Abstract
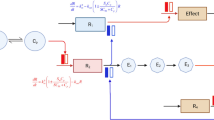
Quantitative systems pharmacology (QSP) models are increasingly used in drug development to provide a deep understanding of the mechanism of action of drugs and to identify appropriate disease targets. Such models are, however, not suitable for estimation purposes due to their high dimensionality. Based on any desired and specific input-output relationship, the system may be reduced to a model with fewer states and parameters. However, any simplification process will be a trade-off between model performance and complexity. In this study, we develop a weighted composite criterion which brings together the opposing indices of performance and dimensionality. The weighting factor can be determined by qualification of the simplified model based on a visual predictive check (VPC) using the precision of each parameter. The weighted criterion and model qualification techniques were illustrated with three examples: a simple compartmental pharmacokinetic model, a physiologically based pharmacokinetic (PBPK) example, and a semimechanistic model for bone mineral density. When considering the PBPK example, this automated search identified the same reduced model which had been detected in a previous report, as well as a simpler model which had not been previously identified. The simpler bone mineral density model provided an adequate description of the response even after 1 year from the initiation of treatment. The proposed criterion together with a VPC provides a natural way for model order reduction that can be fully automated and applied to multiscale models.









Similar content being viewed by others
References
van der Graaf PH, Benson N. Systems pharmacology: bridging systems biology and pharmacokinetics-pharmacodynamics (PKPD) in drug discovery and development. Pharm Res. 2011;28(7):1460–4. https://doi.org/10.1007/s11095-011-0467-9.
Benson N, Matsuura T, Smirnov S, Demin O, Jones HM, Dua P, et al. Systems pharmacology of the nerve growth factor pathway: use of a systems biology model for the identification of key drug targets using sensitivity analysis and the integration of physiology and pharmacology. Interface Focus. 2013;3(2):20120071. https://doi.org/10.1098/rsfs.2012.0071.
Jamei M. Recent advances in development and application of physiologically-based pharmacokinetic (PBPK) models: a transition from academic curiosity to regulatory acceptance. Curr Pharmacol Rep. 2016;2(3):161–9. https://doi.org/10.1007/s40495-016-0059-9.
Peterson MC, Riggs MMFDA. Advisory meeting clinical pharmacology review utilizes a quantitative systems pharmacology (QSP) model: a watershed moment? CPT Pharmacometrics Syst Pharmacol. 2015;4(3):e00020. https://doi.org/10.1002/psp4.20.
Tsamandouras N, Rostami-Hodjegan A, Aarons L. Combining the ‘bottom up’ and ‘top down’ approaches in pharmacokinetic modelling: fitting PBPK models to observed clinical data. Br J Clin Pharmacol. 2015;79(1):48–55. https://doi.org/10.1111/bcp.12234.
Ribba B, Grimm HP, Agoram B, Davies MR, Gadkar K, Niederer S, et al. Methodologies for quantitative systems pharmacology (QSP) models: design and estimation. CPT Pharmacometrics Syst Pharmacol. 2017;6(8):496–8. https://doi.org/10.1002/psp4.12206.
Pilari S, Huisinga W. Lumping of physiologically-based pharmacokinetic models and a mechanistic derivation of classical compartmental models. J Pharmacokinet Pharmacodyn. 2010;37(4):365–405. https://doi.org/10.1007/s10928-010-9165-1.
Dokoumetzidis A, Aarons L. Proper lumping in systems biology models. IET Syst Biol. 2009;3(1):40–51. https://doi.org/10.1049/iet-syb:20070055.
Gulati A, Isbister GK, Duffull SB. Scale reduction of a systems coagulation model with an application to modeling pharmacokinetic-pharmacodynamic data. CPT Pharmacometrics Syst Pharmacol. 2014;3(1):e90. https://doi.org/10.1038/psp.2013.67.
Peters E, Heuberger JAAC, Tiessen R, van Elsas A, Masereeuw R, Arend J, et al. Pharmacokinetic modeling and dose selection in a randomized, double-blind, placebo-controlled trial of a human recombinant alkaline phosphatase in healthy volunteers. Clin Pharmacokinet. 2016;55(10):1227–37. https://doi.org/10.1007/s40262-016-0399-y.
Wendling T, Dumitras S, Ogungbenro K, Aarons L. Application of a Bayesian approach to physiological modelling of mavoglurant population pharmacokinetics. J Pharmacokinet Pharmacodyn. 2015;42(6):639–57. https://doi.org/10.1007/s10928-015-9430-4.
van Schaick E, Zheng J, Perez Ruixo JJ, Gieschke R, Jacqmin PA. Semi-mechanistic model of bone mineral density and bone turnover based on a circular model of bone remodeling. J Pharmacokinet Pharmacodyn. 2015;42(4):315–32. https://doi.org/10.1007/s10928-015-9423-3.
Bauer RJ. NONMEM users guide introduction to NONMEM 7.3.0. Hanover: ICON Development Solutions; 2013.
Goffe WL, Ferrier GD, Rogers J. Global optimization of statistical functions with simulated annealing. J Econom. 1994;60(1–2):65–99. https://doi.org/10.1016/0304-4076(94)90038-8.
Pan S, Duffull S. Automated proper lumping for simplification of systems models. Hersonissos, Crete: Population Approach Group Europe PAGE; 2015.
Shivva V, Korell J, Tucker IG, Duffull SB. An approach for identifiability of population pharmacokinetic–pharmacodynamics models. CPT Pharmacomet Syst Pharmacol. 2013;2(6):e49. https://doi.org/10.1038/psp.2013.25.
Wendling T, Tsamandouras N, Dumitras S, Pigeolet E, Ogungbenro K, Aarons L. Reduction of a whole-body physiologically based pharmacokinetic model to stabilise the Bayesian analysis of clinical data. AAPS J. 2016;18(1):196–209. https://doi.org/10.1208/s12248-015-9840-7.
Brown RP, Delp MD, Lindstedt SL, Rhomberg LR, Beliles RP. Physiological parameter values for physiologically based pharmacokinetic models. Toxicol Ind Health. 1997;13(4):407–84. https://doi.org/10.1177/074823379701300401.
Snowden TJ, van der Graaf PH, Tindall MJ. A combined model reduction algorithm for controlled biochemical systems. BMC Syst Biol. 2017;11(1):17. https://doi.org/10.1186/s12918-017-0397-1.
Wade JR, Edholm M, Salmonson TA. Guide for reporting the results of population pharmacokinetic analyses: a Swedish perspective. AAPS J. 2005;7(2):45. https://doi.org/10.1208/aapsj070245.
Byon W, Smith MK, Chan P, Tortorici MA, Riley S, Dai H, et al. Establishing best practices and guidance in population modeling: an experience with an internal population pharmacokinetic analysis guidance. CPT Pharmacometrics Syst Pharmacol. 2013;2(7):e51. https://doi.org/10.1038/psp.2013.26.
Snowden TJ, van der Graaf PH, Tindall MJ. Methods of model reduction for large-scale biological systems: a survey of current methods and trends. Bull Math Biol. 2017;79(7):1449–86. https://doi.org/10.1007/s11538-017-0277-2.
Wajima T, Isbister GK, Duffull SBA. Comprehensive model for the humoral coagulation network in humans. Clin Pharmacol Ther. 2009;86(3):290–8. https://doi.org/10.1038/clpt.2009.87.
Peterson MC, Riggs MMA. Physiologically based mathematical model of integrated calcium homeostasis and bone remodeling. Bone. 2010;46(1):49–63. https://doi.org/10.1016/j.bone.2009.08.053.
Demin O, Karelina T, Svetlichniy D, Metelkin E, Speshilov G, Demin O Jr, et al. Systems pharmacology models can be used to understand complex pharmacokinetic-pharmacodynamic behavior: an example using 5-lipoxygenase inhibitors. CPT Pharmacometrics Syst Pharmacol. 2013;2(9):e74. https://doi.org/10.1038/psp.2013.49.
Hasegawa C, Duffull SB. Exploring inductive linearization for pharmacokinetic-pharmacodynamic systems of nonlinear ordinary differential equations. J Pharmacokinet Pharmacodyn. 2017. https://doi.org/10.1007/s10928-017-9527-z.
Zheng J, van Schaick E, LS W, Jacqmin P, Perez Ruixo JJ. Using early biomarker data to predict long-term bone mineral density: application of semi-mechanistic bone cycle model on denosumab data. J Pharmacokinet Pharmacodyn. 2015;42(4):333–47. https://doi.org/10.1007/s10928-015-9422-4.
Hazelwood SJ, Bruce Martin R, Rashid MM, Rodrigo JJA. Mechanistic model for internal bone remodeling exhibits different dynamic responses in disuse and overload. J Biomech. 2001;34(3):299–308. https://doi.org/10.1016/S0021-9290(00)00221-9.
Eriksen EF, Hodgson SF, Eastell R, Cedel SL, O'Fallon WM, Riggs BL. Cancellous bone remodeling in type I (postmenopausal) osteoporosis: quantitative assessment of rates of formation, resorption, and bone loss at tissue and cellular levels. J Bone Miner Res. 1990;5(4):311–9. https://doi.org/10.1002/jbmr.5650050402.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Chihiro Hasegawa is an employee of Ono Pharmaceutical Co., Ltd. and a visiting researcher at the University of Otago.
Electronic Supplementary Material
ESM 1
(DOC 151 kb)
Rights and permissions
About this article
Cite this article
Hasegawa, C., Duffull, S.B. Selection and Qualification of Simplified QSP Models When Using Model Order Reduction Techniques. AAPS J 20, 2 (2018). https://doi.org/10.1208/s12248-017-0170-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12248-017-0170-9