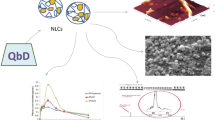
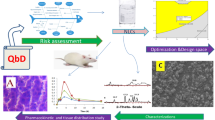
Abstract
Statins are widely prescribed for hyperlipidemia, cancer, and Alzheimer’s disease but are facing some inherent challenges such as low solubility and drug loading, higher hepatic metabolism, as well as instability at gastric pH. So, relatively higher circulating dose, required for exerting the therapeutic benefits, leads to dose-mediated severe toxicity. Furthermore, due to low biocompatibility, high toxicity, and other regulatory caveats such as product conformity, reproducibility, and stability of conventional formulations as well as preferentially higher bioabsorption of lipids in their favorable cuboidal geometry, enhancement in in vivo biopharmaceutical performance of Rosuvastatin could be well manifested in Quality by Design (QbD) integrated cuboidal-shaped mucoadhesive microcrystalline delivery systems (Limicubes). Here, quality-target-product-profile (QTPPs), critical quality attributes (CQAs), Ishikawa fishbone diagram, and integration of risk management through risk assessment matrix for failure mode and effects analysis (FMEA) followed by processing of Plackett-Burman design matrix using different statistical test for the first time established an approach to substantiate the claims that controlling levels of only these three screened out independent process variables, i.e., Monoolein (B = 800–1100 μL), Poloxamer (C = 150–200 mg), and stirring speed (F = 700–1000 rpm) were statistically significant to modulate and improve the biopharmaceutical performance affecting key attributes, viz., average particle size (Y1 = 1.40–2.70 μ), entrapment efficiency (Y2 = 62.60–88.80%), and drug loading (Y3 = 0.817–1.15%), in QbD-enabled process. The optimal performance of developed Limicubes exhibited an average particle size of 1.8 ± 0.2 μ, entrapment efficiency 80.32 ± 2.88%, and drug loading 0.93 ± 0.08% at the level of 1100 μL (+ 1), 200 mg (+ 1), and 700 rpm (− 1) for Monoolein, Poloxamer, and stirring speed, respectively.




Similar content being viewed by others
References
Beg S, Jain S, Kushwah V, Bhatti GK, Sandhu PS, Katare OP, et al. Novel surface-engineered solid lipid nanoparticles of Rosuvastatin calcium for low-density lipoprotein-receptor targeting: a quality by design-driven perspective. Nanomedicine. 2017;12(4):333–56. https://doi.org/10.2217/nnm-2016-0336.
Kostapanos MS, Milionis HJ, Elisaf MS. An overview of the extra-lipid effects of rosuvastatin. J Cardiovasc Pharmacol. 2008;13(3):157–74. https://doi.org/10.1177/1074248408318628.
Rizwanullah M, Amin S, Ahmad J. Improved pharmacokinetics and antihyperlipidemic efficacy of Rosuvastatin-loaded nanostructured lipid carriers. J Drug Target. 2017;25(1):58–74. https://doi.org/10.1080/1061186X.2016.1191080.
Khanfar M, Al-Nimry S. Stabilization and amorphization of Lovastatin using different types of silica. AAPS PharmSciTech. 2017;18(6):1–10.
Glycerol monooleate (gmo)—manufacturing plant, detailed project report, profile, business plan, industry trends, market research, survey, manufacturing process, machinery, raw materials, feasibility study, investment opportunities, cost and revenue. http://www.npcs.in/profile-project-reports/profile/1242/glycerol-monooleate-gmo-manufacturing-plant-detailed-project-report-profile-business-plan-industry-trends-market-research-survey-manufacturing-process-machinery-raw-materials-feasibility-study-investment-opportunities-cost-revenue.html (accessed on 18/09/2017).
Milak S, Zimmer A. Glycerol monooleate liquid crystalline phases used in drug delivery systems. Int J Pharm. 2015;478(2):569–87. https://doi.org/10.1016/j.ijpharm.2014.11.072.
Seddon JM and Templer RH. Polymorphism of lipid-water systems. In: Lipowsky R, Sackmann E, editor. Structure and dynamics of membranes: I. from cells to vesicles/II. Generic and specific interactions. Amsterdam: Elsevier; 1995. p. 97–160.
Ganem-Quintanar A, Quintanar-Guerrero D, Buri P. Monoolein: a review of the pharmaceutical applications. Drug Dev Ind Pharm. 2000;26(8):809–20. https://doi.org/10.1081/DDC-100101304.
Kulkarni CV, Wachter W, Iglesias-Salto G, Engelskirchen S, Ahualli S. Monoolein: a magic lipid? Phys Chem Chem Phys. 2011;13(8):3004–21. https://doi.org/10.1039/C0CP01539C.
Seedher N, Kanojia M. Micellar solubilization of some poorly soluble antidiabetic drugs: a technical note. AAPS PharmSciTech. 2008;9(2):431–6. https://doi.org/10.1208/s12249-008-9057-5.
Ahmad J, Kohli K, Mir SR, Amin S. Formulation of self-nanoemulsifying drug delivery system for Telmisartan with improved dissolution and oral bioavailability. J Dispers Sci Technol. 2011;32(7):958–68. https://doi.org/10.1080/01932691.2010.488511.
Collings PJ, Hird M. Introduction to a special phase of matter. In: Collings PJ, Hird M, editors. Introduction to liquid crystals: chemistry and physics, vol. 1: CRC Press; 1997. p. 1–16.
Collings PJ and Hird M. Lyotropic liquid crystals—anisotropic solutions. In: Collings PJ and Hird M, editors, Introduction to liquid crystals: chemistry and physics. CRC Press; 1997. 7:135–148.
Boyd BJ. Controlled release from cubic liquid-crystalline particles (cubosome). In: Lynch ML, Spicer PT, editors, Bicontinuous liquid crystals. CRC press; 2005. 10:286–303.
Dumortier G, Grossiord JL, Agnely F, Chaumeil JC. A review of Poloxamer 407 pharmaceutical and pharmacological characteristics. Pharm Res. 2006;23(12):2709–272. https://doi.org/10.1007/s11095-006-9104-4.
Wang TY, Liu M, Portincasa P, Wang DQH. New insights into the molecular mechanism of intestinal fatty acid absorption. Eur J Clin Investig. 2013;43(11):1203–23.
Semis R, Nahmias M, Lev S, Frenkel M, Segal E. Evaluation of antifungal combinations of nystatin-intralipid against Aspergillus terreus using checkerboard and disk diffusion methods. J Mycol Méd. 2015;25(1):63–70. https://doi.org/10.1016/j.mycmed.2014.12.002.
Edward RR, Innes JK, Marino LV, Calder PC. Influence of different intravenous lipid emulsions on growth, development and laboratory and clinical outcomes in hospitalised paediatric patients: a systematic review. Clin Nutr. 2017; https://doi.org/10.1016/j.clnu.2017.07.003.
Rothschild L, Bern S, Oswald S, Weinberg G. Intravenous lipid emulsion in clinical toxicology. Scand J Trauma Resusc Emerg Med. 2010;18(1):51. https://doi.org/10.1186/1757-7241-18-51.
Newton MJ, Harjot K. Flunarizine dihydrochloride nanoemulsion for migraine nasal drug delivery: fabrication, characterization and in vitro study. Drug Deliv Lett. 2016;6(2):104–12.
Kawish SM, Ahmed S, Gull A, Aslam M, Pandit J, Aqil M, et al. Development of Nabumetone loaded lipid nano-scaffold for the effective oral delivery; optimization, characterization, drug release and pharmacodynamic study. J Mol Liq. 2017;231:514–22. https://doi.org/10.1016/j.molliq.2017.01.107.
Brito Oliveira TC, Molina CV, Netto FM, Pinho SC. Encapsulation of betacarotene in lipid microparticles stabilized with hydrolyzed soy protein isolate: production parameters, alpha tocopherol coencapsulation and stability under stress conditions. J Food Sci. 2017;82(3):659–69. https://doi.org/10.1111/1750-3841.13642.
Rao Z, Si L, Guan Y, Pan H, Qiu J, Li G. Inhibitive effect of Cremophor RH40 or Tween 80-based self-microemulsiflying drug delivery system on cytochrome P450 3A enzymes in murine hepatocytes. J Huazhong Univ Sci Technolog Med Sci. 2010;30(5):562–8. https://doi.org/10.1007/s11596-010-0543-0.
Dudhipala N, Veerabrahma K. Improved anti-hyperlipidemic activity of Rosuvastatin calcium via lipid nanoparticles: pharmacokinetic and pharmacodynamic evaluation. Eur J Pharm Biopharm. 2017;110:47–57. https://doi.org/10.1016/j.ejpb.2016.10.022.
Pham MN, Van Vo T, Tran VT, Tran PH, Tran TT. Microemulsion-based mucoadhesive buccal wafers: wafer formation, in vitro release, and ex vivo evaluation. AAPS PharmSciTech. 2017;18(7):2727–36. https://doi.org/10.1208/s12249-017-0754-9.
Beg S, Sharma G, Thanki K, Jain S, Katare O, Singh B. Positively charged self-Nano emulsifying oily formulations of Olmesartan medoxomil: systematic development, in vitro, ex vivo and in vivo evaluation. Int J Pharm. 2015;493(1):466–82. https://doi.org/10.1016/j.ijpharm.2015.07.048.
Kothari BH, Fahmy R, Claycamp HG, Moore CM, Chatterjee S, Hoag SWA. Systematic approach of employing quality by design principles: risk assessment and design of experiments to demonstrate process understanding and identify the critical process parameters for coating of the Ethylcellulose pseudolatex dispersion using non-conventional fluid bed process. AAPS PharmSciTech. 2017;18(4):1135–57. https://doi.org/10.1208/s12249-016-0569-0.
Singh B, Kaur A, Dhiman S, Garg B, Khurana RK, Beg S. QbD-enabled development of novel stimuli-responsive gastroretentive systems of Acyclovir for improved patient compliance and biopharmaceutical performance. AAPS PharmSciTech. 2016;17(2):454–65. https://doi.org/10.1208/s12249-015-0367-0.
Shah B, Khunt D, Bhatt H, Misra M, Padh H. Intranasal delivery of Venlafaxine loaded nanostructured lipid carrier: risk assessment and QbD based optimization. J Drug Deliv Sci Technol. 2016;33:37–50. https://doi.org/10.1016/j.jddst.2016.03.008.
Akhtar N, Verma A, Pathak K. Feasibility of binary composition in development of nanoethosomal glycolic vesicles of triamcinolone acetonide using Box-behnken design: in vitro and ex vivo characterization. Artif Cells Nanomed Biotechnol. 2017;45(6):1123–31. https://doi.org/10.1080/21691401.2016.1202261.
Nguyen TH, Hanley T, Porter CJ, Boyd BJ. Nanostructured liquid crystalline particles provide long duration sustained-release effect for a poorly water soluble drug after oral administration. J Control Release. 2011;153(2):180–6. https://doi.org/10.1016/j.jconrel.2011.03.033.
Qadri GR, Ahad A, Aqil M, Imam SS, Ali A. Invasomes of Isradipine for enhanced transdermal delivery against hypertension: formulation, characterization, and in vivo pharmacodynamic study. Artif Cells Nanomed Biotechnol. 2017;45(1):139–45. https://doi.org/10.3109/21691401.2016.1138486.
Gomes GS, Maciel TR, Piegas EM, Michels LR, Colomé LM, Freddo RJ, et al. Optimization of curcuma oil/quinine-loaded nanocapsules for malaria treatment. AAPS PharmSciTech. 2017; https://doi.org/10.1208/s12249-017-0854-6.
Teixeira MT, Sá-Barreto LC, Gratieri T, Gelfuso GM, Silva IC, Cunha-Filho MS. Key technical aspects influencing the accuracy of tablet subdivision. AAPS PharmSciTech. 2017;18(4):1393–401. https://doi.org/10.1208/s12249-016-0615-y.
Yadav NK, Nanda S, Sharma G, Katare OP. Systematically optimized Ketoprofen-loaded novel proniosomal formulation for periodontitis: in vitro characterization and in vivo pharmacodynamic evaluation. AAPS PharmSciTech. 2017;18(5):1863–80. https://doi.org/10.1208/s12249-016-0665-1.
Ma L, Wang L, Tang J, Yang Z. Optimization of arsenic extraction in rice samples by Plackett Burman design and response surface methodology. Food Chem. 2016;204:283–8. https://doi.org/10.1016/j.foodchem.2016.02.126.
Beg S, Jain A, Kaur R, Panda SS, Katare O, Singh B. QbD-driven development and validation of an efficient bioanalytical UPLC method for estimation of Olmesartan medoxomil. J Liq Chromatogr Relat Technol. 2016;39(13):587–97. https://doi.org/10.1080/10826076.2016.1206023.
Teixeira MT, Sá-Barreto LC, Gratieri T, Gelfuso GM, Silva IC, Cunha-Filho MS. Key technical aspects influencing the accuracy of tablet subdivision. AAPS PharmSciTech. 2017;18(4):1393–401. https://doi.org/10.1208/s12249-016-0615-y.
Wissing S, Kayser O, Müller R. Solid lipid nanoparticles for parenteral drug delivery. Adv Drug Deliv Rev. 2004;56(9):1257–72. https://doi.org/10.1016/j.addr.2003.12.002.
Rarokar NR, Saoji SD, Raut NA, Taksande JB, Khedekar PB, Dave VS. Nanostructured cubosomes in a thermoresponsive depot system: an alternative approach for the controlled delivery of Docetaxel. AAPS PharmSciTech. 2016;17(2):436–45. https://doi.org/10.1208/s12249-015-0369-y.
Moghddam SMM, Ahad A, Aqil M, Imam SS, Sultana Y. Optimization of nanostructured lipid carriers for topical delivery of Nimesulide using Box–Behnken design approach. Artif Cells Nanomed Biotechnol. 2017;45(3):617–24. https://doi.org/10.3109/21691401.2016.1167699.
Ha JM, Seo JW, Kim SH, Kim JY, Park CW, Rhee YS, et al. Implementation of quality by design for formulation of Rebamipide gastro-retentive tablet. AAPS PharmSciTech. 2017;18(8):3129–39. https://doi.org/10.1208/s12249-017-0797-y.
Beg S, Sandhu PS, Batra RS, Khurana RK, Singh B. QbD-based systematic development of novel optimized solid self-nanoemulsifying drug delivery systems (SNEDDS) of Lovastatin with enhanced biopharmaceutical performance. Drug Deliv. 2015;22(6):765–84. https://doi.org/10.3109/10717544.2014.900154.
Ahuja S, Ferreira G, Moreira A. Application of Plackett Burman design and response surface methodology to achieve exponential growth for aggregated shipworm bacterium. Biotechnol Bioeng. 2004;85(6):666–75. https://doi.org/10.1002/bit.10880.
Noguchi H, Ojima Y, Yasui S. A practical variable selection for linear models. In: Lenz HJ, Schmid W, Wilrich PT, editors, Frontiers in statistical quality control. Springer; 2012. 10. p. 349–60.
Claycamp HG, Kona R, Fahmy R, Hoag SW, Quality-by-design II. Application of quantitative risk analysis to the formulation of Ciprofloxacin tablets. AAPS PharmSciTech. 2016;17(2):233–44. https://doi.org/10.1208/s12249-015-0349-2.
ACKNOWLEDGEMENTS
The authors are thankful to the Department of Pharmaceutics, School of Pharmaceutical Education and Research (formerly Faculty of Pharmacy), Jamia Hamdard, New Delhi, India for providing all instrument facilities required for carrying out research effectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest.
Additional information
Highlights
• Monoolein-Poloxamer integrated novel biocompatible, mucoadhesive, and cuboidal-shaped microcrystalline system, Limicubes, of hydrophobic drug Rosuvastatin was assessed.
• Quality target product profile (QTPPs), critical quality attributes (CQAs), Ishikawa fishbone diagram, and risk assessment matrix for failure mode and effects analysis (FMEA) were generated for QbD-based product profile.
• Plackett-Burman design screened out Monoolein, Poloxamer, and stirring speed as vital factors which were further analyzed at their three different levels to optimize shape, size, entrapment efficiency, and drug loading.
• Monoolein enhanced drug entrapment, drug loading, and particle size, while higher loading and entrapment of drug with reduced particle size were manifested by Poloxamer.
• Although high stirring speed produced particles with lower dimension due to high association matrix with drug, drug loading and entrapment efficiency were statistically unaffected.
Rights and permissions
About this article
Cite this article
Javed, M.N., Kohli, K. & Amin, S. Risk Assessment Integrated QbD Approach for Development of Optimized Bicontinuous Mucoadhesive Limicubes for Oral Delivery of Rosuvastatin. AAPS PharmSciTech 19, 1377–1391 (2018). https://doi.org/10.1208/s12249-018-0951-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-018-0951-1