Abstract
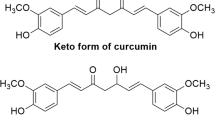
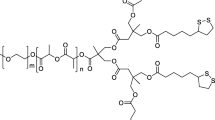
Curcumin, a phenolic compound, possesses many pharmacological activities and is under clinical evaluation to treat different diseases. However, conflicting data about its stability have been reported. In this study, the kinetic degradation of curcumin from a natural curcuminoid mixture under various conditions (pH, temperature, and dielectric constant of the medium) was investigated. Moreover, the degradation of pure curcumin at some selected conditions was also determined. To fully solubilize curcumin and to prevent precipitation of curcumin that occurs when low concentrations of co–solvent are present, a 50:50 (v/v) aqueous buffer/methanol mixture was used as standard medium to study its degradation kinetics. The results showed that degradation of curcumin both as pure compound and present in the curcuminoid mixture followed first order kinetic reaction. It was further shown that an increasing pH, temperature, and dielectric constant of the medium resulted in an increase in the degradation rate. Curcumin showed rapid degradation due to autoxidation in aqueous buffer pH = 8.0 with a rate constant of 280 × 10-3 h-1, corresponding with a half–life (t1/2) of 2.5 h. Dioxygenated bicyclopentadione was identified as the final degradation product. Importantly, curcumin loaded as curcuminoid mixture in ω–methoxy poly (ethylene glycol)–b–(N–(2–benzoyloxypropyl) methacrylamide) (mPEG–HPMA–Bz) polymeric micelles and in Triton X–100 micelles was about 300–500 times more stable than in aqueous buffer. Therefore, loading of curcumin into polymeric micelles is a promising approach to stabilize this compound and develop formulations suitable for further pharmaceutical and clinical studies.








Similar content being viewed by others
REFERENCES
Sharma R, Gescher A, Steward W. Curcumin: the story so far. Eur J Cancer. 2005;41(13):1955–68.
Vogel H, Pelletier J. Curcumin—biological and medicinal properties. J Pharmacol. 1815;2:50–0.
Aggarwal BB, Harikumar KB. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int J Biochem Cell Biol. 2009;41(1):40–59.
Ravindran J, Prasad S, Aggarwal B. Curcumin and cancer cells: how many ways can curry kill tumor cells selectively? AAPS J. 2009;11(3):495–510.
Gupta S, Patchva S, Aggarwal B. Therapeutic roles of curcumin: lessons learned from clinical trials. AAPS J. 2013;15(1):195–218.
Heger M, van Golen RF, Broekgaarden M, Michel MC. The molecular basis for the pharmacokinetics and pharmacodynamics of curcumin and its metabolites in relation to cancer. Pharmacol Rev. 2014;66(1):222–307.
Tønnesen HH, Másson M, Loftsson T. Studies of curcumin and curcuminoids. XXVII. Cyclodextrincomplexation: solubility, chemical and photochemical stability. Int J Pharm. 2002;244(1):127–35.
Schneider C, Gordon ON, Edwards RL, Luis PB. Degradation of curcumin: from mechanism to biological implications. J Agric Food Chem. 2015;63(35):7606–14.
Lao CD, Ruffin MT, Normolle D, Heath DD, Murray SI, Bailey JM, et al. Dose escalation of a curcuminoid formulation. BMC Complement Altern Med. 2006;6(1):6–10.
Garcea G, Berry DP, Jones DJ, Singh R, Dennison AR, Farmer PB, et al. Consumption of the putative chemopreventive agent curcumin by cancer patients: assessment of curcumin levels in the colorectum and their pharmacodynamic consequences. Cancer Epidemiol Biomarkers Prev. 2005;14(1):120–5.
Okada K, Wangpoengtrakul C, Tanaka T, Toyokuni S, Uchida K, Osawa T. Curcumin and especially tetrahydrocurcumin ameliorate oxidative stress-induced renal injury in mice. J Nutr. 2001;131(8):2090–5.
Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB. Bioavailability of curcumin: problems and promises. Mol Pharm. 2007;4(6):807–18.
Wang YJ, Pan MH, Cheng AL, Lin LI, Ho YS, Hsieh CY, et al. Stability of curcumin in buffer solutions and characterization of its degradation products. J Pharm Biomed Anal. 1997;15(12):1867–76.
Suresh D, Gurudutt KN, Srinivasan K. Degradation of bioactive spice compound: curcumin during domestic cooking. Eur Food Res Technol. 2009;228(5):807–12.
Griesser M, Pistis V, Suzuki T, Tejera N, Pratt DA, Schneider C. Autoxidative and cyclooxygenase-2 catalyzed transformation of the dietary chemopreventive agent curcumin. J Biol Chem. 2011;286(2):1114–24.
Gordon O. Oxidative transformation of curcumin: products and reaction mechanisms. Nashville (NSH): Vanderbilt University; 2014. Dissertation.
Gordon ON, Luis PB, Sintim HO, Schneider C. Unraveling curcumin degradation: autoxidation proceeds through spiroepoxide and vinylether intermediates en route to the main bicyclopentadione. J Biol Chem. 2015;290(8):4817–28.
Yallapu MM, Jaggi M, Chauhan SC. Curcumin nanoformulations: a future nanomedicine for cancer. Drug Discov Today. 2012;17(1–2):71–80.
Naksuriya O, Okonogi S, Schiffelers RM, Hennink WE. Curcumin nanoformulations: a review of pharmaceutical properties and preclinical studies and clinical data related to cancer treatment. Biomaterials. 2014;35(10):3365–83.
Lee WH, Loo CY, Young PM, Traini D, Mason RS, Rohanizadeh R. Recent advances in curcumin nanoformulation for cancer therapy. Expert Opin Drug Deliv. 2014;11(8):1183–201.
Thangavel S, Yoshitomi T, Sakharkar MK, Nagasaki Y. Redox nanoparticles inhibit curcumin oxidative degradation and enhance its therapeutic effect on prostate cancer. J Control Release. 2015;209:110–9.
Dey S, Sreenivasan K. Conjugation of curcumin onto alginate enhances aqueous solubility and stability of curcumin. Carbohydr Polym. 2014;99:499–507.
Mohanty C, Sahoo SK. The in vitro stability and in vivo pharmacokinetics of curcumin prepared as an aqueous nanoparticulate formulation. Biomaterials. 2010;31(25):6597–611.
Sandur SK, Pandey MK, Sung B, Ahn KS, Murakami A, Sethi G, et al. Curcumin, demethoxycurcumin, bisdemethoxycurcumin, tetrahydrocurcumin and turmerones differentially regulate anti-inflammatory and anti-proliferative responses through a ROS-independent mechanism. Carcinogenesis. 2007;28(8):1765–73.
Shi Y, van der Meel R, Theek B, Oude Blenke E, Pieters EH, Fens MH, et al. Complete regression of xenograft tumors upon targeted delivery of paclitaxel via π–π stacking stabilized polymeric micelles. ACS Nano. 2015;9(4):3740–52.
Naksuriya O, Shi Y, van Nostrum CF, Anuchapreeda S, Hennink WE, Okonogi S. HPMA-based polymeric micelles for curcumin solubilization and inhibition of cancer cell growth. Eur J Pharm Biopharm. 2015;94:501–12.
Ghasemi J, Niazi A, Kubista M, Elbergali A. Spectrophotometric determination of acidity constants of 4-(2-pyridylazo) resorcinol in binary methanol–water mixtures. Anal Chim Acta. 2002;455(2):335–42.
De Jong S, Arias ER, Rijkers D, van Nostrum C, Kettenes–van den Bosch J, Hennink W. New insights into the hydrolytic degradation of poly (lactic acid): participation of the alcohol terminus. Polymer. 2001;42(7):2795–802.
Tønnesen HH, Karlsen J, van Henegouwen GB. Studies on curcumin and curcuminoids VIII. Photochemical stability of curcumin. Z Lebensm Unters Forsch. 1986;183(2):116–22.
Kurien BT, Singh A, Matsumoto H, Scofield RH. Improving the solubility and pharmacological efficacy of curcumin by heat treatment. Assay Drug Dev Technol. 2007;5(4):567–76.
Bernabé–Pineda M, Ramírez–Silva MT, Romero–Romo M, González–Vergara E, Rojas–Hernández A. Determination of acidity constants of curcumin in aqueous solution and apparent rate constant of its decomposition. Spectrochim Acta A. 2004;60(5):1091–7.
Leung MH, Colangelo H, Kee TW. Encapsulation of curcumin in cationic micelles suppresses alkaline hydrolysis. Langmuir. 2008;24(11):5672–5.
Zhu QY, Zhang A, Tsang D, Huang Y, Chen ZY. Stability of green tea catechins. J Agric Food Chem. 1997;45(12):4624–8.
Yoshioka H, Sugiura K, Kawahara R, Fujita T, Makino M, Kamiya M, et al. Formation of radicals and chemiluminescence during the autoxidation of tea catechins. Agr Biol Chem Tokyo. 1991;55(11):2717–23.
Amorati R, Pedulli GF, Cabrini L, Zambonin L, Landi L. Solvent and pH effects on the antioxidant activity of caffeic and other phenolic acids. J Agric Food Chem. 2006;54(8):2932–7.
Ozgen M, Reese RN, Tulio AZ, Scheerens JC, Miller AR. Modified 2,2-Azino-bis-3-ethylbenzothiazoline-6-sulfonic acid (ABTS) method to measure antioxidant capacity of selected small fruits and comparison to ferric reducing antioxidant power (FRAP) and 2,2‘-Diphenyl-1-picrylhydrazyl (DPPH) methods. J Agric Food Chem. 2006;54(4):1151–7.
Dawidowicz A, Olszowy M. Antioxidant properties of BHT estimated by ABTS assay in systems differing in pH or metal ion or water concentration. Eur Food Res Technol. 2011;232(5):837–42.
Gordon ON, Luis PB, Ashley RE, Osheroff N, Schneider C. Oxidative transformation of demethoxy- and bisdemethoxycurcumin: products, mechanism of formation, and poisoning of human topoisomerase IIα. Chem Res Toxicol. 2015;28(5):989–96.
Leung MH, Kee TW. Effective stabilization of curcumin by association to plasma proteins: human serum albumin and fibrinogen. Langmuir. 2009;25(10):5773–7.
Wei X, Senanayake TH, Bohling A, Vinogradov SV. Targeted nanogel conjugate for improved stability and cellular permeability of curcumin: synthesis, pharmacokinetics, and tumor growth inhibition. Mol Pharm. 2014;11(9):3112–22.
D’Souza A, Jain P, Galdhar CN, Samad A, Degani M, Devarajan P. Comparative in silico–in vivo evaluation of ASGP-R ligands for hepatic targeting of curcumin Gantrez nanoparticles. AAPS J. 2013;15(3):696–706.
Cheng KK, Yeung CF, Ho SW, Chow SF, Chow AH, Baum L. Highly stabilized curcumin nanoparticles tested in an in vitro blood–brain barrier model and in Alzheimer’s disease Tg2576 mice. AAPS J. 2012;15(2):324–36.
Pan Y, Tikekar RV, Wang MS, Avena–Bustillos RJ, Nitin N. Effect of barrier properties of zein colloidal particles and oil-in-water emulsions on oxidative stability of encapsulated bioactive compounds. Food Hydrocoll. 2015;43:82–90.
Chen Y, Zhang X, Lu J, Huang Y, Li J, Li S. Targeted delivery of curcumin to tumors via PEG-derivatized FTS-based micellar system. AAPS J. 2014;16(3):600–8.
ACKNOWLEDGMENTS
The authors are grateful for the support received from the Thailand Research Fund (TRF) through the Royal Golden Jubilee PhD Program (RGJ) Grant No. 5. G. CM/52/D. 2. IN. We thank the Department of Pharmaceutics, Utrecht Institute for Pharmaceutical Sciences (UIPS), Utrecht University and Faculty of Pharmacy, Chiang Mai University for their support.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 5.23 mb)
Rights and permissions
About this article
Cite this article
Naksuriya, O., van Steenbergen, M.J., Torano, J.S. et al. A Kinetic Degradation Study of Curcumin in Its Free Form and Loaded in Polymeric Micelles. AAPS J 18, 777–787 (2016). https://doi.org/10.1208/s12248-015-9863-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-015-9863-0