Abstract
Purpose
The aim of this study is to test the hypothesis that feeding trans fatty acids (TFA) (5%) along with fructose exacerbates obesity and non-alcoholic fatty liver disease (NAFLD) in rats.
Methods
Male Wistar rats were randomized into four groups, i.e., standard diet, 5% TFA + standard diet, fructose + standard diet, and TFA + fructose + standard diet. All the diets were provided for 16 weeks. The body weight, body mass index, calorie intake, adiposity index, and liver index were determined. Serum glucose, insulin, lipid profile, and liver enzymes were estimated. Liver lipids, markers of oxidative stress, inflammation, and collagen were estimated in the liver. The histopathological evaluation of the adipose tissue and liver were carried out.
Results
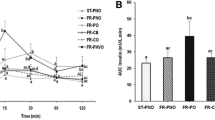
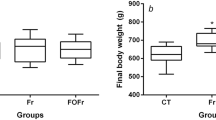
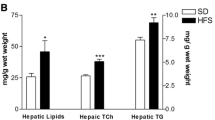
TFA + standard diet caused an increase in body weight while TFA + fructose + standard diet caused significant body weight gain, adiposity index, and hypertrophy of adipocytes. TFA + fructose + standard diet caused insulin resistance and dyslipidemia in the rats. Rats in the TFA + standard diet group showed marked hepatic steatosis and an elevation in alanine aminotransferase, while those in the TFA + fructose + standard diet group showed oxidative stress, inflammation, and fibrosis in the liver.
Conclusion
Feeding of TFA at a concentration of 5% along with the standard diet resulted in an increase in the body weight and hepatic steatosis, but the addition of fructose to 5% TFA and standard diet resulted in obesity and non-alcoholic steatohepatitis. Thus, the reduction in TFA content of foods must be accompanied by a significant decrease in the fructose intake in order to protect against obesity and NAFLD.


Similar content being viewed by others
Abbreviations
- AC/TC:
-
ratio-abdominal circumference to thoracic circumference ratio
- ALP:
-
alkaline phosphatase
- ALT:
-
alanine aminotransferase
- AST:
-
aspartate aminotransferase
- BMI:
-
body mass index
- GGT:
-
gamma-glutamyl transferase
- GSH:
-
reduced glutathione
- HDL-C:
-
high density lipoprotein cholesterol
- HOMA-IR:
-
homeostasis model assessment-insulin resistance
- LDL-C:
-
low density lipoprotein cholesterol
- MDA:
-
malondialdehyde
- NAFLD:
-
non-alcoholic fatty liver disease
- NASH:
-
nonalcoholic steatohepatitis
- PHVO:
-
partially hydrogenated vegetable oil
- SOD:
-
superoxide dismutase
- TFA:
-
trans fatty acids
- TG:
-
triglycerides
- VLDL-C:
-
very low density lipoprotein cholesterol
- WHO:
-
World Health Organization
References
Drewnowski A. The real contribution of added sugars and fats to obesity. Epidemiol Rev. 2007;29:160–71.
WHO calls on countries to reduce sugars intake among adults and children. Available at https://www.who.int/mediacentre/news/releases/2015/sugar-guideline/en/. Accessed 11 June 2019.
Walker RW, Dumke KA, Goran MI. Fructose content in popular beverages made with and without high-fructose corn syrup. Nutrition. 2014;30:928–35.
Dornas WC, de Lima WG, Pedrosa ML, Silva ME. Health implications of high-fructose intake and current research. Adv Nutr. 2015;6:729–37.
Bhardwaj S, Passi SJ, Misra A. Overview of trans fatty acids: biochemistry and health effects. Diabetes Metab Syndr. 2011;5:161–4.
Micha R, Khatibzadeh S, Shi P, Fahimi S, Lim S, Andrews KG, et al. Global, regional, and national consumption levels of dietary fats and oils in 1990 and 2010: a systematic analysis including 266 country-specific nutrition surveys. BMJ. 2014;348:g2272. https://doi.org/10.1136/bmj.g2272.
Brownell KD, Pomeranz JL. The trans-fat ban-food regulation and long term health. N Engl J Med. 2014;370:1773–5.
WHO plan to eliminate industrially-produced trans-fatty acids from global food supply. Available from http://www.who.int/news-room/detail/14-05-2018-who-plan-to-eliminate-industrially-produced-trans-fatty-acids-from-global-food-supply. Accessed 16 August 2018.
FSSAI launches new campaign to eliminate trans fats by 2022. Available from https://www.thehindubusinessline.com/news/national/fssai-launches-new-campaign-to-eliminate-trans-fats-by-2022/article25636525.ece. Accessed 15 December 2018.
Koppe SW, Elias M, Moseley RH, Green RM. Trans fat feeding results in higher serum alanine aminotransferase and increased insulin resistance compared with a standard murine high-fat diet. Am J Physiol Gastrointest Liver Physiol. 2009;297:G378–84.
Jeyapal S, Putcha UK, Mullapudi VS, Ghosh S, Sakamuri A, Kona SR, et al. Chronic consumption of fructose in combination with trans fatty acids but not with saturated fatty acids induces non-alcoholic steatohepatitis with fibrosis in rats. Eur J Nutr. 2018;57:2171–87. https://doi.org/10.1007/s00394-017-1492-1.
de Lima VM, Oliveira CP, Alves VA, Chammas MC, Oliveira EP, Stefano JT, et al. A rodent model of NASH with cirrhosis, oval cell proliferation and hepatocellular carcinoma. J Hepatol. 2008;49:1055–61.
Zhao X, Shen C, Zhu H, Wang C, Liu X, Sun X, et al. Trans-fatty acids aggravate obesity, insulin resistance and hepatic steatosis in C57BL/6 mice, possibly by suppressing the IRS1 dependent pathway Molecules 2016;21(6). pii:E705. doi: https://doi.org/10.3390/molecules21060705.
Machado RM, Stefano JT, Oliveira CP, Mello ES, Ferreira FD, Nunes VS, et al. Intake of trans fatty acids causes nonalcoholic steatohepatitis and reduces adipose tissue fat content. J Nutr. 2010;140:1127–32.
Obara N, Fukushima K, Ueno Y, Wakui Y, Kimura O, Tamai K, et al. Possible involvement and the mechanisms of excess trans-fatty acid consumption in severe NAFLD in mice. J Hepatol. 2010;53:326–34.
Fakhoury-Sayegh N, Trak-Smayra V, Sayegh R, Haidar F, Obeid O, Asmar S, et al. Fructose threshold for inducing organ damage in a rat model of non-alcoholic fatty liver disease. Nutr Res. 2019;62:101–12.
Novelli EL, Diniz YS, Galhardi CM, Ebaid GM, Rodrigues HG, Mani F, et al. Anthropometrical parameters and markers of obesity in rats. Lab Anim. 2007;41:111–9.
Adorni CS, Corrêa CR, Vileigas DF, de Campos DH, Padovani CR, Minatel IO, et al. The influence of obesity by a diet high in saturated fats and carbohydrates balance in the manifestation of systemic complications and comorbidities. Nutrire. 2017;42(1):16.
Wallace TM, Levy JC, Matthews DR. Use and abuse of HOMA modeling. Diabetes Care. 2004;27:1487–95.
Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18:499–502.
Folch J, Lees M, Sloane Stanley GH. A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem. 1957;226:497–509.
Lo AT, Francisqueti FV, Hasimoto FK, Ferraz AP, Minatel IO, Garcia JL, et al. Brazilian Curcuma longa L. attenuates comorbidities by modulating adipose tissue dysfunction in obese rats. Nutrire. 2018;43(1):25.
Neuman RE, Logan MA. The determination of hydroxyproline. J Biol Chem. 1950;184:299–306.
Pai SA, Majumdar AS. Protective effects of melatonin against metabolic and reproductive disturbances in polycycstic ovary syndrome in rats. J Pharm Pharmacol. 2014;66:1710–21.
Kismet K, Ozcan C, Kuru S, Gencay Celemli O, Celepli P, Senes M, et al. Does propolis have any effect on non-alcoholic fatty liver disease? Biomed Pharmacother. 2017;90:863–71.
Mattes R, Foster GD. Food environment and obesity. Obesity. 2014;22:2459–61.
Lindqvist A, Baelemans A, Erlanson-Albertsson C. Effects of sucrose, glucose and fructose on peripheral and central appetite signals. Regul Pept. 2008;150:26–32.
Longhi R, Almeida RF, Machado L, Duarte MM, Souza DG, Machado P, et al. Effect of a trans fatty acid-enriched diet on biochemical and inflammatory parameters in Wistar rats. Eur J Nutr. 2017;56:1003–16.
Mazidi M, Katsiki N, Mikhailidis DP, Banach M. Link between plasma trans-fatty acid and fatty liver is moderated by adiposity. Int J Cardiol. 2018;272:316–22.
Buzzetti E, Pinzani M, Tsochatzis EA. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism. 2016;65:1038–48.
Dhibi M, Brahmi F, Mnari A, Houas Z, Charqui I, Bchir L, et al. The intake of high fat diet with different trans fatty acid levels differentially induces oxidative stress and non alcoholic fatty liver disease (NAFLD) in rats. Nutr Metab. 2011;8:65. https://doi.org/10.1186/1743-7075-8-65.
Mraz M, Haluzik M. The role of adipose tissue immune cells in obesity and low-grade inflammation. J Endocrinol. 2014;222:R113–27.
Bhardwaj S, Passi SJ, Misra A, Pant KK, Anwar K, et al. Effect of heating/reheating of fats/oils, as used by Asian Indians, on trans fatty acid formation. Food Chem. 2016;212:663–70.
Acknowledgements
The authors are grateful to Ms. Deepali Ganachari and Ms. Jaya Verma, research fellows at Dept. of Clinical Pharmacology, T.N. Medical College and BYL Nair Hospital for their help during animal experimentation, and Dr. Sanjay Pawar, veterinary pathologist for carrying out the histopathological evaluation of the liver and adipose tissue.
Funding
The University Grants Commission (BSR Fellowship), New Delhi, India, letter no. F.25–1/2014–15 (BSR)/No. F.5–63/2007 (BSR) dated 16 Feb 2015 is awarded to Ms. Sarayu Pai.
Author information
Authors and Affiliations
Contributions
SAP carried out the experiments and wrote the first draft of the manuscript. RPM helped in designing the study and analyzing the data. ARJ was involved in designing the study and supervision of the conduct of the work. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The protocol was approved by the Institutional Animal Ethics Committee of T N Medical College and BYL Nair Charitable Hospital, Mumbai, India (protocol no. IAEC/2016/8 dated 29 February 2016).
Conflict of interest
The authors declare that they have no conflict of interests.
Electronic supplementary material
Supplementary table 1
(DOCX 29 kb)
Rights and permissions
About this article
Cite this article
Pai, S.A., Munshi, R.P. & Juvekar, A.R. Partially hydrogenated vegetable oil containing 5% trans fats when combined with fructose exacerbates obesity and non-alcoholic fatty liver disease in rats. Nutrire 45, 5 (2020). https://doi.org/10.1186/s41110-019-0105-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41110-019-0105-6