Abstract
Blastocyst implantation and neoplastic invasion have some common properties related to tissue invasion, mediated by various cytokines.
Aim
To compare the expression of IL-6, STAT-3 and telomerase in material of abortions in the first trimester of pregnancy, at term placentas and in choriocarcinomas.
Methods
Immunohistochemical reactions were performed on formalin fixed and included in paraffin samples from 3 groups: abortions, normal at term placentas and choriocarcinomas. Western Blot and Real-Time PCR assays were performed on fresh material from BeWo cell line and in primary culture cells of normal placenta.
Results
Immunohistochemical reactions: IL-6 expression was moderate in the first trimester abortion samples and high in at term placentas and choriocarcinomas. STAT-3 was strongly positive in all groups. Telomerase expression was absent in normal at term placentas but was increased in BeWo cells.
Conclusion
IL-6 and STAT-3 are present in the invasion process of the normal placental development and they are maintained during the malignant transformation to choriocarcinoma. The intense telomerase expression observed in BeWo cells was strongly associated with the malignant phenotype, confirming it as a good marker for cell transformation and tumor progression.
Similar content being viewed by others
Introduction
The establishment of pregnancy starts with blastocyst implantation in the endometrium and depends on the ability of trophoblastic cells to interact with the uterine wall, endometrial stroma and blood vessels. Additionally, the uterus undergoes stimulation by regulatory molecules (Knöfler and Pollheimer 2012), which play a functional role in preparing the endometrial mucosa for embryo implantation and trophoblastic adhesion, invasion and directional trafficking (Dimitriadis et al. 2010).
It is well-established that embryo implantation has many similarities with carcinogenesis because both trophoblastic and tumor cells are capable of tissue invasion. Although it occurs in trophoblastic cells under physiological conditions, changes in this process can result in neoplastic development linked to pregnancy (Fitzgerald et al. 2008). Many of the signaling factors that are operated or produced by trophoblastic cells can trigger the signaling pathway through Janus kinases (JAK), signal transducers and activators of transcription via JAK/STAT pathway that is involved in transcriptional regulation of proteases that are crucially important for invasive cell growth (Konjević 2009).
In normal conditions, the expression of STAT-3 is responsible for the induction of immunoregulatory signals, as well as for the modulation of specific genes in the processes of cell growth, differentiation and survival (Fitzgerald et al. 2010). However, during tumor invasion, its expression or activation become extremely high, due to signaling mediators that are activated via STAT-3, acting on cell growth and motility of tumor and pluripotent cells (Wen et al. 2014).
Like STAT-3, cytokine IL-6 is also another molecule involved in neoplastic process. According Chiesa et al. (Chiesa et al. 2015), in normal conditions, IL-6 is a multifunctional cytokine involved in regulating the immune response, the hematopoiesis, the acute phase of inflammation. IL-6 concentrations correlate with the extent of leukocyte infiltration in preterm placentas. In contrast, in neoplastic process, this cytokine has a role in adhesion of trophoblastic cells to extra-villous elements of the extracellular matrix (ECM), decreasing the expression of integrin and stimulating the production of metalloproteinase inhibitors (TIMP), thus allowing the tumor cell invasion process (Bischof 2001).
Another molecule also involved in the proliferation process is telomerase. Normal cells show limited life span telomerase activity, both in vitro and in vivo, and they have mostly negative expression. Differently, in neoplastic cells the maintenance or increase of telomerase seems to be necessary to prevent loss of telomere length and maintain the capacity for cell proliferation (Chen et al. 2002).
Considering the gestational trophoblastic diseases (GTD), the choriocarcinoma is the most invasive malignant condition, caused by abnormal proliferation of trophoblastic cells whose alterations occur during the trophoblastic differentiation, with the potential to form tumor mass and metastasis (Kishimoto 2005; Monchek and Wiedaseck 2012).
Thus, the aim of this study is to verify if there are differences in the expression of cytokine IL-6, STAT-3 transcription factor and in the expression of telomerase in the invasion of trophoblastic cells of normal placenta in relation to choriocarcinoma.
Materials and methods
Ethics committee
This study was approved by the Ethics Committee of the Faculty of Medical Sciences, Campinas State University (UNICAMP), on 26/06/2009, with CEP n°. 368/2009, C.A.A.E. 0282.0.146.000–09.
Constitution of samples
For the retrospective study from fixed material
Formalin fixed and paraffin embedded tissue blocks from the biopsy files of the Laboratory of Pathology, University of Campinas (UNICAMP), Brazil, from January 2000 to November 2008 were selected totaling 12 curettage material in first trimester abortions, 12 normal at term placentas and 12 choriocarcinomas, in optimal conditions for technical assays. Four μm thick sections were cut from paraffin blocks and the slides were stained with hematoxylin and eosin (H&E) and immunohistochemical reactions for analysis (Table 1).
For the prospective study from fresh material (culture cells)
BeWo cell
BeWo cell is an immortalized cell line established from a human choriocarcinoma. It was acquired from Cell Bank of Rio de Janeiro (Scientific Technical Association Paul Ehrlich - APABCAM). BeWo cells were replicated in culture medium Ham’s F12 with 5% FBS (fetal bovine serum), frozen in liquid nitrogen and stored for further processing. After reaching the semi-confluence status, adherent cells were removed from the substrate using a cell scraper and cryo-preserved in liquid nitrogen for later use in Western Blotting and Real-Time PCR reactions.
Primary culture from normal at term human placenta
Normal human placenta was collected at delivery room, cut into small pieces and washed in saline solution to remove as much blood cells as possible. After rinsing several times, the small fragments underwent enzymatic digestion with TVA solution (trypsin-versine association) for 2 h. Subsequently, the material was collected in tubes and submitted to centrifugation at 800 g with the gathering of the sediment cells. Completing the volume to obtain a cellular suspension of 2 × 106 cells/ml, the cells were placed in several culture flasks with Ham F-12 medium enriched with 20% of bovine fetal serum. At the semi-confluence stage, the cells from the primary culture were removed from the substrate using a cell scraper and cryo-preserved in liquid nitrogen for later use in Western Blotting and Real-Time PCR reactions.
Techniques
Immunohistochemistry (IHC)
Immunohistochemical assays for IL-6 and STAT-3 were performed according to optimized protocols. Sections were deparaffinized and endogenous peroxidase activity was blocked in 3% hydrogen peroxide. Heat antigen retrieval was used with 10 mM sodium citrate buffer, pH 6.0, at 95 °C in a vapor cooker for 30 min. Slides were washed in PBS and after that incubated with the primary antibodies (Table 1) under humidity, at 4 °C overnight. Secondary antibody (anti-mouse, Santa Cruz Biotechonology, CA, USA) was put on slides at 37 °C, for 30 min. Staining was visualized with DAB (diaminobenzine tetrahydrochloride—SIGMA), DMSO, and diluted hydrogen peroxidase for 5 min at 37 °C. The counterstaining was done with Harris’ hematoxylin for 1 min. Positive and negative controls of staining were used in all assays.
Immunostaining was analyzed according to a subjective evaluation: IL-6 (cytoplasmic staining as positive); STAT3 (nuclear or cytoplasmic staining were considered as positive) and was scored by two observers. The scoring ranged from 0 to 2 and considered the staining intensity: a) weak or negative staining (0); b) moderate (1) and c) intense (2). Normal kidney tissue was used as a positive reaction control.
Western blotting
Total protein extracts of BeWo cells and primary culture cells of normal placenta were obtained by using lysing solution containing RIPA buffer and a protease inhibitor mix (1 mM phenylmethylsulfonyl fluoride [PMSF], 3% benzamidine, 3% aprotinin and 3% leupeptin). Samples were run on 10% polyacrylamide gels containing SDS, where 50 μg of protein was applied to each lane. After electrophoresis, the proteins were transferred to nitrocellulose membranes (Hybond-ECL, Amersham, UK) and non-specific binding sites were blocked by incubating the membranes with TBS containing 0.05% Tween 20 (TBST) and 5% dry milk for 4 h, at room temperature. The membranes were subsequently rinsed, incubated overnight at 4 °C with primary antibody (mouse monoclonal anti-IL-6 antibody diluted 1:100 or mouse monoclonal anti-STAT-3 antibody diluted 1:200, all from Santa Cruz Biotechnology). A monoclonal anti-β-actin antibody from Sigma, diluted 1:100, was used as an internal control to detect β-actin. The membranes were washed in TBS and then incubated with secondary antibody (anti-mouse IgG conjugated to peroxidase diluted 1:500 from Advance HRP, Dako, Carpinteria, California, USA). After a final wash with TBS, the bound enzyme was detected by chemiluminescence, according to the manufacturer’s instructions (Amersham®).
Analysis was performed using NIH image analysis software (Image J 1.42q, National Institutes of Health, NIH, Bethesda, Maryland, USA) and expressed in comparison to βeta-actin.
Real-time PCR
mRNA levels of the Telomerase gene - Hs00162669_m1 was measured in BeWo cells and primary culture cells from normal placenta by Real-time PCR. Normal placenta was used as control and GAPDH #4326317E as housekeeping gene. The assays were purchased from Applied Biosystems® and mRNA expression was carried out in an ABI Prism 7500 sequence detection system (Applied Biosystems®). The optimal concentration of cDNA and primers, as well as the maximum efficiency of amplification were obtained through five-point, 2-fold dilution curve analysis for each gene. Each PCR contained 40 ng of reverse-transcribed RNA (High Capacity cDNA Kit - Applied Biosystems®) and was run according to the manufacturer’s recommendations using the TaqMan PCR master mix. Real-time data were analyzed using the Sequence Detector System 1.7 (Applied Biosystems®).
Image analysis and statistics
Descriptive statistics and comparison tests were performed by Chi-Square to compare proportions and the Mann-Whitney test to compare means. Statistical significance was accepted for the values of p ≤ 0.05. All analyzes were performed using the commercial program PASW Statistics (SPSS) 18.0.
Results
Immunohistochemical reactions
IL-6 expression
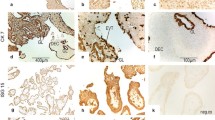
Placental tissue from abortions showed moderate staining for IL-6 in the cyto and syncytiotrophoblast cells of chorionic villi and in the decidual cells (Fig. 1.1-1.3). In normal at term placentas an intense and diffuse IL-6 expression was observed with reactive cells present in different tissue compartments. In the chorionic villi, IL-6 was expressed in the cytoplasm of syncytiotrophoblast, in endothelial cells, in smooth muscle cells of the vessel wall and in stromal mesenchymal cells. In the decidua region, IL-6 was expressed mainly in extra-villous cytotrophoblast but focally in endothelial and smooth muscle cells of blood vessels (Fig. 1.4-1.6). In choriocarcinomas, IL-6 showed an intense and diffuse expression in the cyto and syncytiotrophoblast neoplastic cells, like to the staining observed in normal at term placentas (Fig. 1.7-1-9).
Immunohistochemical expression of IL-6 in first trimester abortion material (1.1–1.3), normal at term placenta (1.4–1.6) and choriocarcinoma (1.7–1.9). At the top of the figures: IL-6 positivity is observed in the cytoplasm of trophoblasts and basal decidua. At the bottom of the figures: in the higher magnification (40x): reactive cells in the decidua (d), cyto (Ct) and sinciciotrophoblastic (Sc) cells of the chorionic villi (v), mesenchymal cells (m) and fetal capillaries (fc)
STAT-3 expression
Placental tissue from abortions showed moderate staining of STAT-3 in decidual cells, in villous cyto and syncytiotrophoblast, in extra-villous trophoblast and in villous mesenchymal cells (Fig. 2.1-2.3). Similar intensity of reaction was observed in normal at term placentas, with predominance of moderate staining in the cyto and syncytiotrophoblast, and in the cells of the extra-villous trophoblast in the decidual region (Fig. 2.4-2.6). In choriocarcinomas, STAT-3 was highly expressed in the cytoplasm of the tumor cells, in endothelial cells, smooth muscle cells of blood vessels, as well as in residual endometrium (Fig. 2.7-2.9; Fig. 3).
Immunohistochemical expression of STAT-3 in first trimester abortion material (2.1–2.3), normal at term placenta (2.4–2.6) and choriocarcinoma (2.7–2.9). At the top of the figures: Positive staining for STAT-3 is observed in the cytoplasm of trophoblastic cells and basal decidua. At the bottom of the figures: in the higher magnification (40x): reactive cells in the decidua (d), cyto (Ct) and sinciciotrophoblastic (Sc) cells of the chorionic villi (v), mesenchymal cells (m) and fetal capillaries (fc)
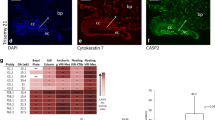
Comparative immunohistochemical analysis of intensity of reactions related to the expressions of IL-6 and STAT-3. Intense IL-6 expression was observed in at term placentas and choriocarcinomas and moderate IL-6 expression in abortion samples, without significant statistical differences (chi-square test, p = 0.092). STAT-3 expression was intense in choriocarcinomas and moderate in abortion and normal placenta samples, with significant statistical differences (chi-square test, *p = 0.037)
A comparative statistical analysis between the 3 groups showed statistically significant differences for the STAT-3 expression (p = 0.037), but not for IL-6 (p = 0.092).
Western blotting
IL-6 and STAT-3 expressions in the normal placenta primary culture were lower than in the BeWo cells. In all situations, the immunoreactive bands were compared to the presence of β-actin antibody as control reaction (Fig. 4).
Western blotting for IL-6 and STAT-3 in BeWo cell line and in primary culture of normal placenta. Note that there are three independent samples (1, 2 and 3) for BeWo cells and for primary culture of placenta normal. IL-6 and STAT-3 expressions were higher in BeWo cells as compared to primary cultured of normal placenta
Real-time PCR
The analysis of telomerase mRNA showed a high expression in choriocarcinomas and no expression in normal placenta primary culture (p = 0.0001). Null expression of telomerase was ensured by mRNA expression of the human housekeeping gene Gapdh, used as a control to normalize the relative expression values of the gene of interest, validating the target gene system (Fig. 5).
Telomerase gene expression by Real time PCR. No expression of telomerase in primary cultures of normal placenta contrasting to its high expression in BeWo cells. The comparative statistical analysis by Mann-Whitney showed statistically significant differences for the telomerase expression (*p = 0.0001)
Discussion
The success of embryonic implantation depends on the adequate orchestration between proliferation, migration, and invasion of the trophoblast in the endometrium, to establish the anchorage and blood supply of the fetus, through the formation of the villous chamber (Dubinsky et al. 2010). Studies have indicated the role of IL-6 in controlling these events and the maternal immune system is a critical participant in the establishment of pregnancy, its progression and maintenance (Munoz-Suano et al. 2011).
During the pre-implantation period, a pro-inflammatory environment facilitates the preparation of the embryo implantation and the acquisition of maternal immune tolerance. After implantation and with the evolution of the pregnancy, there will be an anti-inflammatory environment that restricts the immune system through suppressor regulatory cells, characteristic of the immunological tolerance state of pregnancy. At the end of pregnancy, a pro-inflammatory environment appears again, defining the scenario of childbirth and birth (Prins et al. 2012).
These data are confirmed by studies by Prins et al. (Prins et al. 2012), who demonstrated that the IL-6 protein and its mRNA are expressed in the decidua and in the placenta throughout pregnancy, in different concentrations depending on the period (De et al. 1992). IL-6 is related to angiogenesis and to the vascular remodeling necessary for morphogenesis in early pregnancy. In addition, it also regulates the balance between the invasion properties acquired by the trophoblast (Champion et al. 2012) and its activity during pregnancy. These data are confirmed by our results, because IL-6 was present since the early stages of pregnancy, expressed in the first trimester abortion samples, with increased expression at the end of pregnancy, as observed in at term placentas. This high expression was also seen in carcinogenesis, in the choriocarcinoma trophoblastic tissue.
According to Jasper et al. (Jasper et al. 2007) the moderate expression of IL-6 in abortion materials is consistent with the known functions of IL-6 in coordinating the balance of T cell phenotypes, generating cell populations necessary for the establishment of immunological tolerance in pregnancy. The high expression of IL-6 in at term placentas is related to the physiological processes of childbirth, because IL-6 stimulates uterine contractility through the induction of oxytocin expression (Fang et al. 2000). In choriocarcinomas IL-6 maintains the high proliferative invasion of the carcinogenic process.
On the other hand, the loss of IL-6 protein expression in tissues grown in vitro reinforces the idea that this cytokine should be required in the processes of cell invasion and proliferation, which are gradually deactivated during the formation of the confluent sheet in the culture (Kruse and Miedema 1965). Perhaps, the results could be different if we analyzed the production of IL-6 during the phases of adhesion to the substrate and formation of colonies, during the first 3 days of implantation of the cells to the in vitro system.
In relation to STATs, these are known as transcription-activating proteins, responsible for carrying information from the cytoplasm to the nucleus, expressed by many cell types and activated by a variety of cytokines and growth factors. The precise way of regulating STAT activation is critical in terms of causing an appropriate extracellular signaling pathway. Studies show that when the control of STAT activation becomes unregulated, there is a change in it signaling, which contributes to the malignant cell transformation, leading to cell cycle progression and cell survival (Fitzgerald et al. 2008).
According to Takahashi et al. (Takahashi et al. 2008), the JAK-STAT-3 signaling cascade is an important pathway for trophoblastic cell differentiation, since STAT-3 is known for its ability to regulate the expression of proteins that increase the invasion and migration of tumor cells (Germain and Frank 2007; Chan et al. 2008). Thus, the intense and progressive presence of STAT-3 observed in our immunohistochemical study during pregnancy (abortions and at term placentas) and choriocarcinoma, seems to confirm the possibility that the JAK-STAT-3 pathway is progressively deactivated after nesting and implantation of the fetus in the endometrium. From then on, both the deactivation of this preferential route and the activation of alternative routes are required to sustain the pregnancy until its end.
We could think that in the process of carcinogenesis, the deactivation of STAT-3 would not trigger apoptosis, which favors the stability of atypical cell clones of choriocarcinoma. However, considering that cytokines lead to proliferative and antiproliferative responses, depending on the cell type, the role of STAT-3 signaling pathways in cell control and apoptosis still requires further investigations.
Telomerase
Telomerase is a cellular reverse transcriptase, which adds 5 ‘-d (TTAGGG) -3’ hexameric repeats at the 3 ‘end of the chromosomal DNA helix, providing stability and integrity to the chromosome telomeric regions. Studies show that telomerase activity is associated with most human malignant tumors, since the increase in its activity in tumor cells maintains the integrity of the telomeres and ensures the capacity for cell proliferation (Chen et al. 2002; Collins and Mitchell 2002).
According to Castellucci and Kaufmann (Castellucci et al. 2000), in normal pregnancies, the villous trophoblast consists of a proliferative population of cytotrophoblasts, which differentiates and merges to form the syncytiotrophoblast. In the first trimester of pregnancy, the proliferation rate of cytotrophoblasts is high, gradually decreasing in the following months and, in the third trimester, this proliferation of trophoblasts falls to about 10% of the values observed in the first trimester (Chen et al. 2002; Huppertz et al. 1998).
Our results confirmed this behavior in the at term placenta group, as the telomerase expression for the Hs00162669_m1 gene was absent in normal mature placenta and in the culture of this fresh placental tissue. On the other hand, there was a high expression of telomerase in the BeWo cells, demonstrating its importance in the process of neoplastic proliferation.
According to data from CHEN et al. (Chen et al. 2002), of 37 samples of normal term placenta, only 11 (30%) expressed some telomerase activity, contrasting with the null expression of 26 placentas (70%). In the same study, 100% of the samples of choriocarcinoma showed telomerase activity.
Thus, the increase in telomerase activity in BeWo cells reinforces its relationship with the malignant phenotype, indicating it as a good marker for disease progression.
Conclusions
Our study showed that, in choriocarcinomas, IL-6 and STAT-3 are involved in tumor progression. Finally, the presence of telomerase gene expression allows us to assert that increased telomerase is associated with tumor progression and cell immortalization, as observed in BeWo cells.
Availability of data and materials
Not applicable.
Abbreviations
- IL-6:
-
Interleukin-6
- STAT-3:
-
Signal transducer and activator of transcription-3
- JAK:
-
Signaling pathway through Janus kinases
- ECM:
-
Extracellular matrix
- TIMP:
-
Metalloproteinase inhibitors
- GTD:
-
Gestational trophoblastic diseases
- H&E:
-
Hematoxylin and eosin
- FBS:
-
Fetal bovine serum
- PCR:
-
Polymerase chain reaction
- TVA:
-
Trypsin-versine association
- IHC:
-
Immunohistochemistry
- PBS:
-
Phosphate buffered saline
- DAB:
-
Diaminobenzine tetrahydrochloride
- DMSO:
-
Dimethyl sulfoxide
- PMSF:
-
Phenylmethylsulfonyl fluoride
- SDS:
-
Sodium dodecyl sulphate
- TBS:
-
Tris buffered saline
- TBST:
-
Tween tris-buffered saline
- DNA:
-
Deoxyribonucleic acid
- cDNA:
-
Complementary DNA
- RNA:
-
Ribonucleic acid
- mRNA:
-
Messenger RNA
References
Bischof P (2001) Endocrine, paracrine and autocrine regulation of trhophoblastic metalloproteinases. Early Pregnancy 5(1):30–31
Castellucci M, Kosanke G, Verdenelli F, Huppertz B, Kaufmann P (2000) Villous sprouting: fundamental mechanisms of human placental development. Hum Reprod Update 6(5):485–494
Champion H, Innes BA, Robson SC, Lash GE, Bulmer JN (2012) Effects of interleukin-6 on extravillous trophoblast invasion in early human pregnancy. Mol Hum Reprod 18(8):391–400
Chan HY, Siu MKY, Zhang HJ, Wong ESY, Ngan HYS, Chan KYK, Cheung ANY (2008) Activated Stat3 expression in gestational trophoblastic disease: correlation with clinicopathological parameters and apoptotic indices. Histopathology 53:139–146
Chen RJ, Chu CT, Huang SC, Chow SN, Hsieh CY (2002) Telomerase activity in gestational trophoblastic disease and placental tissue from early and late human pregnancies. Hum Reprod 17:463–468
Chiesa C, Pacifico L, Natale F, Hofer N, Osborn JF, Resch B (2015) Fetal and early neonatal interleukin-6 response. Cytokine 76(1):1–12
Collins K, Mitchell JR (2002) Telomerase in the human organism. Oncogene. 21:564–579
De M, Sanford TH, Wood GW (1992) Detection of interleukin-1, interleukin-6, and tumor necrosis factor-alpha in the uterus during the second half of pregnancy in the mouse. Endocrinol 131:14–20
Dimitriadis E, Nie G, Hannan NJ et al (2010) Local regulation of implantation at the human fetal-maternal interface. Int J Dev Biol 54:313–322
Dubinsky V, Poehlmann TG, Suman P et al (2010) Role of regulatory and angiogenic cytokines in invasion of trophoblastic cells. Am J Reprod Immun 63:193–199
Fang X, Wong S, Mitchell BF (2000) Effects of LPS and IL-6 on oxytocin receptor in non-pregnant and pregnant rat uterus. Am J Reprod Immunol 44:65–72
Fitzgerald JS, Germeyer A, Huppertz B, Jeschke U, Knöfler M, Moser G, Scholz C, Sonderegger S, Toth B, Markert UR (2010) Governing the invasive Trophoblast: current aspects on intra- and extracellular regulation. Am J Reprod Immunol 63:492–505
Fitzgerald JS, Poehlmann TG, Schleussner E et al (2008) Trophoblast invasion: the role of intracellular cytokine signaling via signal transducer and activator of transcription 3 (STAT-3). Hum Reprod Update 14(4):335–344
Germain D, Frank DA (2007) Targeting the cytoplasmatic and nuclear functions of signal transducers and activators of transcription 3 for cancer therapy. Clin Cancer Res 13:5665–5669
Huppertz B, Frank HG, Kingdom JC, Reister F, Kaufmann P (1998) Villous cytotrophoblast regulation of the syncytial apoptotic cascade in the human placenta. Histochem Cell Biol 110(5):495–508
Jasper MJ, Tremellen KP, Robertson SA (2007) Reduced expression of IL-6 and IL-1alpha mRNAs in secretory phase endometrium of women with recurrent miscarriage. J Reprod Immunol 73:74–84
Kishimoto T (2005) IL-6: from laboratory to bedside. Clin Rev Allergy Immunol 28:177–186
Knöfler M, Pollheimer J (2012) IFPA award in Placentology lecture: molecular regulation of human trophoblast invasion. Placenta 26:S55–S62
Konjević G (2009) STAT proteins in cancerogenesis and therapy of malignancies. Srp Arh Celok Lek 137(1–2):98–105
Kruse PF, Miedema E (1965) Production and characterization of multiple-layered populations of animal cells. J Cell Biol 27:273–279
Monchek R, Wiedaseck S (2012) Gestational trophoblastic disease: an overview. J Midwifery Womens Health 57:255–259
Munoz-Suano A, Hamilton AB, Betz AG (2011) Gimme shelter: the immune system during pregnancy. Immunol Rev 241:20–38
Prins JR, Lopeza NG, Robertsona SA (2012) Interleukin-6 in pregnancy and gestational disorders. J Reprod Immun 95:1–14
Takahashi U, Takahashi M, Carpino N et al (2008) Leukemia inhibitory factor regulates trophoblast giant cell differentiation via Janus kinase 1-signal transducer and activator of transcription 3-suppressor of cytokine signaling 3 pathway. Mol Endocrinol 22(7):1673–1681
Wen W, Liang W, Wu J et al (2014) Targeting JAK1/STAT3 signaling suppresses tumor progression and metastasis in a peritoneal model of human ovarian cancer. Mol Cancer Ther 13(12):3037–3048
Acknowledgements
The authors thank Prof. Dr. Lício Augusto Velloso and the student Joseane Morari for their excellent technical assistance in the Medical Physiopathology Laboratory; Dr. Marcelo Nomura of Department of Obstetrics and Gynecology, Faculty of Medical Sciences for their assistance with the collection of human placentas and Dr. Antonio Fazano and Josianne Nascimento for the loan of their excellent framework for the achievement of cell cultures.
Funding
This research was supported by Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP), Coordenadoria de Aperfeiçoamento do Ensino Superior (CAPES) and Conselho Nacional de Pesquisa e Desenvolvimento Tecnológico (CNPq).
Author information
Authors and Affiliations
Contributions
Prof. Dr. Lício Augusto Velloso and the technique Joseane Morari helped with technical assistance in the Medical Physiopathology Laboratory; Dr. Marcelo Nomura of Department of Obstetrics and Gynecology, Faculty of Medical Sciences helped in the assistance with the collection of human placentas; Profa. Dra. Fátima Bottcher and Profa. Dra. Liliana Andrade guided me, Luciana Pietro, in conducting the work, that is, my doctoral thesis. The author(s) read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was approved by the Ethics Committee of the Faculty of Medical Sciences, Campinas State University (UNICAMP), on 26/06/2009, with CEP n°. 368/2009, C.A.A.E. 0282.0.146.000–09.
Consent for publication
Informed consent was obtained from all individual participants included in the study.
Competing interests
The author(s) declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Summary sentence: Blastocyst implantation and neoplastic invasion have some common properties related to tissue invasion. With the aim of investigating the role of IL-6 (Interleukin-6) and the transcription activator STAT-3, we compared the expression of these molecules in fresh and fixed materials from placentas of abortion, normal at term placentas and choriocarcinomas.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pietro, L., Bottcher-Luiz, F., Velloso, L.A. et al. Expression of interleukin-6 (IL-6), signal transducer and activator of transcription-3 (STAT-3) and telomerase in choriocarcinomas. Surg Exp Pathol 3, 28 (2020). https://doi.org/10.1186/s42047-020-00080-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s42047-020-00080-1