Abstract
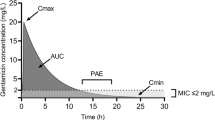
This study aimed to determine the optimal starting dose of gentamicin in paediatric oncology patients. A population pharmacokinetic model describing drug exposure, a semi-mechanistic model describing bacterial killing and an Emax model describing renal cortex accumulation were linked in a utility function using NONMEM®. The optimal gentamicin starting dose was estimated in patients aged from 0.1 to 18.2 years, by balancing the probability of efficacy on day 1 against relative renal function reduction on day 7 with continued dosing. Using achievement of a gentamicin area under the concentration time curve to bacterial minimum inhibitor concentration (MIC) ratio of ≥ 100 and maximum concentration to MIC ratio of ≥ 10 as the efficacy endpoints, a starting dose of 7.1, 9.5, 10.8 and 14.6 mg/kg/q24h was optimal at a MIC of 0.5, 1, 2 and 4 mg/L respectively, with ≥ 75% probability of obtainment. Using achievement of a 2-log10 bacterial count reduction at 24-h post-dose as the efficacy endpoint, a starting dose of 12.8 mg/kg/q24h was optimal, with 85.6% probability of obtainment. Under these different dosing scenarios, relative reduction in renal function ranged on average from 6.9 to 14.5% on day 7. The current recommended starting dose of gentamicin of 7.5 mg/kg/q24h may not be sufficient to achieve efficacy on day 1 if bacterial MIC is > 0.5 mg/L. A higher initial dose (up to 14.6 mg/kg/q24h), in less sensitive microorganisms, would likely cause only a relatively small reduction in renal function at day 7. Close monitoring is crucial if high doses are given, especially for longer than 7 days.





Similar content being viewed by others
References
Pizzo PA, Poplack DG, Ovid Technologies I. Principles and practice of pediatric oncology. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins Health; 2011.
Moore RD, Lietman PS, Smith CR. Clinical response to aminoglycoside therapy: importance of the ratio of peak concentration to minimal inhibitory concentration. J Infect Dis. 1987;155(1):93–9. https://doi.org/10.1093/infdis/155.1.93.
Kashuba AD, Nafziger AN, Drusano GL, Bertino JS Jr. Optimizing aminoglycoside therapy for nosocomial pneumonia caused by gram-negative bacteria. Antimicrob Agents Chemother. 1999;43(3):623–9.
Drusano GL, Liu W, Fikes S, Cirz R, Robbins N, Kurhanewicz S, et al. Interaction of drug- and granulocyte-mediated killing of Pseudomonas aeruginosa in a murine pneumonia model. J Infect Dis. 2014;210(8):1319–24. https://doi.org/10.1093/infdis/jiu237.
Mohamed AF, Nielsen EI, Cars O, Friberg LE. Pharmacokinetic-pharmacodynamic model for gentamicin and its adaptive resistance with predictions of dosing schedules in newborn infants. Antimicrob Agents Chemother. 2012;56(1):179–88. https://doi.org/10.1128/AAC.00694-11.
Zhuang L, He Y, Xia H, Liu Y, Sy SK, Derendorf H. Gentamicin dosing strategy in patients with end-stage renal disease receiving haemodialysis: evaluation using a semi-mechanistic pharmacokinetic/pharmacodynamic model. J Antimicrob Chemother. 2016;71(4):1012–21. https://doi.org/10.1093/jac/dkv428.
Spanggaard MH, Honge BL, Schonheyder HC, Nielsen H. Short-term gentamicin therapy and risk of renal toxicity in patients with bacteraemia. Scand J Infect Dis. 2011;43(11–12):953–6. https://doi.org/10.3109/00365548.2011.601755.
Paterson DL, Robson JM, Wagener MM. Risk factors for toxicity in elderly patients given aminoglycosides once daily. J Gen Intern Med. 1998;13(11):735–9. https://doi.org/10.1046/j.1525-1497.1998.00224.x.
Rybak MJ, Abate BJ, Kang SL, Ruffing MJ, Lerner SA, Drusano GL. Prospective evaluation of the effect of an aminoglycoside dosing regimen on rates of observed nephrotoxicity and ototoxicity. Antimicrob Agents Chemother. 1999;43(7):1549–55.
Rougier F, Claude D, Maurin M, Sedoglavic A, Ducher M, Corvaisier S, et al. Aminoglycoside nephrotoxicity: modeling, simulation, and control. Antimicrob Agents Chemother. 2003;47(3):1010–6. https://doi.org/10.1128/AAC.47.3.1010-1016.2003.
Croes S, Koop AH, van Gils SA, Neef C. Efficacy, nephrotoxicity and ototoxicity of aminoglycosides, mathematically modelled for modelling-supported therapeutic drug monitoring. Eur J Pharm Sci. 2012;45(1–2):90–100. https://doi.org/10.1016/j.ejps.2011.10.022.
Miron D. Once daily dosing of gentamicin in infants and children. Pediatr Infect Dis J. 2001;20(12):1169–73. https://doi.org/10.1097/00006454-200112000-00016.
Queensland Government. Tobramycin/gentamicin. Ther Drug Monit. 2016 [Available from: https://www.childrens.health.qld.gov.au/wp-content/uploads/PDF/ams/658202.pdf.]
Bialkowski S, Staatz CE, Clark J, Lawson R, Hennig S. Gentamicin pharmacokinetics and monitoring in pediatric febrile neutropenic patients. Ther Drug Monit. 2016;38(6):693–8. https://doi.org/10.1097/FTD.0000000000000341.
Standing JF, Morris J, Germovsek E, Lutsar I, Cortina-Borja M. Methods for optimising neonatal antimicrobial use: time- and concentration-dependent agents. PAGE 20 Abstr 2225 [www.page-meetingorg/?abstract=2225]; Athens; 2011.
Hennig S, Standing JF, Staatz CE, Thomson AH. Population pharmacokinetics of tobramycin in patients with and without cystic fibrosis. Clin Pharmacokinet. 2013;52(4):289–301. https://doi.org/10.1007/s40262-013-0036-y.
Viberg A, Cars O, Karlsson MO, Jonsson S. Estimation of cefuroxime dosage using pharmacodynamic targets, MIC distributions, and minimization of a risk function. J Clin Pharmacol. 2008;48(11):1270–81. https://doi.org/10.1177/0091270008320923.
Llanos-Paez CC, Staatz CE, Lawson R, Hennig S. A population pharmacokinetic model of gentamicin in pediatric oncology patients to facilitate personalized dosing. Antimicrob Agents Chemother. 2017;61(8):e00205–17. https://doi.org/10.1128/AAC.00205-17.
Zhang L, Beal SL, Sheinerz LB. Simultaneous vs. sequential analysis for population PK/PD data II: robustness of methods. J Pharmacokinet Pharmacodyn. 2003;30(6):405–16. https://doi.org/10.1023/B:JOPA.0000012999.36063.4e.
Zhang L, Beal SL, Sheiner LB. Simultaneous vs. sequential analysis for population PK/PD data I: best-case performance. J Pharmacokinet Pharmacodyn. 2003;30(6):387–404. https://doi.org/10.1023/B:JOPA.0000012998.04442.1f.
Schnermann J, Häberle DA, Davis JM, Thurau K. Tubuloglomerular feedback control of renal vascular resistance. Compr Physiol 2011:1675–705.
Pottel H, Mottaghy FM, Zaman Z, Martens F. On the relationship between glomerular filtration rate and serum creatinine in children. Pediatr Nephrol. 2010;25(5):927–34. https://doi.org/10.1007/s00467-009-1389-1.
Schwartz GJ, Munoz A, Schneider MF, Mak RH, Kaskel F, Warady BA, et al. New equations to estimate GFR in children with CKD. J Am Soc Nephrol. 2009;20(3):629–37. https://doi.org/10.1681/ASN.2008030287.
Du Bois D, Du Bois EF. A Formula to estimate the approximate surface area if height and weight be known. Nutrition. 1989;5(5):303–11.
Rhodin MM, Anderson BJ, Peters AM, Coulthard MG, Wilkins B, Cole M, et al. Human renal function maturation: a quantitative description using weight and postmenstrual age. Pediatr Nephrol. 2009;24(1):67–76. https://doi.org/10.1007/s00467-008-0997-5.
Ceriotti F, Boyd JC, Klein G, Henny J, Queralto J, Kairisto V, et al. Reference intervals for serum creatinine concentrations: assessment of available data for global application. Clin Chem. 2008;54(3):559–66. https://doi.org/10.1373/clinchem.2007.099648.
Giuliano RA, Verpooten GA, Verbist L, Wedeen RP, De Broe ME. In vivo uptake kinetics of aminoglycosides in the kidney cortex of rats. J Pharmacol Exp Ther. 1986;236(2):470–5.
The children's hospital at Westmead. Aminoglycoside Dosing and Monitoring 2015 [Available from: http://www.schn.health.nsw.gov.au/_policies/pdf/2011-8018.pdf.]
Antibiotic Expert Group. Therapeutic guidelines: antibiotic. Version 15. Melbourne: Therapeutic Guidelines Limited. 2014.
Anderson BJ, Holford NH. Tips and traps analyzing pediatric PK data. Paediatr Anaesth. 2011;21(3):222–37. https://doi.org/10.1111/j.1460-9592.2011.03536.x.
Mueller M, de la Pena A, Derendorf H. Issues in pharmacokinetics and pharmacodynamics of anti-infective agents: kill curves versus MIC. Antimicrob Agents Chemother. 2004;48(2):369–77. https://doi.org/10.1128/AAC.48.2.369-377.2004.
Craig WA, Andes D. Correlation of the magnitude of the AUC[24]/MIC for 6 fluoroquinolones against Streptococcus pneumoniae (SP) with survival and bactericidal activity in an animal model. Abstr Intersci Conf Antimicrob Agents Chemother; Toronto, 2000.
Prins JM, Buller HR, Kuijper EJ, Tange RA, Speelman P. Once versus thrice daily gentamicin in patients with serious infections. Lancet. 1993;341(8841):335–9. https://doi.org/10.1016/0140-6736(93)90137-6.
Llanos-Paez CC, Staatz C, Lawson R, Hennig S. Comparison of methods to estimate glomerular filtration rate in paediatric oncology patients. J Paediatr Child Health. 2017; https://doi.org/10.1111/jpc.13752.
Goldstein SL, Kirkendall E, Nguyen H, Schaffzin JK, Bucuvalas J, Bracke T, et al. Electronic health record identification of nephrotoxin exposure and associated acute kidney injury. Pediatrics. 2013;132(3):756–67.
Kent A, Turner MA, Sharland M, Heath PT. Aminoglycoside toxicity in neonates: something to worry about? Expert Rev Anti-Infect Ther. 2014;12(3):319–31. https://doi.org/10.1586/14787210.2014.878648.
Wicha SG, Kees MG, Solms A, Minichmayr IK, Kratzer A, Kloft C. TDMx: a novel web-based open-access support tool for optimising antimicrobial dosing regimens in clinical routine. Int J Antimicrob Agents. 2015;45(4):442–4. https://doi.org/10.1016/j.ijantimicag.2014.12.010.
Hennig S, Nyberg J, Fanta S, Backman JT, Hoppu K, Hooker AC, et al. Application of the optimal design approach to improve a pretransplant drug dose finding design for ciclosporin. J Clin Pharmacol. 2012;52(3):347–60. https://doi.org/10.1177/0091270010397731.
Retout S, Duffull S, Mentre F. Development and implementation of the population Fisher information matrix for the evaluation of population pharmacokinetic designs. Comput Methods Prog Biomed. 2001;65(2):141–51. https://doi.org/10.1016/S0169-2607(00)00117-6.
Merle Y, Mentre F. Optimal sampling times for Bayesian estimation of the pharmacokinetic parameters of nortriptyline during therapeutic drug monitoring. J Pharmacokinet Biopharm. 1999;27(1):85–101. https://doi.org/10.1023/A:1020634813296.
Acknowledgements
The authors would like to thank the Australian Centre of Pharmacometrics for the NONMEM® licences. C C Llanos-Paez acknowledged the BECAS-Chile CONICYT scholarship for supporting her PhD during the time this manuscript was written.
Author information
Authors and Affiliations
Contributions
CLLP analysed the data and wrote the manuscript. CS and SH provided a critical review of the manuscript and contributed to the writing of the manuscript.
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Llanos-Paez, C.C., Staatz, C. & Hennig, S. Balancing Antibacterial Efficacy and Reduction in Renal Function to Optimise Initial Gentamicin Dosing in Paediatric Oncology Patients. AAPS J 20, 14 (2018). https://doi.org/10.1208/s12248-017-0173-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12248-017-0173-6