo and in vitro tests. Therefore, IB-CP SRCs could play a great role in rapid and long-term analgesic.





Similar content being viewed by others
References
Dorner TE. Pain and chronic pain epidemiology : implications for clinical and public health fields. Wien Klin Wochenschr. 2018;130(1–2):1–3.
Donahue RE, Bradbury GR, Zychowicz ME, Muckler VC. Multimodal perioperative analgesia regimen to improve patient outcomes after Total knee arthroplasty: a multidisciplinary quality improvement project. J Perianesth Nurs. 2018;33(2):138–52.
Golladay GJ, Balch KR, Dalury DF, Satpathy J, Jiranek WA. Oral multimodal analgesia for total joint arthroplasty. J Arthroplast. 2017;32(9S):S69–73.
Lopes DG, Garsuch V, Becker K, Paudel A, Stehr M, Zimmer A, et al. Improving the granule strength of roller-compacted ibuprofen sodium for hot-melt coating processing. Int J Pharm. 2016;510(1):285–95.
Byrne GA, Wood PJ, Spark MJ. Non-prescription supply of combination analgesics containing codeine in community pharmacy: a simulated patient study. Res Social Adm Pharm. 2018;14(1):96–105.
Hamer AM, Spark MJ, Wood PJ, Roberts E. The upscheduling of combination analgesics containing codeine: the impact on the practice of pharmacists. Res Soc Adm Pharm. 2014;10(4):669–78.
Gibbins AK, Wood PJ, Spark MJ. Managing inappropriate use of non-prescription combination analgesics containing codeine: a modified Delphi study. Res Social Adm Pharm. 2017;13(2):369–77.
Foley M, Harris R, Rich E, Rapca A, Bergin M, Norman I, et al. The availability of over-the-counter codeine medicines across the European Union. Public Health. 2015;129(11):1465–70.
Le May S, Gouin S, Fortin C, Messier A, Robert MA, Julien M. Efficacy of an ibuprofen/codeine combination for pain management in children presenting to the emergency department with a limb injury: a pilot study. J Emerg Med. 2013;44(2):536–42.
McLaughlin DC, Cheah JW, Aleshi P, Zhang AL, Ma CB, Feeley BT. Multimodal analgesia decreases opioid consumption after shoulder arthroplasty: a prospective cohort study. J Shoulder Elb Surg. 2018;27(4):686–91.
Tornero Tornero C, Fernández Rodríguez LE, Orduña Valls J. Multimodal analgesia and regional anaesthesia. Revista Española de Anestesiología y Reanimación (English Edition). 2017;64(7):401–5.
Singla NK, Meske DS, Desjardins PJ. Exploring the interplay between rescue drugs, data imputation, and study outcomes: conceptual review and qualitative analysis of an acute pain data set. Pain and Therapy. 2017;6(2):165–75.
Renton T. Chronic orofacial pain. Oral Dis. 2017;23(5):566–71.
Sun Z, Yuan S, Zhao H, Wang Z, Liu Z. Preparation and evaluation of 1-deoxynojirimycin sustained-release pellets vs conventional immediate-release tablets. J Microencapsul. 2017;34(3):293–8.
Verstraete G, De Jaeghere W, Vercruysse J, Grymonpre W, Vanhoorne V, Stauffer F, et al. The use of partially hydrolysed polyvinyl alcohol for the production of high drug-loaded sustained release pellets via extrusion-spheronisation and coating: in vitro and in vivo evaluation. Int J Pharm. 2017;517(1–2):88–95.
Abbaspour MR, Sadeghi F, Garekani HA. Preparation and characterization of ibuprofen pellets based on Eudragit RS PO and RL PO or their combination. Int J Pharm. 2005;303(1–2):88–94.
Gouyon JB, Kibleur Y. Efficacy and tolerability of enteral formulations of ibuprofen in the treatment of patent ductus arteriosus in preterm infants. Clin Ther. 2010;32(10):1740–8.
Qazi F, Shoaib MH, Yousuf RI, Nasiri MI, Ahmed K, Ahmad M. Lipids bearing extruded-spheronized pellets for extended release of poorly soluble antiemetic agent-meclizine HCl. Lipids in Health & Dis. 2017;16(1):75.
Zhao X, Li G, Zhang L, Tao X, Guan T, Hong M, et al. Preparation and evaluation of nicotinic acid sustained-release pellets combined with immediate release simvastatin. Int J Pharm. 2010;400(1–2):42–8.
Varca GH, Lopes PS, Ferraz HG. Development of papain containing pellets produced by extrusion-spheronization: an operational stage approach. Drug Dev Ind Pharm. 2015;41(3):430–5.
de Barros JM, Lechner T, Charalampopoulos D, Khutoryanskiy VV, Edwards AD. Enteric coated spheres produced by extrusion/spheronization provide effective gastric protection and efficient release of live therapeutic bacteria. Int J Pharm. 2015;493(1–2):483–94.
Gao Y, Hong Y, Xian J, Lin X, Shen L, Zhang X, et al. A protocol for the classification of wet mass in extrusion-spheronization. Eur J Pharm Biopharm: Off J Arbeitsgemeinschaft fur Pharmazeutische Verfahrenstechnik eV. 2013;85(3 Pt B):996–1005.
Zhang S, Yan H, Yu P, Xia Y, Zhang W, Liu J. Development of protocatechualdehyde proliposomes-based sustained-release pellets with improved bioavailability and desired pharmacokinetic behavior for angina chronotherapy. Eur. J. Pharm. Sci. : Off J Eur Fed Pharm Sci. 2016;93:341–50.
Matji A, Carvajal L, Conde F, Peña MA, Donato N, Serrano DR, et al. Effect of the characteristics of raw material ibuprofen on roller compaction and dissolution. J Drug Delivery Sci Technol. 2017;42:237–44.
Ibrahim MA, El-Badry M. Formulation of immediate release pellets containing famotidine solid dispersions. Saudi Pharm J. 2014;22(2):149–56.
Abbaspour MR, Sadeghi F, Afrasiabi GH. Design and study of ibuprofen disintegrating sustained-release tablets comprising coated pellets. Eur J Pharm Biopharm : official journal of Arbeitsgemeinschaft fur Pharmazeutische Verfahrenstechnik eV. 2008;68(3):747–59.
Dukic-Ott A, Remon JP, Foreman P, Vervaet C. Immediate release of poorly soluble drugs from starch-based pellets prepared via extrusion/spheronisation. Eur J Pharm Biopharm : official journal of Arbeitsgemeinschaft fur Pharmazeutische Verfahrenstechnik eV. 2007;67(3):715–24.
Główka FK. Stereoselective pharmacokinetics of ibuprofen and its lysinate from suppositories in rabbits. Int J Pharm. 2000;199(2):159–66.
Boscarino C, Edginton AN, Peng H, Riggs KW, Szeitz A, Cheung B. The effect of operational stressors on ibuprofen pharmacokinetics. Eur J Clin Pharmacol. 2013;69(1):31–41.
Li Y, Zhang Y, Zhu C-Y. Pharmacokinetics and correlation between in vitro release and in vivo absorption of bio-adhesive pellets of panax notoginseng saponins. Chin J Nat Med. 2017;15(2):142–51.
Kapil R, Nolting A, Roy P, Fiske W, Benedek I, Abramowitz W. Pharmacokinetic properties of combination oxycodone plus racemic ibuprofen: two randomized, open-label, crossover studies in healthy adult volunteers. Clin Ther. 2004;26(12):2015–25.
Przejczowska-Pomierny K, Wlodyka M, Cios A, Wyska E. Enantioselective analysis of ibuprofen enantiomers in mice plasma and tissues by high-performance liquid chromatography with fluorescence detection: application to a pharmacokinetic study. Chirality. 2017;29(9):500–11.
Gu Z, Shi X, Omari-Siaw E, Zhu Y, Li H, Guo M, et al. Self-microemulsifying sustained-release pellet of Ginkgo biloba extract: preparation, in vitro drug release and pharmacokinetics study in beagle dogs. J Drug Delivery Sci Technol. 2017;37:184–93.
Zhang Y, Huang Z, Omari-Siaw E, Lu S, Zhu Y, Jiang D, et al. Preparation and in vitro-in vivo evaluation of sustained-release matrix pellets of capsaicin to enhance the oral bioavailability. AAPS PharmSciTech. 2016;17(2):339–49.
Zhang X, Li Q, Ye M, Zhao Z, Sun J, Yang X, et al. Preparation, characterization and in vitro / in vivo evaluation of oral time-controlled release etodolac pellets. AAPS PharmSciTech. 2017.
Yan H-X, Li J, Li Z-H, Zhang W-L, Liu J-P. Tanshinone IIA-loaded pellets developed for angina chronotherapy: deconvolution-based formulation design and optimization, pharmacokinetic and pharmacodynamic evaluation. Eur J Pharm Sci. 2015;76:156–64.
Yu P, Zhang S, Zhang W, Yang J, Lu J, Liu J. Tanshinol sustained-release pellets with absorption enhancer: optimization, characterization, pharmacokinetics and pharmacodynamics. Drug Dev Ind Pharm. 2017;43(7):1093–102.
Zhang S, Xia Y, Yan H, Zhang Y, Zhang W, Liu J. The water holding ability of powder masses: characterization and influence on the preparation of pellets via extrusion/spheronization. Powder Technol. 2016;301:940–8.
Goyanes A, Martinez-Pacheco R. New co-processed MCC-based excipient for fast release of low solubility drugs from pellets prepared by extrusion-spheronization. Drug Dev Ind Pharm. 2015;41(3):362–8.
Wang S, Wang Y, Luo Y, Liu Y, Su W. In vitro and in vivo évaluation of naringin sustained-release pellets compared with immediate-release tablets. J. Drug Delivery Sci. Technol. 2013;23(5):459–64.
Sonaglio D, Beringhs AO, Porfírio A, Bataille B. On the factors influencing the extrusion strain, particle size and dissolution behavior of multiparticulate systems obtained by extrusion/spheronization. Powder Technol. 2012;230:54–62.
Muley S, Nandgude T, Poddar S. Extrusion–spheronization a promising pelletization technique: in-depth review. Asian J Pharm Sci. 2016;11(6):684–99.
Podczeck F, Knight PE, Newton JM. The evaluation of modified microcrystalline cellulose for the preparation of pellets with high drug loading by extrusion/spheronization. Int J Pharm. 2008;350(1–2):145–54.
López EV, Luzardo Álvarez A, Blanco Méndez J, Otero Espinar FJ. Cellulose-polysaccharide film-coating of cyclodextrin based pellets for controlled drug release. J Drug Delivery Sci Technol. 2017;42:273–83.
Mallipeddi R, Saripella KK, Neau SH. Use of fine particle ethylcellulose as the diluent in the production of pellets by extrusion-spheronization. Saudi Pharm J. 2014;22(4):360–72.
Nasiri MI, Yousuf RI, Shoaib MH, Fayyaz M, Qazi F, Ahmed K. Investigation on release of highly water soluble drug from matrix-coated pellets prepared by extrusion–spheronization technique. J Coat Technol Res. 2016;13(2):333–44.
Chen T, Li J, Sun CC, Zheng Y. Tablets of multi-unit pellet system for controlled drug delivery. J Control Release. 2017;262:222–31.
Kuang C, Sun Y, Li B, Fan R, Zhang J, Yao Y, et al. Preparation and evaluation of duloxetine hydrochloride enteric-coated pellets with different enteric polymers. Asian J Pharm Sci. 2017;12(3):216–26.
Sheon CP, Nallamolu S, Heng PWS. A study on the plasticization of sustained release coatings for their ability to withstand the damaging effects of compaction on coated pellets. Asian J Pharm Sci. 2016;11(1):209–10.
Wlosnewski JC, Kumpugdee-Vollrath M, Sriamornsak P. Effect of drying technique and disintegrant on physical properties and drug release behavior of microcrystalline cellulose-based pellets prepared by extrusion/spheronization. Chem Eng Res Des. 2010;88(1):100–8.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Dong, L., Yang, F., Zhu, Z. et al. Preparation, Characterization and Pharmacokinetics Evaluation of the Compound Capsules of Ibuprofen Enteric-Coated Sustained-Release Pellets and Codeine Phosphate Immediate-Release Pellets. AAPS PharmSciTech 19, 3057–3066 (2018). https://doi.org/10.1208/s12249-018-1119-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-018-1119-8