Abstract
The purpose of this research was to use inline real-time near-infrared (NIR) to measure the moisture content of granules manufactured using a commercial production scale continuous twin-screw granulator fluid-bed dryer milling process. A central composite response surface statistical design was used to study the effect of inlet air temperature and dew point on granule moisture content. The NIR moisture content was compared to Karl Fischer (KF) and loss on drying (LOD) moisture determinations. Using multivariate analysis, the data showed a statistically significant correlation between the conventional methods and NIR. The R 2 values for predicted moisture content by NIR versus KF and predicted moisture values by NIR versus LOD were 0.94 (p < 0.00001) and 0.85 (p < 0.0002), respectively. The adjusted R 2 for KF versus LOD correlation was 0.85 (p < 0.0001). Analysis of the response surface design data showed that inlet air temperature over a range of 35–55°C had a significant linear impact on granule moisture content as measured by predicted NIR (adjusted R 2 = 0.84, p < 0.02), KF (adjusted R 2 = 0.91, p < 0.0001), and LOD (adjusted R 2 = 0.85, p < 0.0006). The inlet air dew point range of 10–20°C did not have a significant impact on any of the moisture measurements.






Similar content being viewed by others
References
Herschel W. Investigation of the powers of the prismatic colours to heat and illuminate objects; with remarks, that prove the different refrangibility of radiant heat, to which is added, an inquiry into the method of viewing the sun advantageously, with telescopes of large apertures and high magnifying powers. Phil Trans Roy Soc. 1800;90:255–83.
Reich G. Near-infrared spectroscopy and imaging: basic priniciples and pharmaceutical applications. Adv Drug Delivery Rev. 2005;57:1109–43.
Swarbrick B. Process analytical technology: a strategy for keeping manufacturing viable in Australia. Vib Spectrosc. 2007;44:171–8.
Ufret C, Morris K. Modeling of powder blending using on-line near-infrared measurements. Drug Dev Ind Pharm. 2001;27:719–29.
Gupta A, Peck GE, Miller RW, Morris KR. Real-time near-infrared monitoring of content uniformity, moisture content, compact density, tensile strength, and Young’s modulus of roller compacted powder blends. J Pharm Sci. 2005;94:1589–97.
Gupta A, Peck GE, Miller RW, Morris KR. Influence of ambient moisture on the compaction behavior of microcrystalline cellulose powder undergoing uni-axial compression and roller compaction: a comparative study using near-infrared spectroscopy. J Pharm Sci. 2005;94:2301–13.
Morris KR, Stowell JG, Byrn SR, Placette AW, Davis TD, Peck GE. Accelerated fluid bed drying using NIR monitoring and phenomenological modeling. Drug Dev Ind Pharm. 2000;26:985–8.
Findlay WP, Peck GE, Morris KR. Determination of fluidized bed granulation end point using near-infrared spectroscopy and phenomenological analysis. J Pharm Sci. 2005;94:604–12.
Nieuwmeyer FJS, Damen M, Gerich A, Rusmini F, van der Voort Maarschalk K, Vromans H. Granule characterization during fluid bed drying by development of a near infrared method to determine water content and median granule size. Pharm Res. 2007;24:1854–61.
Tabasi SH, Fahmy R, Bensley D, O’Brien C, Hoag SW. Quality by design, part I: application of NIR spectroscopy to monitor tablet manufacturing process. J Pharm Sci. 2008;97:4040–51.
Tabasi SH, Fahmy R, Bensley D, O’Brien C, Hoag SW. Quality by design, part II: application of NIR spectroscopy to monitor the coating process for a pharmaceutical sustained release product. J Pharm Sci. 2008;97:4052–66.
Poli JE, Hoag SW, Flank S. Comparison of authentic and suspect pharmaceuticals. Pharm Technol. 2009;33:46–52.
U.S. FDA. Office of Pharmaceutical Science (OPS). Process analytical technology (PAT) initiative. http://www.fda.gov/cder/OPS/PAT.htm. Accessed 1 Mar 2011.
De Beer T, Burggraeve A, Fonteyne M, Saerens L, Remon JP, Vervaet C. Near infrared and Raman spectroscopy for the in-process monitoring of pharmaceutical production processes. Int J Pharm. 2010. doi:10.1016/j.ijphar.2010.12.012.
Simca-P+ version 11.5. Umetrics AB. Umea, Sweden.
JMP® 8 Design of experiments software. SAS Institute Inc. Cary, NC: SAS Publishing, 2008.
Acknowledgments
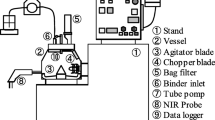
We appreciate the generous support of GlaxoSmithKline (GSK). We also appreciate the support of Mr. Scott Staton, operation and formulation manager at Campbell University Pharmaceutical Sciences Institute for editing the flow diagram of ConsiGma™ kit provided by GSK (Zebulon).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chablani, L., Taylor, M.K., Mehrotra, A. et al. Inline Real-Time Near-Infrared Granule Moisture Measurements of a Continuous Granulation–Drying–Milling Process. AAPS PharmSciTech 12, 1050–1055 (2011). https://doi.org/10.1208/s12249-011-9669-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-011-9669-z