Abstract
Nanoparticles (NPs) of metal oxides, sometimes referred to as engineered nanoparticles have been used to protect building surfaces against biofilm formation for many years, but their history in the Cultural Heritage world is rather short. Their first reported use was in 2010. Thereafter, a wealth of reports can be found in the literature, with Ti, Ag and Zn oxides being the major protagonists. As with all surface treatments, NPs can be leached into the surrounding environment, leading to potential ecotoxicity in soil and water and associated biota. Dissolution into metal ions is usually stated to be the main mode of toxic action and the toxic effects, when determined in the marine environment, decrease in the order Au > Zn > Ag > Cu > Ti > C60, but direct action of NPs cannot be ruled out. Although ecotoxicity has been assessed by a variety of techniques, it is important that a suitable standard test be developed and the European Unions’s Biocidal Product Registration group is working on this, as well as a standard test for antimicrobial efficacy to determine their impact on ecological processes of surrounding non-target organisms and their transformation products under realistic scenarios.
Similar content being viewed by others
Use of nanoparticles in the protection of cultural heritage buildings
Nanoparticles (NPs) are produced both as a result of natural events such as volcanic eruption, terrestrial dust storms, erosion, and forest fires and are thus considered naturally produced nanoparticles. However, there is another type of nanoparticles that are produced by man, known as engineered nanoparticles (ENPs), which are manufactured using different materials such as metal oxides, metals, carbon, polymers and lipids [1]. The potential of nanoparticles (NPs) for conservation of built structures has been established for the consolidation of decayed materials, production of de-polluting surfaces, self-cleaning, surface enhancement or as a biocide to reduce biodeterioration [2,3,4,5]. Titanium dioxide (anatase) has been employed for many years [6, 7] and in some respects has become the gold standard for self-cleaning surfaces. TiO2 NPs have high stability and photo-reactivity, are active against a broad spectrum of microorganisms and, importantly, have low cost. However, the conservation community was slow to realize the potential of this photocatalytic process for cultural heritage buildings. In 2009, a research paper was published to demonstrate the effectiveness of TiO2 in preventing fungal colonization on concrete, and the authors included historic buildings in their discussion as structures that could be protected by this means [8]. Fonseca et al. [9] tested the efficacy of TiO2 (anatase) on the “Palacio Nacional da Pena in Sintra, Portugal”, and showed that it was more effective than two conventional biocides. It has also been suggested that TiO2 NPs may decrease the bioreceptivity of the treated surface because of its self-cleaning ability [10], and this would reduce recolonization after remediative treatment.
Since then, an increasing number of studies of the use of these photocatalytic nanoparticles on buildings of cultural heritage have been published and tested, with different metals or hybrids containing more than one type of nanoparticle (NPs) [e.g. 4, 11]. Suspensions of mixed NPs or NPs mixed with other chemicals have been tested for their efficacy against biofouling of building envelopes. Frequently the mixtures are found to be more effective than a single NP, advocating synergistic action. Substances that have been mixed with nanoparticles to improve function include biocides, heavy metals, and water repellants/stone consolidants. La Russa et al. [5] found that Ag, Fe and Sr could increase the activity of TiO2 and could be applied for stone preservation. Although Becerra et al. [12] confirmed the additive effect of Ag on TiO2 nanoparticles for protection of limestone, Graziani et al. [13] found that the addition of neither Cu nor Ag improved the self-cleaning and anti-biofouling activities of TiO2 nanoparticles, while Pinna et al. [14] found that, although copper nanoparticles could be a suitable alternative to traditional biocides such as tin derivatives for protection of archaeological stone (marble, sandstone and plaster), they did not reduce regrowth sufficiently. Further examples can be found in Fonseca et al. [8], Pinna et al. [15], Banach et al. [16], and Batista Goffredo et al. [17].
A potential new nanosystem has been suggested for constructional materials, based on a natural halloysite nanotube-based carrier. The aluminosilicate clay mineral nanotubes were loaded with a biocide and the system found to effectively prevent biological growth on mortar over 2 years [18]. This could be a useful addition to the armoury of the historic building restorer. A potential application, specific for cultural heritage buildings as opposed to modern constructions, is the eco-friendly application of biocidal nanoparticles in a water repellant. Such a nano-composite, composed of TiO2 and poly(carbonate urethane), was produced and tested by D’Orazio and Grippo [19]. It was shown to have self-cleaning and water desorption activities.
Recently, Toniolo and Gherardi [20] overviewed the protection of historic marble by NP coatings, concluding that suitably modified nanocomposites based on TiO2 seem very promising. They also considered the use of laboratory versus field testing of coatings; each has its place, but only field tests can predict the true protective value of an experimental treatment. The influence of test site (environment) is exemplified by the following two publications. An extended testing of TiO2, Ag and ZnO NPs in siloxane wax for protection of underwater marble resulted in the patenting of a new product for underwater cultural property, based on NPs containing TiO2 and Ag [21]. On a terrestrial site, however, a historic stone building in Italy, TiO2 NPs had less success. Ruffolo et al. [22] tested pure TiO2 (anatase 25 nm) and TiO2 mixed with silver nanoparticles at a ratio of 100/1 in aqueous dispersion of nanosilica. The solutions were applied to selected sites on the stone walls of Villa dei Papiri in Herculanum, Naples, Italy by brushing, 3 months after cleaning with an organic biocide (Biotin R). Results were monitored over 8 months. Although the biocide cleaning was effective, no difference was seen between control and NP-treated areas after 4 months and, in fact, higher recolonization occurred on treated areas in high humidity zones, close to the ground.
Similarly, spray application of TiO2 NPs, with or without Ag and Cu NPs, was unable to increase significantly the durability of travertine (limestone) subjected to artificial accelerated ageing [23]. Factors affecting efficacy of NPs on building materials include porosity and roughness; when these are high, NP treatment is less likely to be effective [24]. The latter authors record the effectiveness (or lack of it) on phototroph growth on three types of limestone, two of sandstone and a tuff. They suggest that TiO2 is not a suitable treatment for preservation of porous and rough stone. Many examples, where NP treatment is effective in one case, but not in another, can be found. Hence NPs are still not used routinely in the cultural heritage field, and their efficacy needs to be proven for each particular material and environment.
Probably the main metals currently used in NPs for cultural heritage buildings are Ti, Ag, Zn and Si. These are the same NPs that show the highest worldwide production values overall, with Si > Ti > Zn > Ag, followed by Cu [25]. Ortega-Morales et al. [26] have recently reviewed some of the literature on the use and testing of Ti, Ag, Zn and Cu NPs for protection of cultural heritage, as have Sierra-Fernandez et al. [27]. SiO2 NPs are used mainly as stone consolidants [28] and not as protective antimicrobial surfaces with anticipated inhibitory ecological effects, although a nanocomposite containing both SiO2 and a biocidal NP of CuO has been found useful as a biofilm inhibiting consolidant [29] and silica nanocontainers have been shown to be suitable in the production of a slow-release system for a natural antifouling agent [30].
The lack of really convincing evidence for the efficacy of nanoparticle coatings on cultural heritage buildings, together with the need for specific testing of relevant materials and environments, has limited the use of these potentially useful materials by conservators. As conservationists not only by name, but also by outlook, the potentially adverse effects on the environment of these relatively new tools are also considered of immense importance by those concerned with control and maintenance of our heritage. The next, and larger, section of this article considers these aspects.
Ecotoxicological consequences of release of nanoparticles in the environment
There is limited understanding of the environmental fate of engineered nanoparticles (ENPs) after release from treated surfaces. Ecotoxicological studies report different results of bacterial inhibition, stimulation, survival and death, which depend on dose, species and test procedures [31].
However, it is important to determine their impact on surrounding non-target organisms and ecological processes not only following leaching, but also during their production, especially since that production is estimated to grow to 58,000 tons per year by 2020. In 2010, it was estimated that up to 309,000 metric tons of ENPs were produced and that 63–91% of these ended up in landfills [32]. Therefore, the risks of engineered nanoparticles (ENPs) have been widely debated in recent years. Since the early discussion about the risks of NPs, chemical risk assessment (CRA) has been presented as the most relevant approach to understand, evaluate and quantify the ecotoxicological risks. CRA is an international process [recognized by WHO (World Health Organization), OECD (Organization for Economic Cooperation and Development)] and a fundamental ingredient of the REACH (Registration, Evaluation and Authorization of Chemicals) in which scientific and regulatory principles are applied in a systematic way to describe the hazards associated with environmental and/or human exposure to chemicals. CAR is a four-step process, consisting of: (1) hazard identification, (2) dose–response assessment, (3) exposure assessment and (4) risk characterization. Its main result is a statement of the probability that when human or other environmental receptors (e.g. plants, animals) are exposed to a chemical agent, they will be harmed and to what degree [33].
Gladis et al. [34] stressed the importance of active agents under development being subject to ecotoxicological assessments. Indeed, ENPs may potentially be released into the environment throughout their life cycle, from production to the end of their useful life. Ferrari et al. [35] carrying out a life cycle assessment of TiO2 NPs used in the protection of cultural heritage, determined, with the limited information available at that time, that the most important environmental damage was related to the production and application of TiO2 NPs sprays to the surface of the stone, with a major impact factor being the energy-intensive method of spray application; they stated that this could readily be reduced by using manual application. More recently, González-Gálvez et al. [36] reviewed the processes that occur throughout the life cycle of nano-products, including release studies.
After application, release of ENPs may result when the coatings are not fixed adequately to stone or when the durability of materials is not sufficiently effective to remain adhered to the substrate over a long period of time [37, 38]. Air is the principal factor that affects the fate of nanoparticles in the environment and is determined by three main factors: (1) the duration of time in which the particles remain in the air, (2) their interaction with other particles or molecules in the atmosphere, and (3) the distance that they can travel in the air. The important processes for understanding the dynamics of ENPs in the atmosphere are diffusion, agglomeration, wet and dry deposition and gravitational sedimentation. When considering the duration of time that ENPs remain in the air, they are assumed to follow the laws of gaseous diffusion. The rate of diffusion is inversely proportional to the particle diameter and the rate of gravitational sedimentation is proportional to it. In general, particles on the nanometer scale are considered to have a shorter residence time in air, compared to medium-sized particles, because they quickly agglomerate into much larger particles and settle to the ground. Here again ENPs with anti-agglomeration coatings differ and their residence time cannot be predicted. It is considered that deposited ENPs are generally not resuspended or re-aerosolized in the atmosphere [33].
When the nanoparticles enter water systems, they may have considerable impact on the ecosystem. For instance, through their antimicrobial action they can change the composition of the bacterial community in biofilms within the marine environment, thus affecting the settlement of invertebrate larvae and causing disequilibrium of the ecosystem [39]. Chen et al. [40] showed that Ag and ZnO NPs altered the functional bacterial community in activated sludge, inhibiting denitrifying bacteria at higher concentrations. It has been suggested that NP pollution would be greater in soils than in other recipient environments [41] and this gives cause for concern. Kaegi et al. [37, 42] using run-off experimental set-ups, were able to demonstrate unequivocally the direct release of silver and titanium oxide ENPs from façade paints (aged and new coatings) and their transport into surface waters and soils. Levard et al. [43] reported the transformation processes of Ag NPs that occur in various aqueous environments and how these impact on their stability and toxicity, while Bondarenko et al. [44] reviewed the ecotoxicity of ENPs containing Ag, CuO and ZnO, comparing the 50% lethal concentrations or minimum inhibitory concentrations (MIC) against algae, crustaceans, fish, bacteria, yeast, nematodes, protozoa and mammalian cell lines. Crustaceans, fish and, especially, algae were generally the most sensitive, and MIC values for bacteria were much higher, indicating that the levels needed to prevent biofilm formation would be well above those that are toxic to aquatic organisms. This implies that the risk of ENP release from coated surfaces in buildings represents a real issue in environmental terms, so that proper assessment is necessary.
Giese et al. [45], however, consider that ENPs are not a threat to most environmental compartments, apart from those located close to manufacturing or waste disposal plants. The authors point out, though, that many of the published studies in this area have flaws; hence it would be advisable to reduce release of these substances into the environment.
The development of slow-release formulations will help to ensure not only that antimicrobials have prolonged life at the site, but also that large amounts of NPs are not released rapidly into the environment. In a step towards this aim, ecofriendly, superhydrophobic coatings containing TiO2 and SiO2 nanoparticles were produced by Chen et al. [46]; the completely water-based coatings were self-healing and durable.
Graphene oxide has been shown to increase the stability of copper NPs [47] among others, and it not only stabilizes, but also increases the antimicrobial activity [48]. In this respect, silver graphene oxide (AgGO) has been shown to be a more effective antimicrobial than ZnGO [49]. Slate et al. [50] reviewed various modified graphenes and their use in advanced 2D antimicrobial coatings.
Halloysite, an aluminosilicate nanotubular material, can be loaded with antimicrobial NPs to produce a novel, slow-release product [51]. The nanotubes with metal oxide NPs have enhanced antimicrobial efficacy [52, 53].
Ecological effects of NPs in soils
ENPs may enter the soil system directly, by leaching from building surfaces, or indirectly, via wastewater sludges used as fertilizers [54]. ENPs tend to partition into the sludge in wastewater treatment plants [55] and their disposal into soil can result in contamination by the particles themselves, or their transformation products. Lahive et al. [56] showed that, in contradiction to several publications [e.g. 57], ENPs were more toxic to earthworms than their respective metal ions; earthworm reproduction was reduced by 90% when they were exposed to ENP-containing soil but only 22–27% in soil containing the metal ions Ag or, especially, Zn.
There are several controls dictating the biological impact and ultimate fate of ENP in soils. As well as pH, porosity of soils plays a key role, allowing ENPs to pass through the pore system, leaching into aquifer systems and eventually marine environments [42]. Engineered nanoparticle mobility is more efficient across mineral soils than in soils rich in organic colloids; therefore, this condition is determinant in establishing mobility and ultimate fate of NPs [58]. As ENPs reach the soil compartment, nanoparticles interact with organisms thriving in the surroundings of cultural heritage buildings. The effect of different metal nanoparticles can be seen on soil non-target macroorganisms (plants and invertebrates such as annelids, nematodes and insects) and microorganisms. Table 1 describes the important impacts for metal ENPs reviewed so far, relevant to the conservation of built cultural heritage. ENPs influence organisms at the biochemical and cellular levels. They also affect organisms at tissue, organismal and community levels, with various outcomes (Table 1).
Metal oxide NPs, including Ti, Zn [80] and Ag [81], have been shown to alter enzymatic activities and bacterial community structure in soils, especially when these are alkaline. In plants, Ag-based ENPs have been shown to exert negative effects at low doses, causing chromosomal aberrations [69]. These ENPs are also active against natural enemies of the mosquito, suggesting a potential public health problem through disruption of biological control of mosquito populations [64]. There is evidence that CuO-based ENPs are toxic to the reproduction of Enchytraeus crypticus (earthworm) and this may alter soil processes, since earthworms are key players in soil health [75]. Soil processes may also be altered via the nitrogen cycle in plants when impacted by TiO2, as well as by Ag-based and CuO ENPs.
Kim et al. [82] found reduced levels of dehydrogenase, phosphatase, and β-glucosidases in soil mesocosms treated with CuO NPs. Similarly, ZnO NPs have been found to negatively affect microbial diversity in soils, reducing respiration and ammonification in periods as short as 30 days after exposure [83]. Not only microbial diversity, but also community composition, could be affected in soil impacted by ENPs released from treated surfaces. Doolette et al. [84] studied the influence of Ag NPs on soil microbial populations. They found a differential response of the community with some phyla (Acidobacteria, Actinobacteria, Cyanobacteria and Nitrospirae) being significantly decreased as Ag NPs concentration increased. Conversely, Proteobacteria and Planctomycetes increased and became dominant. TiO2 NPs, often stated to have low environmental impact, were found to reduce ammonia-oxidizing Archaea in soil systems at very low concentrations [85]. Even small apparent changes in microbial composition of a soil can have significant impact on function. These studies are in line with a recent review [86] about the impact of ENPs on soil microbial communities. The authors reported that contrasting responses can be observed according to the type of NP, the concentration, the exposure time, and other factors. They advocate for the study of effects of realistic concentrations of nanoparticles and the identification of soil variables controlling the bioavailability and toxicity of nanoparticles is order to provide a better environmental risk assessment. It is on good scientific grounds that it is stated that ENPs may pose an environmental concern, and should be the subject of proper assessment, as in the case of biocides [87]. A suggested standard method to determine ENP release during outdoor weathering of treated surfaces was published by Lankone et al. [88] and it is important that an official standard be developed, tested, approved and published to enable future production and use of acceptable NPs.
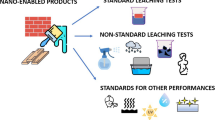
Unfortunately, there are, as yet, no standard tests for the ecotoxicity of ENPs, although the International Standards Organization test, ISO 11348-3 [89], using luminescence inhibition of the marine Gram-negative bacterium Vibrio fischeri strain NRRL B-11177, has been employed [90]. There is much discussion within the European Union’s Biocidal Products Regulation group about suitable tests for ENPs [91]. Holden et al. [92] recommended the development of standard ecotoxicity tests using bacteria, and Chifiriuc et al. [93] suggested that Drosophila should be used in a standard test, mainly because of its low maintenance cost and lack of ethical objections.
More recently, Holden et al. [94] have reviewed the various test methods present in the literature and suggested that the employment of more realistic conditions would improve the methodology. Hjorth et al. [95] insisted that OECD (Organization for Economic Co-operation and Development) guidelines must be published to allow controlled regulation of ENP release into the environment. OECD draft test guidelines for dissolution and agglomeration of NPs exist, but require modification and extension to include methods to estimate heteroagglomeration, the process by which NPs are adsorbed on cells, (contrasting with homoagglomeration which refers to NPs clustering with other NPs) and the tendency for NPs to transform in the environment [96, 97]. Such tests are essential for the future safe development of ENP technology.
Ecological effects of NPs in the aqueous environment
Not only soil, but also water, is affected by run-off from NP-treated surfaces. Griffitt et al. [98] used zebrafish, daphnids, and an algal species to determine the toxic effects of Ag, Cu, Al, Ni, and Co, both as NPs and as their soluble salts. They compared the results to those for TiO2 NPs. Ag and Cu in nano form were toxic in all the organisms tested, while TiO2 NPs were completely negative. Filter-feeding invertebrates were more susceptible to nanometals than zebrafish and nanometals were less toxic than their soluble forms. Vicario-Pares et al. [99] showed that the health of zebrafish was impaired by both ionic Ag and Ag NPs, but that the effects on liver transcriptome regulation were different. Aruoja et al. [100] showed that ZnO NPs were more toxic to the microalga Pseudokirchneriella subcapitata than CuO NPs. The toxicity was attributed mainly to bioavailable metal ions released into the water. They agreed, therefore, with the conclusions reached by Griffitt et al. [98], and Ivask et al. [101], and compared the effects of several NPs in their original and dissolved forms, adding to the debate.
Zhang et al. [102], emphasizing the importance of testing NPs toxicity in natural waters, rather than artificial ones, also confirmed that ZnO and Ag NP ecotoxicity was mostly due to release of the toxic ions.
Transport of NPs in the aquatic environment can be affected by aggregation, dissolution, and/or transformation, and these, in turn, depend on the size and shape of the particles, the composition and concentration of electrolytes in the environment, and biogeochemical and hydrodynamic conditions [103,104,105]. The presence of extracellular polymeric substances (EPS) from aquatic microorganisms can affect ENP stability; the dissolution of CuO NPs was increased by EPS [106], impacting on their ultimate permanency in the environment. Modelling and predicting the environmental fate and distribution of NPs is, therefore, challenging.
Hegde et al. [107] reviewed and discussed the environmental problems associated with ENPs and Mahaye et al. [108] reviewed their genotoxicity in aquatic organisms. Minetto et al. [109], whilst criticizing the lack of research on ecotoxicity of ENPs and particularly mixed NP formulations, stated that research up to 2016 suggested that the toxicity effects of NPs in the marine environment were, in decreasing order, Au > ZnO > Ag > CuO > TiO2 > C60. However, the frequently reported influence of environmental factors like salinity, pH and presence of pollutants on ENPs toxicity means that this may not be applicable in all situations. The biological components of the environment must also be considered; in the presence of algae, for example, Ag NP toxicity for Daphnia is reduced [110].
While ENPs released into soil and water are obvious concerns, John et al. [111] pointed out that air contamination can also occur accidentally or, especially, when sprays are used. This is less common than soil and water pollution but should be further studied. The only publication found that examines interactions between NPs and airborne particles is that of Baysal et al. [112].
NP interactions with chemical pollutants
Only recently has the issue of reaction between released NPs and other pollutants in the environment been raised. The presence of existing contaminants may decrease bioavailability of the active metal ions by complex formation, aggregation, or absorption, thus reducing environmental toxicity; on the other hand, they may have synergistic effects, increasing the ecotoxicological risk [113, 114]. Deng et al. [115] pointed out that NPs released into water bodies may react with organic contaminants, metal/metalloid ions, dissolved organic matter, inorganic ligands and other NPs, as well as influencing the bioaccumulation of organic contaminants and heavy metal ions. Zhang et al. [116] reported that TiO2 NPs had a synergistic inhibitory effect with atrazine against the alga Chlorella pyrenoidosa, an additive effect with pentachlorobenzene, and an antagonistic effect for hexachlorobenzene and 3,3′,4,4′-tetrachlorobiphenyl. Okupnik et al. [117] showed that cyanobacterial toxins could adsorb to TiO2 NPs, affecting the transport and transformation of both materials, while the antibiotic tetracycline has been shown to stabilize TiO2 NPs in aquatic systems [118].
Many unknown reactions can take place once ENPs are released into the (often unstudied) environment. For instance, microbial extracellular polymeric substances (EPS) can react in various ways with ENPs [119], modifying their surfaces and hence their fate in the surroundings and their potential toxic effects. This is an area that requires further research if we are to understand and predict the levels of toxicity exerted by different ENPs in the natural environment.
Summary and perspectives
Nanoparticles (NPs) are becoming more widely used for the protection of Cultural Heritage buildings, but they are a long way from being routine treatments. Probably the major barrier to this is the fact that treatments have to be tailored not only to the particular substrate, but also to the surrounding environment. Even the changes in climatic conditions between the seasons can alter the immediate result and the longevity of NP coatings. Given the relative cost, compared to current and traditional conservation treatments, it seems likely that more development will be necessary to improve NP coating efficacy before they become accepted routinely by conservators and restorers.
The fate, transport and transformation of NPs in different environments are still little understood. Complex interactions mean that the exact physical, chemical and biological make-up of an environment will have a tremendous effect on the final ecotoxic effects of released ENPs. Hence predictive modeling is problematic.
There is some information on ways in which NPs can be modified in order to reduce their release, or reduce their environmental effects once released; however, this research is in its infancy. In view of the projected increase in use of NPs in the future, this should be one of the foci of current research.
In order to enable direct comparison of newly produced and already available ENPs, standard tests must be used. One of the most important conclusions from this brief review is the necessity for the development of suitable standard tests, not only for assessing antimicrobial efficacy of NPs, but also for determining their ecotoxicological effects. This is a readily achievable goal and will facilitate basic research on testing and production of more environmentally acceptable ENPs products. It is also necessary, as suggested by Selck et al. [120], to re-direct the study in various environments to (1) ENP characterization and quantification in environmental and biological matrices; (2) ENP transformation in the environment and consequences for bioavailability and toxicity; (3) alternative methods to assess exposure and monitoring; (4) influence of exposure scenarios on bioavailability and toxicity; and (5) uptake, internal distribution, and depuration of ENPs.
References
Grillo R, Rosa AH, Fraceto LF. Engineered nanoparticles and organic matter: a review of the state-of-the-art. Chemosphere. 2015;119:608–19. https://doi.org/10.1016/j.chemosphere.2014.07.049.
Baglioni P, Carretti E, Chelazzi D. Nanomaterials in art conservation. Nat Nanotechnol. 2015. https://doi.org/10.1038/nnano.2015.38.
Munafò P, Goffredo GB, Quagliarini E. TiO2-based nanocoatings for preserving architectural stone surfaces: an overview. Constr Build Mater. 2015. https://doi.org/10.1016/j.conbuildmat.2015.02.083.
Sierra-Fernandez A, De la Rosa-García SC, Gomez-Villalba LS, Gómez-Cornelio S, Rabanal ME, Fort R, et al. Synthesis, photocatalytic, and antifungal properties of MgO, ZnO and Zn/Mg oxide nanoparticles for the protection of calcareous stone heritage. ACS Appl Mater Interfaces. 2017. https://doi.org/10.1021/acsami.7b06130.
La Russa MF, Macchia A, Ruffolo SA, De Leo F, Barberio M, Barone P, et al. Testing the antibacterial activity of doped TiO2 for preventing biodeterioration of cultural heritage building materials. Int Biodeterior Biodegrad. 2014. https://doi.org/10.1016/j.ibiod.2014.10.002.
Benedix R, Dehn F, Quaas J, Orgass M. Application of titanium dioxide photocatalysis to create self-cleaning building materials. Lacer. 2000;5:158–68.
Parkin IP, Palgrave RG. Self-cleaning coatings. J Mater Chem. 2005;15:1689–95.
Giannantonio DJ, Kurth JC, Kurtis KE, Sobecky PA. Effects of concrete properties and nutrients on fungal colonization and fouling. Int Biodeterior Biodegrad. 2009. https://doi.org/10.1016/j.ibiod.2008.10.002.
Fonseca AJ, Pina F, Macedo MF, Leal N, Romanowska-Deskins A, Laiz L, et al. Anatase as an alternative application for preventing biodeterioration of mortars: evaluation and comparison with other biocides. Int Biodeterior Biodegrad. 2010. https://doi.org/10.1016/j.ibiod.2010.04.006.
Baneriee S, Dionysiou DD, Pillai SC. Self-cleaning applications of TiO2 by photo-induced hydrophilicity and photocatalysis. Appl Catal B. 2015;176–177:396–428. https://doi.org/10.1016/j.apcatb.2015.03.058.
Pino F, Fermo P, La Russa M, Ruffolo S, Comite V, Baghdachi J, et al. Advanced mortar coating for cultural heritage protection. Durability towards prolonged UV and outdoor exposure. Environ Sci Pollut Res. 2016. https://doi.org/10.1007/s11356-016-7611-3.
Becerra J, Zaderenko AP, Sayagués MJ, Ortiz R, Ortiz P. Synergy achieved in silver-TiO2 nanocomposites for the inhibition of biofouling on limestone. Build Environ. 2018;141:80–90. https://doi.org/10.1016/j.buildenv.2018.05.020.
Graziani L, Quagliarini E, D’Orazio M. The role of roughness and porosity on the self-cleaning and anti-biofouling efficiency of TiO2–Cu and TiO2–Ag nanocoatings applied on fired bricks. Constr Build Mater. 2016;129:116–24.
Pinna D, Galeotti M, Perito B, Daly G, Salvadori B. In situ long-term monitoring of recolonization by fungi and lichens after innovative and traditional conservative treatments of archaeological stones in Fiesole (Italy). Int Biodeterior Biodegrad. 2018;132:49–58.
Pinna D, Salvadori B, Galeotti M. Monitoring the performance of innovative and traditional biocides mixed with consolidants and water-repellents for the prevention of biological growth on stone. Sci Total Environ. 2012;423:132–41.
Banach M, Szczyglowska R, Pulit J, Bryk M. Building materials with antifungal efficacy enriched with silver nanoparticles. Chem Sci J. 2014;5:1. https://doi.org/10.4172/2150-3494.1000085.
Battista Goffredo G, Accoroni S, Totti C, Romagnoli T, Valentini L, Manufò P. Titanium dioxide based nanotreatments to inhibit microalgal fouling on building stone surfaces. Build Environ. 2017;112:209–22.
Scarfato P, Avallone E, Incarnato L, Di Maio L. Development and evaluation of halloysite nanotube-based carrier for biocide activity in construction materials protection. Appl Clay Sci. 2016;132–133:336–42.
D’Orazio L, Grippo A. A water dispersed Titanium dioxide/poly(carbonate urethane) nanocomposite for protecting cultural heritage: preparation and properties. Prog Org Coat. 2015;79:1–7. https://doi.org/10.1016/j.porgcoat.2014.09.017.
Toniolo L, Gherardi F. The protection of marble surfaces: the challenge to develop suitable nanostructured treatments. In: Hosseini M, Karapanagiotis I, editors. Advanced materials for the conservation of stone. Cham: Springer; 2018. p. 57–78.
Ruffolo SA, Ricca M, Macchia A, La Russa MF. Antifouling coatings for underwater archaeological stone materials. Prog Org Coat. 2017. https://doi.org/10.1016/j.porgcoat.2016.12.004.
Ruffolo SA, Ricca M, De Leo F, La Russa MF. Medium-term in situ experiment using organic biocides and titanium dioxide for the mitigation of microbial colonization on stone surfaces. Int Biodeterior Biodegrad. 2017. https://doi.org/10.1016/j.ibiod.2017.05.016.
Goffredo GB, Terlizzi V, Munafò P. Multifunctional TiO2-based hybrid coatings on limestone: initial performances and durability over time. J Build Eng. 2017. https://doi.org/10.1016/j.jobe.2017.10.006.
Quagliarini E, Graziani L, Diso D, Licciulli A, D’Orazio M. Is nano-TiO2 alone an effective strategy for the maintenance of stones in cultural heritage? J Cult Herit. 2017. https://doi.org/10.1016/j.culher.2017.09.016.
Nowack B, Bornhöft N, Ding Y, Riediker M, Sánchez-Jiménez A, Sun T, et al. The flows of engineered nanomaterials from production, use, and disposal to the environment. In: Viana M, editor. Indoor and outdoor nanoparticles. The handbook of environmental chemistry, vol. 48. Cham: Springer; 2015. p. 209–31.
Ortega-Morales BO, Reyes-Estebanez M, Gaylarde CC, Camacho-Chab JC, Sanmartín P, Chan-Bacab M, Granados-Echegoyen C, Pereañez-Sacarias JE. Antimicrobial properties of nanomaterials used to control microbial colonization of stone substrata. In: Hosseini M, Karapanagiotis I, editors. Advanced materials for the conservation of stone, volume 1, chapter 13. Cham: Springer; 2018. p. 277–98.
Sierra-Fernandez A, Gomez-Villalba LS, Rabanal ME, Fort R. New nanomaterials for applications in conservation and restoration of stony materials: a review. Mater Constr. 2017. https://doi.org/10.3989/mc.2017.07616.
Zornoza-Indart A, López-Arce P. Silica nanoparticles (SiO2): influence of relative humidity in stone consolidation. J Cult Herit. 2016. https://doi.org/10.1016/j.culher.2015.06.002.
Zarzuela R, Carbú M, Gil MLA, Cantoral JM, Mosquera MJ. CuO/SiO2 nanocomposites: a multifunctional coating for application on building stone. Mater Des. 2017. https://doi.org/10.1016/j.matdes.2016.11.009.
Ruggiero L, Crociani L, Zendri E, El Habra N, Guerriero P. Incorporation of the zosteric sodium salt in silica nanocapsules: synthesis and characterization of new fillers for antifouling coatings. Appl Surf Sci. 2018;439:705–11. https://doi.org/10.1016/j.apsusc.2017.12.228.
Eduok S, Coulon F. Engineered nanoparticles in the environments: interactions with microbial systems and microbial activity. In: Cravo-Laureau C, Cagnon C, Lauga B, Duran R, editors. Microbial ecotoxicology, Cham. Springer: Cham; 2017. p. 63–107.
Keller AA, McFerran S, Lazareva A, Suh S. Global life cycle releases of engineered nanomaterials. J Nanopart Res. 2013;15:1692.
Hristozov D, Malsch I. Hazard and risk of engineered nanoparticles for the environment and human health. Sustainability. 2009;1(4):1161–94. https://doi.org/10.3390/su1041161.
Gladis G, Eggert A, Karsten U, Schumann R. Prevention of biofilm growth on man-made surfaces: evaluation of antialgal activity of two biocides and photocatalytic nanoparticles. Biofouling. 2010. https://doi.org/10.1080/08927010903278184.
Ferrari AM, Pini M, Neri P, Bondioli F. Nano-TiO2 coatings for limestone: which sustainability for cultural heritage? Coatings. 2015. https://doi.org/10.3390/coatings5030232.
González-Gálvez D, Janer G, Vilar G, Vílchez A, Vázquez-Campos S. The life cycle of engineered nanoparticles. In: Tran L, Bañares M, Rallo R, editors. Modelling the toxicity of nanoparticles. Advances in experimental medicine and biology, vol. 947. Cham: Springer; 2017. p. 41–69.
Kaegi R, Sinnet B, Zuleeg S, Hagendorfer H, Mueller E, Vonbank R, et al. Release of silver nanoparticles from outdoor facades. Environ Pollut. 2010. https://doi.org/10.1016/j.envpol.2010.06.009.
Carmona ER, Inostroza-Blancheteau C, Obando V, Rubio L, Marcos R. Genotoxicity of copper oxide nanoparticles in Drosophila melanogaster. Mutat Res. 2015. https://doi.org/10.1016/j.mrgentox.2015.07.006.
Kegler P, Kegler HF, Gärdes A, Ferse SCA, Lukman M, Alfiansah YR, Hassenrück C, Kunzman A. Bacterial biofilm communities and coral larvae settlement at different levels of anthropogenic impact in the Spermonde Archipelago, Indonesia. Front Mar Sci. 2017. https://doi.org/10.3389/fmars.2017.00270.
Chen J, Tang Y, Li Y, Nie Y, Hou L, Li X, et al. Impacts of different nanoparticles on functional bacterial community in activated sludge. Chemosphere. 2014. https://doi.org/10.1016/j.chemosphere.2013.10.082.
Chai H, Yao J, Sun J, Zhang C, Liu W, Zhu M, et al. The effect of metal oxide nanoparticles on functional bacteria and metabolic profiles in agricultural. Bull Environ Contam Toxicol. 2015. https://doi.org/10.1007/s00128-015-1485-9.
Kaegi R, Ulrich A, Sinnet B, Vonbank R, Wichser A, Zuleeg S, et al. Synthetic TiO2 nanoparticle emission from exterior facades into the aquatic environment. Environ Pollut. 2008. https://doi.org/10.1016/j.envpol.2008.08.004.
Levard C, Hotze EM, Lowry GV, Brown GE Jr. Environmental transformations of silver nanoparticles: impact on stability and toxicity. Environ Sci Technol. 2012;46:6900–14.
Bondarenko O, Juganson K, Ivask A, Kasemets K, Mortimer M, Kahru A. Toxicity of Ag, CuO and ZnO nanoparticles to selected environmentally relevant test organisms and mammalian cells in vitro: a critical review. Arch Toxicol. 2013. https://doi.org/10.1007/s00204-013-1079-4.
Giese B, Klaessig F, Park B, Kaegi R, Steinfeldt M, Wigger H, von Gleich A, Gottschalk F. Risks, release and concentrations of engineered nanomaterial in the environment. Sci Rep. 2018;8:1565. https://doi.org/10.1038/s41598-018-19275-4.
Chen K, Zhou S, Yang S, Wu L. Fabrication of all-water-based self-repairing superhydrophobic coatings based on UV-responsive microcapsules. Adv Funct Mater. 2015;25:1035–41. https://doi.org/10.1002/adfm.201403496.
Alves DCB, Silva R, Voiry D, Asefa T, Chhowalla M. Copper nanoparticles stabilized by reduced graphene oxide for CO2 reduction reaction. Mater Renew Sustain Energy. 2015;4:2. https://doi.org/10.1007/s40243-015-0042-0.
Ji H, Sun H, Qu X. Antibacterial applications of graphene-based nanomaterials: recent achievements and challenges. Adv Drug Deliv Rev. 2016;105(Part B):176–89. https://doi.org/10.1016/j.addr.2016.04.009.
Whitehead KA, Vaidya M, Liauw CM, Brownson DAC, Ramalingam P, Kamieniak J, Rowley-Neale SJ, Tetlow LA, Wilson-Nieuwenhuis JST, Brown D, McBain AJ, Kulandaivel J, Banks CE. Antimicrobial activity of graphene oxide-metal hybrids. Int Biodeterior Biodegrad. 2017;123:182–90. https://doi.org/10.1016/j.ibiod.2017.06.020.
Slate AJ, Karaky N, Whitehead KA. Antimicrobial properties of modified graphene and other advanced 2D material coated surfaces, Chap. 5. In: Banks C, Brownson D, editors. 2D Materials. Boca Raton: CRC Press; 2018.
Lvov Y, Wang W, Zhang L, Fakhrullin R. Halloysite clay nanotubes for loading and sustained release of functional compounds. Adv Mater. 2015;28:1227–50.
Jana S, Kondakova AV, Shevchenko SN, Sheval EV, Gonchar KA, Timoshenko VY, Vasiliev AN. Halloysite nanotubes with immobilized silver nanoparticles for anti-bacterial application. Colloids Surf B. 2017;151:249–54. https://doi.org/10.1016/j.colsurfb.2016.12.017.
Shu Z, Zhang Y, Ouyang J, Yang H. Characterization and synergetic antibacterial properties of ZnO and CeO2 supported by halloysite. Appl Surf Sci. 2017;420:833–8. https://doi.org/10.1016/j.apsusc.2017.05.219.
Loureiro S, Tourinho PS, Cornelis G, VanDenBrink NW, Díez-Ortiz M, Vázquez-Campos S, et al. Nanomaterials as soil pollutants. In: Duarte AC, Cachada A, Rocha-Santos TAP, editors. Soil pollution. From monitoring to remediation; 2018. p. 161–190.
Ma R, Levard C, Judy JD, Unrine JM, Durenkamp M, Martin B, et al. Fate of zinc oxide and silver nanoparticles in a pilot wstewater treatment plant and in processed biosolids. Environ Sci Technol. 2014;48:104–12.
Lahive E, Matzke M, Durenkamp M, Lawlor AJ, Thacker SA, Pereira MG, et al. Sewage sludge treated with metal nanomaterials inhibits earthworm reproduction more strongly than sludge treated with metal metals in bulk/salt forms. Environ Sci Nano. 2017;4:78–88.
Coutris C, Joner EJ, Oughton DH. Aging and soil organic matter content affect the fate of silver nanoparticles in soil. Sci Total Environ. 2012. https://doi.org/10.1016/j.scitotenv.2012.01.027.
Pachapur VL, Larios AD, Cledón M, Brar SK, Verma M, Surampalli RY. Behavior and characterization of titanium dioxide and silver nanoparticles in soils. Sci Total Environ. 2016. https://doi.org/10.1016/j.scitotenv.2012.01.027.
Philbrook NA, Winn LM, Nabiul-Afrooz ARM, Saleh NB, Walker VK. The effect of TiO(2) and Ag nanoparticles on reproduction and development of Drosophila melanogaster and CD-1 mice. Toxicol Appl Pharmacol. 2011. https://doi.org/10.1016/j.taap.2011.09.027.
Kim E, Kim SH, Kim HC, Lee SG, Lee SJ, Jeong SW. Growth inhibition of aquatic plant caused by silver and titanium oxide nanoparticles. Toxicol Environ Health Sci. 2011;3:1–6.
Song U, Jun H, Waldman B, Roh J, Kim Y, Yi J, et al. Functional analyses of nanoparticle toxicity: a comparative study of the effects of TiO2 and Ag on tomatoes (Lycopersicon esculentum). Ecotoxicol Environ Saf. 2013. https://doi.org/10.1016/j.ecoenv.2013.03.033.
Zheng L, Hong F, Lu S, Liu C. Effect of nano-TiO(2) on strength of naturally aged seeds and growth of spinach. Biol Trace Elem Res. 2005. https://doi.org/10.1385/BTER:104:1:083.
Balakrishnan S, Srinivasan M, Mohanraj J. Biosynthesis of silver nanoparticles from mangrove plant (Avicennia marina) extract and their potential mosquito larvicidal property. J Parasit Dis. 2016. https://doi.org/10.1007/s12639-014-0621-5.
Kumar PM, Murugan K, Madhiyazhagan P, Kovendan K, Amerasan D, Chandramohan B, et al. Biosynthesis, characterization, and acute toxicity of Berberis tinctoria-fabricated silver nanoparticles against the Asian tiger mosquito, Aedes albopictus, and the mosquito predators Toxorhynchites splendens and Mesocyclops thermocyclopoide. Parasitol Res. 2016. https://doi.org/10.1007/s00436-015-4799-y.
Ramkumar G, Karthi S, Suganya R, Shivakumar MS. Evaluation of silver nanoparticle toxicity of Coleus aromaticus leaf extracts and its larvicidal toxicity against dengue and filariasis vectors. Bionanoscience. 2016;6:308–15.
Velu K, Elumalai D, Hemalatha P, Janaki A, Babu M, Hemavathi M, et al. Evaluation of silver nanoparticles toxicity of Arachis hypogaea peel extracts and its larvicidal activity against malaria and dengue vectors. Environ Sci Pollut Res. 2015. https://doi.org/10.1007/s11356-015-4919-3.
Velicogna JR, Schwertfeger DM, Jesmer AH, Scroggins RP, Princz JI. The bioaccumulation of silver in Eisenia andrei exposed to silver nanoparticles and silver nitrate in soil. Nanoimpact. 2017. https://doi.org/10.1016/j.impact.2017.03.001.be.
Diez-Ortiz M, Lahive E, George S, Schure AT, van Gestel CAM, Jurkschat K, et al. Short-term soil bioassays may not reveal the full toxicity potential for nanomaterials; bioavailability and toxicity of silver ions (AgNO3) and silver nanoparticles to earthworm Eisenia foetida in long-term aged soils. Environ Pollut. 2015. https://doi.org/10.1016/j.envpol.2015.03.033.
Saha N, Dutta Gupta S. Low-dose toxicity of biogenic silver nanoparticles fabricated by Swertia chirata on root tips and flower buds of Allium cepa. J Hazard Mater. 2017. https://doi.org/10.1016/j.jhazmat.2017.01.021.
Shams G, Ranjbar MR, Amiri A. Effect of silver nanoparticles on concentration of silver heavy element and growth indexes in cucumber (Cucumis sativus L. negeen). J Nanopart Res. 2013;15:1630.
Kaveh R, Li YS, Ranjbar S, Tehrani R, Brueck CL, Van Aken B. Changes in Arabidopsis thaliana gene expression in response to silver nanoparticles and silver ions. Environ Sci Technol. 2013;47:10637–44.
Mirzajani F, Askari H, Hamzelou S, Schober Y, Römpp A, Ghassempour A, Spengler B. Proteomics study of silver nanoparticles toxicity on Oryza sativa L. Ecotoxicol Environ Saf. 2014. https://doi.org/10.1016/j.ecoenv.2014.07.013.
Cvjetko P, Milošić A, Domijan AM, Vinković I, Peharec Tolić S, Štefanić P, et al. Toxicity of silver ions and differently coated silver nanoparticles in Allium cepa roots. Ecotoxicol Environ Saf. 2017. https://doi.org/10.1016/j.ecoenv.2016.11.009.
Gomes SIL, Murphy M, Nielsen MT, Kristiansen SM, Amorim MJB, Scott-Fordsmand JJ. Cu-nanoparticles ecotoxicity—explored and explained? Chemosphere. 2015. https://doi.org/10.1016/j.chemosphere.2015.06.045.
Gopalakrishnan Nair PM, Kim SH, Chung IM. Copper oxide nanoparticle toxicity in mung bean (Vigna radiata L.) seedlings: physiological and molecular level responses of in vitro grown plants. Acta Physiol Plant. 2014. https://doi.org/10.1007/s11738-014-1667-9.
Liu R, Zhang H, Lal R. Effects of stabilized nanoparticles of copper, zinc, manganese, and iron oxides in low concentrations on lettuce (Lactuca sativa) seed germination: nanotoxicants or nanonutrients? Water Air Soil Pollut. 2016. https://doi.org/10.1007/s11270-015-2738-2.
Murugan K, Roni M, Panneerselvam C, Aziz AT, Suresh U, Rajaganesh R, et al. Sargassum wightii-synthesized ZnO nanoparticles reduce the fitness and reproduction of the malaria vector Anopheles stephensi and cotton bollworm Helicoverpa armigera. Physiol Mol Plant Pathol. 2017. https://doi.org/10.1016/j.pmpp.2017.02.004.
Hu CW, Li M, Cui YB, Li DS, Chen J, Yang LY. Toxicological effects of TiO2 and ZnO nanoparticles in soil on earthworm Eisenia fetida. Soil Biol Biochem. 2010. https://doi.org/10.1016/j.soilbio.2009.12.007.
Khare P, Sonane M, Pandey R, Ali S, Gupta KC, Satish A. Adverse effects of TiO2 and ZnO nanoparticles in soil nematode, Caenorhabditis elegans. J Biomed Nanotechnol. 2011;7:116–7.
You T, Liu D, Chen J, Yang Z, Dou R, Gao X, et al. Effects of metal oxide nanoparticles on soil enzyme activities and bacterial communities in two different soil types. J Soils Sediments. 2017. https://doi.org/10.1007/s11368-017-1716-2.
Asadishad B, Chahal S, Akbari A, Cianciarelli V, Azodi M, Ghoshal S, et al. Amendment of agricultural soil with metal nanoparticles: effects on soil enzyme activity and microbial community composition. Environ Sci Technol. 2018. https://doi.org/10.1021/acs.est.7b05389.
Kim S, Sin H, Lee S, Lee I. Influence of metal oxide particles on soil enzyme activity and bioaccumulation of two plants. J Microbiol Biotechnol. 2013;23:1279–86.
Shen Z, Chen Z, Hou Z, Li T, Lu X. Ecotoxicological effect of zinc oxide nanoparticles on soil microorganisms. Front Environ Sci Eng. 2015;9:912–8.
Doolette CL, Gupta VVSR, Lu Y, Payne JL, Batstone DJ, Kirby JK, et al. Quantifying the sensitivity of soil microbial communities to silver sulfide nanoparticles using metagenome sequencing. PLoS ONE. 2016. https://doi.org/10.1371/journal.pone.0161979.
Simonin M, Martins JMF, LeRoux X, Uzu G, Calas A, Richaume A. Toxicity of TiO2 nanoparticles on soil nitrification at environmentally relevant concentrations: lack of classical dose response relationships. Nanotoxicology. 2017. https://doi.org/10.1080/17435390.2017.1290845.
Simonin M, Richaume A. Impacto f engineered nanoparticles on the activity, abundance, and diversity of soil microbial communities: a review. Environ Sci Pollut Res. 2015;22(18):13710–23. https://doi.org/10.1007/s11356-015-4171-x.
Pinna D. Coping with biological growth on stone heritage objects: methods, products, applications, and perspectives. 1st ed. Boca Raton: CRC Press Taylor & Francis Group; 2017.
Lankone RS, Challis KE, Bi Y, Hanigan D, Reed RB, Zaikova T, et al. Methodology for quantifying engineered nanomaterial release from diverse product matrices under outdoor weathering conditions and implications for life cycle assessment. Environ Sci Nano. 2017. https://doi.org/10.1039/C7EN00410A.
ISO/IEC IS 1348-3:2007. Water quality—determination of the inhibitory effect of water samples on the light emission of vibrio fischeri (luminescent bacteria test)—part 3: method using freeze-dried bacteria. Geneva: International Organization for Standardization. 2007.
Nuzzo A, Hosseinkhani B, Boon N, Zanaroli G, Fava F. Impact of bio-palladium nanoparticles (bio-Pd NPs) on the activity and structure of a marine microbial community. Environ Pollut. 2017. https://doi.org/10.1016/j.envpol.2016.11.036.
Brinch A, Hansen SF, Hartmann NB, Baun A. EU regulation of nanobiocides: challenges in implementing the biocidal product regulation (BPR). Nanomater. 2016. https://doi.org/10.3390/nano6020033.
Holden PA, Schimel JP, Godwin HA. Five reasons to use bacteria when assessing manufactured nanomaterial environmental hazards and fates. Curr Opin Biotechnol. 2014. https://doi.org/10.1016/j.copbio.2013.11.008.
Chifiriuc MC, Ratiu AC, Popa M, Ecovoiu AA. Drosophotoxicology: an emerging research area for assessing nanoparticles interaction with living organisms. Int J Mol Sci. 2016. https://doi.org/10.3390/ijms17020036.
Holden PA, Gardea-Torresdey JL, Klaessig F, Turco RF, Mortimer M, Hund-Rinke K, et al. Considerations of environmentally relevant test conditions for improved evaluation of ecological hazards of engineered nanomaterials. Environ Sci Technol. 2016. https://doi.org/10.1021/acs.est.6b00608.
Hjorth R, Skjolding LM, Sørensen SN, Baun A. Regulatory adequacy of aquatic ecotoxicity testing of manufactured nanomaterials. Nanoimpact. 2017. https://doi.org/10.1016/j.impact.2017.07.003.
Baun A, Sayre P, Steinhäuser KG, Rose J. Regulatory relevant and reliable methods and data for determining the environmental fate of manufactured nanomaterials. Nanoimpact. 2017. https://doi.org/10.1016/j.impact.2017.06.00.
You T, Liu D, Chen J, Yang Z, Dou R, Gao X, Wang L. Effects of metal oxide nanoparticles on soil enzyme activities and bacterial communities in two different soil types. J Soils Sediments. 2018;18:211. https://doi.org/10.1007/s11368-017-1716-2.
Griffitt RJ, Luo J, Gao J, Bonzongo JC, Barber DS. Effects of particle composition and species on toxicity of metallic nanomaterials in aquatic organisms. Environ Toxicol Chem. 2008. https://doi.org/10.1897/08-002.1.
Vicario-Parés U, Lacave JM, Reip P, Cajaraville MP, Orbea A. Cellular and molecular responses of adult zebrafish after exposure to CuO nanoparticles or ionic copper. Ecotoxicology. 2018. https://doi.org/10.1007/s10646-017-1873-5.
Aruoja V, Dubourguier HC, Kasemets K, Kahru A. Toxicity of nanoparticles of CuO, ZnO and TiO2 to microalgae Pseudokirchneriella subcapitata. Sci Total Environ. 2009. https://doi.org/10.1016/j.scitotenv.2008.10.053.
Ivask A, George S, Bondarenko O, Kahru A. Metal-containing nano-antimicrobials: differentiating the impact of solubilized metals and particles. In: Cioffi N, Rai M, editors. Nano-antimicrobials: progress and prospects. Berlin: Springer; 2012. p. 253–90.
Zhang L, Li J, Yang K, Liu J, Lin D. Physicochemical transformation and algal toxicity of engineered nanoparticles in surface water samples. Environ Pollut. 2016. https://doi.org/10.1016/j.envpol.2015.12.041.
Hammes J, Gallego-Urrea JA, Hasseliöv M. Geographically distributed classification of surface water chemical parameters influencing fate and behavior of nanoparticles and colloid facilitated contaminant transport. Water Res. 2013. https://doi.org/10.1016/j.watres.2013.06.015.
Peng C, Zhang W, Gao H, Li Y, Tong X, Li K, et al. Behavior and potential impacts of metal-based engineered nanoparticles in aquatic environments. Nanomaterials. 2017. https://doi.org/10.3390/nano7010021.
Pulido-Reyes G, Leganes F, Fernández-Piñas F, Rosal R. Bio-nano interface and environment: a critical review. Environ Toxicol Chem. 2017. https://doi.org/10.1002/etc.3924.
Adeleye AS, Conway JR, Perez T, Rutten P, Keller AA. Influence of extracellular polymeric substances on the long-term fate, dissolution, and speciation of copper-based nanoparticles. Environ Sci Technol. 2014;48:12561–8.
Hegde K, Brar SK, Verma M, Surampalli RY. Current understanding of toxicity, risk and regulations of engineered nanoparticles with respect to environmental microorganisms. Nanotechnol Environ Eng. 2016. https://doi.org/10.1007/s41204-016-0005-4.
Mahaye N, Thwala N, Cowan DA, Musee N. Genotoxicity of metal based engineered nanoparticles in aquatic organisms: a review. Mutat Res. 2017. https://doi.org/10.1016/j.mrrev.2017.05.004.
Minetto D, Ghirardini AV, Libralato G. Saltwater ecotoxicology of Ag, Au, CuO, TiO2, ZnO and C 60 engineered nanoparticles: an overview. Environ Int. 2016. https://doi.org/10.1016/j.envint.2016.03.041.
Conine AL, Frost PC. Variable toxicity of silver nanoparticles to Daphnia magna: effects of algal particles and animal nutrition. Ecotoxicology. 2017. https://doi.org/10.1007/s10646-016-1747-2.
John AC, Küpper M, Manders-Groot AMM, Debray BB, Lacome JM, Kuhlbusch TAJ. Emissions and possible environmental implication of engineered nanomaterials (ENMs) in the atmosphere. Atmosphere. 2017. https://doi.org/10.3390/atmos8050084.
Baysal A, Saygin H, Ustabasi GS. Interaction of PM2.5 airborne particulates with ZnO and TiO2 nanoparticles and their effect on bacteria. Environ Monit Assess. 2018. https://doi.org/10.1007/s10661-017-6408-2.
Pavagadhi S, Sathishkumar M, Balasubramanian R. Uptake of Ag and TiO2 nanoparticles by zebrafish embryos in the presence of other contaminant in the aquatic environment. Water Res. 2014. https://doi.org/10.1016/j.watres.2014.02.036.
Martín-de-Lucía I, Campos-Mañas MC, Agüera A, Rodea-Palomares I, Pulido-Reyes G, Leganés F, et al. Reverse Trojan-horse effect decreased wastewater toxicity in the presence of inorganic nanoparticles. Environ Sci Nano. 2017;4:1273–82.
Deng R, Lin D, Zhu L, Majumdar S, White JC, Gardea-Torresday JL, et al. Nanoparticle interactions with co-existing contaminants: joint toxicity, bioaccumulation and risk. Nanotoxicology. 2017. https://doi.org/10.1080/17435390.2017.1343404.
Zhang S, Deng R, Lin D, Wu F. Distinct toxic interactions of TiO2 nanoparticles with four coexisting organochlorine contaminants on algae. Nanotoxicology. 2017. https://doi.org/10.1080/17435390.2017.1398358.
Okupnik A, Contardo-Jara V, Pflugmacher S. Potential role of engineered nanoparticles as contaminant carriers in aquatic ecosystems: estimating sorption processes of the cyanobacterial toxin microcystin-LR by TiO2 nanoparticles. Colloids Surf A. 2015. https://doi.org/10.1016/j.colsurfa.2015.06.013.
Qi N, Wang P, Wang C, Ao Y. Effect of a typical antibiotic (tetracycline) on the aggregation of TiO2 nanoparticles in an aquatic environment. J Hazard Mater. 2018. https://doi.org/10.1016/j.jhazmat.2017.07.046.
Adeleye AS, Keller AA. Interactions between algal extracellular polymeric substances and commercial TiO2 nanoparticles in aqueous media. Environ Sci Technol. 2016. https://doi.org/10.1021/acs.est.6b03684.
Selck H, Handy RD, Fernandes TF, Klaine SJ, Petersen EJ. Nanomaterials in the aquatic environment: a European Union-United States perspective on the status of ecotoxicity testing, research priorities, and challenges ahead. Environ Toxicol Chem. 2016;35:1055–67. https://doi.org/10.1002/etc.3385.
Authors’ contributions
MRE, MCB, CGE, JCCC, and JEPS critically analyzed the literature and drafted the manuscript. BOOM conceived of the study, participated in its design, and English translation. CG read and co-drafted the paper. All authors read and approved the final manuscript.
Acknowledgements
This research was financially supported by National Council of Science and Technology (CONACYT-Mexico) (Grant No. 257449), Project CONACYT-Ciencia Básica 2015 “Influencia de tratamientos con nano y biomateriales en la colonización microbiana de roca monumental” to Benjamín Otto Ortega Morales.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Reyes-Estebanez, M., Ortega-Morales, B.O., Chan-Bacab, M. et al. Antimicrobial engineered nanoparticles in the built cultural heritage context and their ecotoxicological impact on animals and plants: a brief review. Herit Sci 6, 52 (2018). https://doi.org/10.1186/s40494-018-0219-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40494-018-0219-9