Abstract
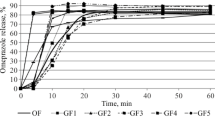
Despite extensive research in the field of gastroretentive dosage forms, this “holy grail” of oral drug delivery yet remained an unmet goal. Especially under fasting conditions, the reproducible retention of dosage forms in the stomach seems to be an impossible task. This is why such systems are often advised to be taken together with food. But also the postprandial motility can contribute significantly to the failure of gastroretentive dosage forms. To investigate the influence of postprandial pressure conditions on drug release from such systems, we used a novel in vitro dissolution tool, the dissolution stress test device. With the aid of this device, we simulated three different intragastric pressure profiles that may occur after postprandial intake. These transit scenarios were based on recently obtained, postprandial SmartPill® data. The tested systems, Glumetza® 1000 and Madopar® HBS 125, are marketed dosage forms that are based on different approaches to achieve proper gastric retention. All three transit scenarios revealed a highly pressure-sensitive drug release behavior, for both drugs. For Madopar® HBS 125, nearly complete drug release was observed even after early occurring pressures. Glumetza® 1000 seemed to be more resistant to these, most likely due to incomplete wetting of the system. On the contrary to these findings, data from standard dissolution tests using the paddle apparatus displayed controlled drug release for both systems for about 6 h. Based on these results, it can be doubted that established gastroretentive systems stay intact over a longer period of time, even under postprandial conditions.





Similar content being viewed by others
References
Lopes CM, Bettencourt C, Rossi A, Buttini F, Barata P. Overview on gastroretentive drug delivery systems for improving drug bioavailability. Int J Pharm. 2016;510(1):144–58. https://doi.org/10.1016/j.ijpharm.2016.05.016.
Streubel A, Siepmann J, Bodmeier R. Gastroretentive drug delivery systems. Expert Opin Drug Deliv. 2006;3(2):217–33. https://doi.org/10.1517/17425247.3.2.217.
Awasthi R, Kulkarni GT. Decades of research in drug targeting to the upper gastrointestinal tract using gastroretention technologies: where do we stand? Drug Deliv. 2016;23(2):378–94. https://doi.org/10.3109/10717544.2014.936535.
Koziolek M, Garbacz G, Neumann M, Weitschies W. Simulating the postprandial stomach: physiological considerations for dissolution and release testing. Mol Pharm. 2013;10:1610–22. https://doi.org/10.1021/mp300604u.
Malagelada JR, Go VL, Summerskill WHJ. Different gastric, pancreatic, and biliary responses to solid-liquid or homogenized meals. Dig Dis Sci. 1979;24(2):101–10. https://doi.org/10.1007/BF01324736.
Sauter MM, Steingoetter A, Curcic J, Treier R, Kuyumcu S, Fried M, et al. Quantification of meal induced gastric secretion and its effect on caloric emptying by magnetic resonance imaging (MRI). Gastroenterology. 2011;140:297.
Goetze O, Treier R, Fox M, Steingoetter A, Fried M, Boesiger P, et al. The effect of gastric secretion on gastric physiology and emptying in the fasted and fed state assessed by magnetic resonance imaging. Neurogastroenterol Motil. 2009;21(7):725–e72. https://doi.org/10.1111/j.1365-2982.2009.01293.x.
Mudie DM, Murray K, Hoad CL, Pritchard SE, Garnett MC, Amidon GL, et al. Quantification of gastrointestinal liquid volumes and distribution following a 240 mL dose of water in the fasted state. Mol Pharm. 2014;11(9):3039–47. https://doi.org/10.1021/mp500210c.
Schiller C, Fröhlich CP, Giessmann T, Siegmund W, Mönnikes H, Hosten N, et al. Intestinal fluid volumes and transit of dosage forms as assessed by magnetic resonance imaging. Aliment Pharmacol Ther. 2005;22(10):971–9. https://doi.org/10.1111/j.1365-2036.2005.02683.x.
Cassilly D, Kantor S, Knight LC, Maurer AH, Fisher RS, Semler J, et al. Gastric emptying of a non-digestible solid: assessment with simultaneous SmartPill pH and pressure capsule, antroduodenal manometry, gastric emptying scintigraphy. Neurogastroenterol Motil. 2008;20(4):311–9. https://doi.org/10.1111/j.1365-2982.2007.01061.x.
Deloose E, Janssen P, Depoortere I, Tack J. The migrating motor complex: control mechanisms and its role in health and disease. Nat Rev Gastroenterol Hepatol. 2012;9(5):271–85. https://doi.org/10.1038/nrgastro.2012.57.
Mojaverian P, Ferguson RK, Vlasses PH, Rocci ML, Oren A, Fix JA, et al. Estimation of gastric residence time of the Heidelberg capsule in humans: effect of varying food composition. Gastroenterology. 1985;89(2):392–7. https://doi.org/10.1016/0016-5085(85)90342-7.
Waterman KC. A critical review of gastric retentive controlled drug delivery. Pharm Dev Technol. 2007;12(1):1–10. https://doi.org/10.1080/10837450601168680.
Kararli TT. Comparison of the gastrointestinal anatomy, physiology, and biochemistry of humans and commonly used laboratory animals. Biopharm Drug Dispos. 1995;16(5):351–80. https://doi.org/10.1002/bdd.2510160502.
Garbacz G, Wedemeyer RS, Nagel S, Giessmann T, Mönnikes H, Wilson CG, et al. Irregular absorption profiles observed from diclofenac extended release tablets can be predicted using a dissolution test apparatus that mimics in vivo physical stresses. Eur J Pharm Biopharm. 2008;70(2):421–8. https://doi.org/10.1016/j.ejpb.2008.05.029.
Kamba M, Seta Y, Kusai A, Ikeda M, Nishimura K. A unique dosage form to evaluate the mechanical destructive force in the gastrointestinal tract. Int J Pharm. 2000;208(1):61–70. https://doi.org/10.1016/S0378-5173(00)00552-4.
Koziolek M, Schneider F, Grimm M, Modeβ C, Seekamp A, Roustom T, et al. Intragastric pH and pressure profiles after intake of the high-caloric, high-fat meal as used for food effect studies. J Control Release. 2015;220:71–8. https://doi.org/10.1016/j.jconrel.2015.10.022.
Garbacz G, Golke B, Wedemeyer RS, Axell M, Söderlind E, Abrahamsson B, et al. Comparison of dissolution profiles obtained from nifedipine extended release once a day products using different dissolution test apparatuses. Eur J Pharm Sci. 2009;38(2):147–55. https://doi.org/10.1016/j.ejps.2009.06.010.
Schneider F, Grimm M, Koziolek M, Modeß C, Dokter A, Roustom T, et al. Resolving the physiological conditions in bioavailability and bioequivalence studies: comparison of fasted and fed state. Eur J Pharm Biopharm. 2016;108:214–9. https://doi.org/10.1016/j.ejpb.2016.09.009.
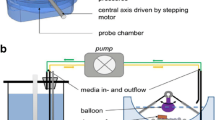
Schneider F, Beeck R, Hoppe M, Koziolek M, Weitschies W. In vitro simulation of realistic gastric pressure profiles. Eur J Pharm Sci. 2017;107:71–7. https://doi.org/10.1016/j.ejps.2017.06.037.
Homšek I, Parojčić J, Dačević M, Petrović L, Jovanović D. Justification of metformin hydrochloride biowaiver criteria based on bioequivalence study. Arzneimittelforschung. 2010;60(9):553–9. https://doi.org/10.1055/s-0031-1296324.
Adis Editorial. Metformin extended release—DepoMed. Drugs R & D. 2004;5(4):231–3. https://doi.org/10.2165/00126839-200405040-00009.
Hair PI, Curran MP, Keam SJ. Tramadol extended-release tablets in moderate to moderately severe chronic pain in adults. CNS Drugs. 2007;21(3):259–63. https://doi.org/10.2165/00023210-200721030-00005.
Kasim NA, Whitehouse M, Ramachandran C, Bermejo M, Lennernäs H, Hussain AS, et al. Molecular properties of WHO essential drugs and provisional biopharmaceutical classification. Mol Pharm. 2004;1(1):85–96. https://doi.org/10.1021/mp034006h.
Erni W, Held K. The hydrodynamically balanced system: a novel principle of controlled drug release. Eur Neurol. 1987;27:21–7. https://doi.org/10.1159/000116171.
Mandal UK, Chatterjee B, Senjoti FG. Gastro-retentive drug delivery systems and their in vivo success: a recent update. Asian Journal of Pharmaceutical Sciences. 2016;11(5):575–84. https://doi.org/10.1016/j.ajps.2016.04.007.
Hatton GB, Yadav V, Basit AW, Merchant HA. Animal farm: considerations in animal gastrointestinal physiology and relevance to drug delivery in humans. J Pharm Sci. 2015;104(9):2747–76. https://doi.org/10.1002/jps.24365.
Ewe K, Press AG, Bollen S, Schuhn I. Gastric emptying of indigestible tablets in relation to composition and time of ingestion of meals studied by metal detector. Dig Dis Sci. 1991;36(2):146–52. https://doi.org/10.1007/BF01300748.
Schwartz SL, Gordi T, Hou E, Cramer M, Heritier M, Cowles VE. Clinical development of metformin extended-release tablets for type 2 diabetes: an overview. Expert Opin Drug Metab Toxicol. 2008;4(9):1235–43. https://doi.org/10.1517/17425255.4.9.1235.
Berner B, Cowles VE. Case studies in swelling polymeric gastric retentive tablets. Expert Opin Drug Deliv. 2006;3(4):541–8. https://doi.org/10.1517/17425247.3.4.541.
Nakagawa T, Kondo SI, Sasai Y, Kuzuya M. Preparation of floating drug delivery system by plasma technique. Chem Pharm Bull. 2006;54(4):514–8. https://doi.org/10.1248/cpb.54.514.
Aoki S, Uesugi K, Tatsuishi K, Ozawa H, Kayano M. Evaluation of the correlation between in vivo and in vitro release of phenylpropanolamine HCl from controlled-release tablets. Int J Pharm. 1992;85(1–3):65–73. https://doi.org/10.1016/0378-5173(92)90135-O.
Aoki S, Ando H, Tatsuishi K, Uesugi K, Ozawa H. Determination of the mechanical impact force in the in vitro dissolution test and evaluation of the correlation between in vivo and in vitro release. Int J Pharm. 1993;95(1–3):67–75. https://doi.org/10.1016/0378-5173(93)90391-R.
Brun R, Michalek W, Surjanhata BC, Parkman HP, Semler JR, Kuo B. Comparative analysis of phase III migrating motor complexes in stomach and small bowel using wireless motility capsule and antroduodenal manometry. Neurogastroenterol Motil. 2012;24(4):332–e165. https://doi.org/10.1111/j.1365-2982.2011.01862.x.
Desipio J, Friedenberg FK, Korimilli A, Richter JE, Parkman HP, Fisher RS. High-resolution solid-state manometry of the antropyloroduodenal region. Neurogastroenterol Motil. 2007;19(3):188–95. https://doi.org/10.1111/j.1365-2982.2006.00866.x.
Garbacz G, Cadé D, Benameur H, Weitschies W. Bio-relevant dissolution testing of hard capsules prepared from different shell materials using the dynamic open flow through test apparatus. Eur J Pharm Sci. 2014;57:264–72. https://doi.org/10.1016/j.ejps.2013.08.039.
Koziolek M, Grimm M, Garbacz G, Kühn JP, Weitschies W. Intragastric volume changes after intake of a high-caloric, high-fat standard breakfast in healthy human subjects investigated by MRI. Mol Pharm. 2014;11(5):1632–9. https://doi.org/10.1021/mp500022u.
Grahnen A, Eckernäs SÅ, Collin C, Ling-Andersson A, Tiger G, Nilsson M. Comparative multiple-dose pharmacokinetics of controlled-release levodopa products. Eur Neurol. 1992;32(6):343–8. https://doi.org/10.1159/000116858.
Funding
This work has received financial support from the Innovative Medicines Initiative Joint Undertaking (http://www.imi.europa.eu) under Grant Agreement No. 115369, resources of which are composed of financial contribution from the European Union Seventh Framework Program and EFPIA companies in kind contribution, as well as from the Federal Ministry of Education and Research, Germany (FKZ 03IPT612X, InnoProfile-Transfer).
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editor: Sandra Klein
Rights and permissions
About this article
Cite this article
Schneider, F., Hoppe, M., Koziolek, M. et al. Influence of Postprandial Intragastric Pressures on Drug Release from Gastroretentive Dosage Forms. AAPS PharmSciTech 19, 2843–2850 (2018). https://doi.org/10.1208/s12249-018-1022-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-018-1022-3