Abstract
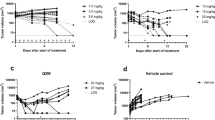
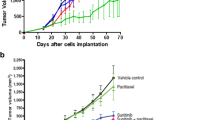
Timing of the anti-angiogenic agent with respect to the chemotherapeutic agent may be crucial in determining the success of combination therapy in cancer. We investigated the effects of sequential therapy with the potent VEGF inhibitor, aflibercept, and doxorubicin (DOX) in preclinical acute myeloid leukemia (AML) models. Mice were engrafted with human HL-60 and HEL-luciferase leukemia cells via S.C. and/or I.V. injection and treated with two to three doses of aflibercept (5–25 mg/kg) up to 3–7 days prior to doxorubicin (30 mg/kg) administration. Leukemia growth was determined by local tumor measurements (days 0–16) and systemic bioluminescent imaging (days 0–28) in animals receiving DOX (3 mg/kg) with or without aflibercept. A PK/PD model was developed to characterize how prior administration of aflibercept altered intratumoral DOX uptake. DOX concentration–time profiles were described using a four-compartment PK model with linear elimination. We determined that intratumoral DOX concentrations were 6-fold higher in the aflibercept plus DOX treatment group versus DOX alone in association with increased drug uptake rates (from 0.125 to 0.471 ml/h/kg) into tumor without affecting drug efflux. PD modeling demonstrated that the observed growth retardation was mainly due to the combination of DOX plus TRAP group; 0.00794 vs. 0.0043 h−1. This PK/PD modeling approach in leukemia enabled us to predict the effects of dosing frequency and sequence for the combination of anti-VEGF and cytotoxic agents on AML growth in both xenograft and marrow, and may be useful in the design of future rational combinatorial dosing regimens in hematological malignancies.





Similar content being viewed by others
References
Ma J, Waxman DJ. Combination of antiangiogenesis with chemotherapy for more effective cancer treatment. Mol Cancer Ther. 2008;7(12):3670–84. Epub 2008/12/17.
Saltz LB, Clarke S, Diaz-Rubio E, Scheithauer W, Figer A, Wong R, et al. Bevacizumab in combination with oxaliplatin-based chemotherapy as first-line therapy in metastatic colorectal cancer: a randomized phase III study. J Clin Oncol. 2008;26(12):2013–9. Epub 2008/04/19.
Gerber HP, Ferrara N. Pharmacology and pharmacodynamics of bevacizumab as monotherapy or in combination with cytotoxic therapy in preclinical studies. Cancer Res. 2005;65(3):671–80. Epub 2005/02/12.
Tong RT, Boucher Y, Kozin SV, Winkler F, Hicklin DJ, Jain RK. Vascular normalization by vascular endothelial growth factor receptor 2 blockade induces a pressure gradient across the vasculature and improves drug penetration in tumors. Cancer Res. 2004;64(11):3731–6.
Van der Veldt AA, Lubberink M, Bahce I, Walraven M, de Boer MP, Greuter HN, et al. Rapid decrease in delivery of chemotherapy to tumors after anti-VEGF therapy: implications for scheduling of anti-angiogenic drugs. Cancer Cell. 2012;21(1):82–91. Epub 2012/01/24.
Pastuskovas CV, Mundo EE, Williams SP, Nayak TK, Ho J, Ulufatu S, et al. Effects of anti-VEGF on pharmacokinetics, biodistribution, and tumor penetration of trastuzumab in a preclinical breast cancer model. Mol Cancer Ther. 2012;11(3):752–62. Epub 2012/01/10.
Aguayo A, Kantarjian H, Manshouri T, Gidel C, Estey E, Thomas D, et al. Angiogenesis in acute and chronic leukemias and myelodysplastic syndromes. Blood. 2000;96(6):2240–5. Epub 2000/09/09.
Wellbrock J, Fiedler W. Clinical experience with antiangiogenic therapy in leukemia. Curr Cancer Drug Targets. 2011;11(9):1053–68. Epub 2011/10/18.
Lal D, Park JA, Demock K, Marinaro J, Perez AM, Lin MH, et al. Aflibercept exerts antivascular effects and enhances levels of anthracycline chemotherapy in vivo in human acute myeloid leukemia models. Mol Cancer Ther. 2010;9(10):2737–51. Epub 2010/10/07.
Holash J, Davis S, Papadopoulos N, Croll SD, Ho L, Russell M, et al. VEGF-Trap: a VEGF blocker with potent antitumor effects. Proc Natl Acad Sci U S A. 2002;99(17):11393–8. Epub 2002/08/15.
Thai HT, Veyrat-Follet C, Vivier N, Dubruc C, Sanderink G, Mentre F, et al. A mechanism-based model for the population pharmacokinetics of free and bound aflibercept in healthy subjects. Br J Clin Pharmacol. 2011;72(3):402–14. Epub 2011/05/18.
Coleman RL, Duska LR, Ramirez PT, Heymach JV, Kamat AA, Modesitt SC, et al. Phase 1–2 study of docetaxel plus aflibercept in patients with recurrent ovarian, primary peritoneal, or fallopian tube cancer. Lancet Oncol. 2011;12(12):1109–17. Epub 2011/10/14.
Leighl NB, Raez LE, Besse B, Rosen PJ, Barlesi F, Massarelli E, et al. A multicenter, phase 2 study of vascular endothelial growth factor trap (Aflibercept) in platinum- and erlotinib-resistant adenocarcinoma of the lung. J Thorac Oncol. 2010;5(7):1054–9. Epub 2010/07/02.
Twardowski P, Stadler WM, Frankel P, Lara PN, Ruel C, Chatta G, et al. Phase II study of Aflibercept (VEGF-Trap) in patients with recurrent or metastatic urothelial cancer, a California Cancer Consortium Trial. Urology. 2010;76(4):923–6. Epub 2010/07/22.
de Groot JF, Lamborn KR, Chang SM, Gilbert MR, Cloughesy TF, Aldape K, et al. Phase II study of aflibercept in recurrent malignant glioma: a North American Brain Tumor Consortium study. J Clin Oncol. 2011;29(19):2689–95. Epub 2011/05/25.
Tarhini AA, Frankel P, Margolin KA, Christensen S, Ruel C, Shipe-Spotloe J, et al. Aflibercept (VEGF Trap) in inoperable stage III or stage IV melanoma of cutaneous or uveal origin. Clin Cancer Res. 2011;17(20):6574–81. doi:10.1158/1078-0432.CCR-11-1463.
Andersen A, Warren DJ, Slordal L. A sensitive and simple high-performance liquid chromatographic method for the determination of doxorubicin and its metabolites in plasma. Ther Drug Monit. 1993;15(5):455–61. Epub 1993/10/01.
Efferth T, Konkimalla VB, Wang YF, Sauerbrey A, Meinhardt S, Zintl F, et al. Prediction of broad spectrum resistance of tumors towards anticancer drugs. Clin Cancer Res. 2008;14(8):2405–12. Epub 2008/04/17.
Primeau AJ, Rendon A, Hedley D, Lilge L, Tannock IF. The distribution of the anticancer drug doxorubicin in relation to blood vessels in solid tumors. Clin Cancer Res. 2005;11(24 Pt 1):8782–8.
Lee EM, Bachmann PS, Lock RB. Xenograft models for the preclinical evaluation of new therapies in acute leukemia. Leuk Lymphoma. 2007;48(4):659–68. Epub 2007/04/25.
Baxter LT, Zhu H, Mackensen DG, Jain RK. Physiologically based pharmacokinetic model for specific and nonspecific monoclonal antibodies and fragments in normal tissues and human tumor xenografts in nude mice. Cancer Res. 1994;54(6):1517–28. Epub 1994/03/15.
Brown RP, Delp MD, Lindstedt SL, Rhomberg LR, Beliles RP. Physiological parameter values for physiologically based pharmacokinetic models. Toxicol Ind Heal. 1997;13(4):407–84. Epub 1997/07/01.
Davies B, Morris T. Physiological parameters in laboratory animals and humans. Pharm Res. 1993;10(7):1093–5. Epub 1993/07/01.
Gjedde SB, Gjedde A. Organ blood flow rates and cardiac output of the BALB/c mouse. Comp Biochem Physiol Part A: Physiology. 1980;67(4):5.
D’Argenio DZ, A. Schumitzky and X. Wang. ADAPT 5 User’s Guide: Pharmacokinetic/Pharmacodynamic Systems Analysis Software. Biomed Simul Resour. 2009.
Akaike H. A Bayesian extension of the minimum AIC procedure of autoregressive model fitting. Biometrika. 1979;66:237–43.
Simeoni M, Magni P, Cammia C, De Nicolao G, Croci V, Pesenti E, et al. Predictive pharmacokinetic-pharmacodynamic modeling of tumor growth kinetics in xenograft models after administration of anticancer agents. Cancer Res. 2004;64(3):1094–101. Epub 2004/02/12.
Jain RK. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science. 2005;307(5706):58–62. Epub 2005/01/08.
Goteti K, Garner CE, Utley L, Dai J, Ashwell S, Moustakas DT, et al. Preclinical pharmacokinetic/pharmacodynamic models to predict synergistic effects of co-administered anti-cancer agents. Cancer Chemother Pharmacol. 2010;66(2):245–54. Epub 2009/10/21.
Pruijn FB, van Daalen M, Holford NH, Wilson WR. Mechanisms of enhancement of the antitumour activity of melphalan by the tumour-blood-flow inhibitor 5,6-dimethylxanthenone-4-acetic acid. Cancer Chemother Pharmacol. 1997;39(6):541–6.
Troiani T, Serkova NJ, Gustafson DL, Henthorn TK, Lockerbie O, Merz A, et al. Investigation of two dosing schedules of vandetanib (ZD6474), an inhibitor of vascular endothelial growth factor receptor and epidermal growth factor receptor signaling, in combination with irinotecan in a human colon cancer xenograft model. Clin Cancer Res. 2007;13(21):6450–8. Epub 2007/11/03.
Ma J, Chen CS, Blute T, Waxman DJ. Antiangiogenesis enhances intratumoral drug retention. Cancer Res. 2011;71(7):2675–85. Epub 2011/03/31.
Urva SR, Shin BS, Yang VC, Balthasar JP. Sensitive high performance liquid chromatographic assay for assessment of doxorubicin pharmacokinetics in mouse plasma and tissues. J Chromatogr B, Anal Technol Biomed Life Sci. 2009;877(8–9):837–41. Epub 2009/02/28.
Webster LK, Cosson EJ, Stokes KH, Millward MJ. Effect of the paclitaxel vehicle, Cremophor EL, on the pharmacokinetics of doxorubicin and doxorubicinol in mice. Br J Cancer. 1996;73(4):522–4. Epub 1996/02/01.
Sim H, Bibee K, Wickline S, Sept D. Pharmacokinetic modeling of tumor bioluminescence implicates efflux, and not influx, as the bigger hurdle in cancer drug therapy. Cancer Res. 2011;71(3):686–92. Epub 2010/12/03.
Ayala F, Dewar R, Kieran M, Kalluri R. Contribution of bone microenvironment to leukemogenesis and leukemia progression. Leukemia. 2009;23(12):2233–41.
Schaefer C, Krause M, Fuhrhop I, Schroeder M, Algenstaedt P, Fiedler W, et al. Time-course-dependent microvascular alterations in a model of myeloid leukemia in vivo. Leukemia. 2008;22(1):59–65.
Iversen PO, Thing-Mortensen B, Nicolaysen G, Benestad HB. Decreased blood flow to rat bone marrow, bone, spleen, and liver in acute leukemia. Leuk Res. 1993;17(8):663–8. Epub 1993/08/01.
Nooter K, Sonneveld P, Martens A. Differences in the pharmacokinetics of daunomycin in normal and leukemic rats. Cancer Res. 1985;45(9):4020–5. Epub 1985/09/01.
Nooter K, de Vries A, Martens AC, Hagenbeek A. Effect of cyclophosphamide pretreatment on daunorubicin in rat acute leukaemia model. Eur J Cancer. 1990;26(6):729–32. Epub 1990/01/01.
Lowenberg B, Ossenkoppele GJ, van Putten W, Schouten HC, Graux C, Ferrant A, et al. High-dose daunorubicin in older patients with acute myeloid leukemia. N Engl J Med. 2009;361(13):1235–48. Epub 2009/09/25.
Fernandez HF, Sun Z, Yao X, Litzow MR, Luger SM, Paietta EM, et al. Anthracycline dose intensification in acute myeloid leukemia. N Engl J Med. 2009;361(13):1249–59.
Feldman EJ, Lancet JE, Kolitz JE, Ritchie EK, Roboz GJ, List AF, et al. First-in-man study of CPX-351: a liposomal carrier containing cytarabine and daunorubicin in a fixed 5:1 molar ratio for the treatment of relapsed and refractory acute myeloid leukemia. J Clin Oncol. 2011;29(8):979–85. Epub 2011/02/02.
Gourdeau H, Genne P, Kadhim S, Bibeau L, Duchamp O, Ouellet F, et al. Antitumor activity of troxacitabine (Troxatyl) against anthracycline-resistant human xenografts. Cancer Chemother Pharmacol. 2002;50(6):490–6. Epub 2002/11/27.
Lobo ED, Balthasar JP. Pharmacodynamic modeling of chemotherapeutic effects: application of a transit compartment model to characterize methotrexate effects in vitro. AAPS PharmSci. 2002;4(4):E42. Epub 2003/03/21.
Yang J, Mager DE, Straubinger RM. Comparison of two pharmacodynamic transduction models for the analysis of tumor therapeutic responses in model systems. AAPS J. 2010;12(1):1–10. Epub 2009/11/11.
Fiedler W, Mesters R, Heuser M, Ehninger G, Berdel WE, Zirrgiebel U, et al. An open-label, phase I study of cediranib (RECENTIN) in patients with acute myeloid leukemia. Leuk Res. 2010;34(2):196–202. Epub 2009/08/14.
Giles FJ, Bellamy WT, Estrov Z, O’Brien SM, Verstovsek S, Ravandi F, et al. The anti-angiogenesis agent, AG-013736, has minimal activity in elderly patients with poor prognosis acute myeloid leukemia (AML) or myelodysplastic syndrome (MDS). Leuk Res. 2006;30(7):801–11. Epub 2005/12/08.
Fiedler W, Serve H, Dohner H, Schwittay M, Ottmann OG, O’Farrell AM, et al. A phase 1 study of SU11248 in the treatment of patients with refractory or resistant acute myeloid leukemia (AML) or not amenable to conventional therapy for the disease. Blood. 2005;105(3):986–93. Epub 2004/10/02.
Fiedler W, Mesters R, Tinnefeld H, Loges S, Staib P, Duhrsen U, et al. A phase 2 clinical study of SU5416 in patients with refractory acute myeloid leukemia. Blood. 2003;102(8):2763–7.
Karp JE, Gojo I, Pili R, Gocke CD, Greer J, Guo C, et al. Targeting vascular endothelial growth factor for relapsed and refractory adult acute myelogenous leukemias: therapy with sequential 1-beta-d-arabinofuranosylcytosine, mitoxantrone, and bevacizumab. Clin Cancer Res. 2004;10(11):3577–85.
Roboz GJ, Giles FJ, List AF, Cortes JE, Carlin R, Kowalski M, et al. Phase 1 study of PTK787/ZK 222584, a small molecule tyrosine kinase receptor inhibitor, for the treatment of acute myeloid leukemia and myelodysplastic syndrome. Leukemia. 2006;20(6):952–7.
Ortholan C, Durivault J, Hannoun-Levi JM, Guyot M, Bourcier C, Ambrosetti D, et al. Bevacizumab/docetaxel association is more efficient than docetaxel alone in reducing breast and prostate cancer cell growth: a new paradigm for understanding the therapeutic effect of combined treatment. Eur J Cancer. 2010;46(16):3022–36. Epub 2010/08/24.
Acknowledgments
We would like to acknowledge Ashleigh Lamson M. S. for technical support. This research was supported by an American Cancer Society Mentored Research Scientist Award MRSG-06-044-01-LIB (to E.S.W) and the Szefel Leukemia Research Fund (to E.S.W). Core institute resource support was provided by the NCI Cancer Center Support Grant (CA016156).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fetterly, G.J., Aras, U., Lal, D. et al. Development of a Preclinical PK/PD Model to Assess Antitumor Response of a Sequential Aflibercept and Doxorubicin-Dosing Strategy in Acute Myeloid Leukemia. AAPS J 15, 662–673 (2013). https://doi.org/10.1208/s12248-013-9480-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-013-9480-8