Abstract
Background
Nonalcoholic fatty liver disease (NAFLD) is highly prevalent worldwide. Experimental studies have shown that cholecystectomy (XGB) increases hepatic fat content in mice and appears associated to NAFLD in large retrospective population-based studies. The aim of this study was to prospectively assess the effects of XGB on hepatic fat content (HFC) and insulin resistance (IR) in non-obese, middle aged Hispanic subjects.
Methods
Twenty-six gallstone patients undergoing elective XGB and 16 control subjects with normal livers and gallbladders at ultrasonography were prospectively followed 24 months for changes in HFC and IR. Clinical, biochemical determinations and hepatic imaging were performed at baseline and 24 months after surgery. MRI technique quantified HFC in four hepatic segments. IR was assessed by the Homeostasis Model Assessment (HOMA-IR) index.
Results
Initial body mass index (BMI) was 25.6 ± 0.4 and 24.3 ± 1.0 in the control and XGB groups of subjects, respectively. Serum insulin level increased from 8.1 ± 0.7 to 10.0 ± 1.9 (μU/ml) 24 months after surgery in XGB patients (p < 0.05); no significant changes were detected in control individuals. Median HOMA-IR index increased from 1.31 (interquartile range, 1.01-1.68) to 2.20 (interquartile range, 1.57 - 2.60) 24 months after XGB, (p < 0.003). Median HOMA-IR index of control subjects remained unchanged at the end of the study. Serum apoB concentration increased from 61.5 ± 3.4 to 79.0 ± 7.8 (μg/ml) in XGB patients (p < 0.03). Serum apoB levels remained within normal ranges in both periods of the study in control subjects. HFC significantly increased in 2 of the 4 segments 24 months after XGB: right posterior hepatic lobe (from 5.3 ± 0.2% to 6.0 ± 0.2%, p > 0.04) and right anterior hepatic lobe (from 5.8 ± 0.2% to 6.6 ± 0.3%, p < 0.02). The average HFC of the four hepatic segments studied slightly increased from 5.4 ± 0.2 to 5.8 ± 0.3 2 years after XGB (p < 0.03). No significant changes were found in HFC in the control subjects at the end of the study.
Conclusions
Elective XGB increases HFC, HOMA-IR index and serum apoB concentration. These results support the notion that XGB is a risk factor non-alcoholic fatty liver disease and other IR – associated disease conditions.
Similar content being viewed by others
Background
Nonalcoholic fatty liver disease (NAFLD) is highly prevalent worldwide with an estimated rate of 30% among the adult population diagnosed by abdominal ultrasound [1,2,3]. South American and Mexican American Hispanics have the highest prevalence rates of NAFLD with figures ranging between 30 to 50% of the population [4]. NAFLD is frequently associated to obesity, type 2 diabetes, atherosclerosis and cholesterol gallstones [5,6,7] with these entities sharing some metabolic alterations. NAFLD has a complex bidirectional pathogenic interrelationship with the metabolic syndrome (MS). NAFLD is considered both, a metabolic manifestation of MS and in some cases, a primary determinant of insulin resistance (IR) and the metabolic abnormalities clustered in the MS [8,9,10,11,12]. The natural history and possible evolution of NAFLD varies greatly in seriousness, starting as an accumulation of fat in the hepatocyte and potentially evolving towards nonalcoholic steatohepatitis (NASH), cirrhosis and hepatocellular carcinoma [13,14,15].
The cause of the increase of triglycerides (TG) in NAFLD has not been completely unveiled. However, it is known that IR is a fundamental pathogenic factor that alter each of the processes that regulate TG concentration in the liver by increasing hepatic lipogenesis and lipolysis from adipocytes, and decreasing peripheral lipoprotein lipase activity, producing an increase of chylomicron and VLDL remnants, which are rapidly cleared by the liver, thus resulting in a net TG accumulation in the organ [16,17,18].
Cholecystectomy (XGB) is the recommended treatment for gallbladder (GB) diseases, including gallstone disease (GSD) and cholecystitis [19]. Indeed, XGB is one of the most commonly performed surgical procedures worldwide [20, 21] and is considered a low-risk surgical procedure with no major long-term health implications. However, recent evidence shows that the GB may not only be a simple reservoir that stores, concentrates and delivers bile into the intestine for lipid absorption. Through its critical role in regulating bile acid (BA) metabolism and the recent finding that GB mucosa is rich in the hormone Fibroblast Growth Factor 19 (FGF19), this organ may have a physiological role in whole body metabolic homeostasis [22, 23]. In fact, the possibility that GB ablation may have metabolic consequences has emerged [24] due to recent retrospective epidemiological studies showing that XGB may be a risk factor of NAFLD) [25, 26] and metabolic syndrome [27]. Concordantly, experimental studies in mice have shown that XGB increases basal metabolic rate, serum and hepatic triglycerides concentration and very low-density lipoprotein (VLDL) production [28, 29]. Moreover, XGB has been associated with elevated VLDL levels [30], deteriorated postprandial glycemic control [31] and weight gain [32] in humans. The underlying mechanisms of these effects remain ill defined. Since BAs are important signaling molecules in controlling lipid and carbohydrate metabolism [33,34,35] it is plausible that XGB may influence whole-body metabolic regulation through changes in BA physiology and potentially contributing to the development of IR and MS - associated conditions, particularly NAFLD [19–21].
To better define the potential role of XGB as a risk factor of NAFLD, the present study aimed to prospectively evaluate the effects of XGB on liver fat content and IR, one of the most relevant factors underlying NAFLD development [36]. To that end, we determined IR by the homeostatic model assessment index (HOMA-IR) [37] and hepatic fat by magnetic resonance imaging (MRI) [38, 39] in a series of non-obese GSD patients over a period of 24 months after elective XGB.
Methods
Patient selection
This study agreed with the ethical guidelines of Declaration of Helsinki (1975) and was approved by the Institutional Review Board for Human Studies of the Faculty of Medicine at Pontificia Universidad Católica de Chile. All participants gave informed written consent prior to participate in this survey.
We arbitrarily included 30 patients harboring asymptomatic gallstone disease subjected to elective laparoscopic XGB at the Clinical Hospital of the Pontificia Universidad Católica de Chile, between January and August 2013. We also studied a group of 20 control subjects with similar clinical and biochemical characteristics and normal GBs at abdominal ultrasound. Twenty-six cholecystectomized patients and 16 control subjects completed the study 24 months later.
All subjects were 35 to 55 years old, non-diabetic and non-obese who agreed to participate in the study. Inclusion criteria were: a) body mass index (BMI) < 28 and no change in weight of more than 3 kg in the last 6 months before entering the study; b) fasting glucose, lipid levels and liver function tests within normal values; c) absence of hepatic steatosis assessed by abdominal ultrasound; d) no use of glucose- and lipid-lowering drugs, estrogens or androgens. Exclusion criteria were: a) abnormal serum alanine aminotransferase (ALT) or aspartate aminotransferase (AST) concentrations; b) hepatic pathologies on MRI; c) alcohol consumption >20 g per day in the last 3 years; e) substance abuse; f) significant systemic diseases and g) inability to undergo MRI.
Clinical and biochemical assessments
Baseline clinical evaluation included medical history, physical exam, and height and weight measurements to calculate body mass index (BMI). Venous blood samples were obtained after 12 h of fasting. Glucose, lipids, insulin, apolipoprotein B (apoB), ALT and AST serum levels were measured at the central laboratory of Pontificia Universidad Católica de Chile Health Network. We used the homeostasis model assessment index (HOMA-IR) to define IR at fasting [20, 24]. We defined IR for non-obese middle-aged subjects when HOMA-IR values were ≥2.53 according to a population –based Chilean study [40].
Magnetic resonance imaging protocol and imaging analysis
A magnetic resonance imaging (MRI) scanner (1.5 T Philips Achieva MRI scanner, Best, The Netherlands) was used to quantitate hepatic fat content (HFC) A fat fraction map of the liver was generated based on IDEAL method [41]. Briefly, echo times were set to TE = 2.3/ 3.5/ 4.7 ms, corresponding to the optimal echo times in IDEAL algorithm for 1.5 T [42]. TRs and flip angles were chosen experimentally using fat-water-emulsion phantoms [43].
MRI data was processed with OSIRIX software (OsiriX Foundation, Geneva, Switzerland) to extract water and fat images. Afterwards, fat fraction map was extracted using image calculator tool of Image J software (NIH, Bethesda, MA, USA). In the fat fraction map, one region of interest (ROI) was set in four segments of the liver avoiding vascular structures. Four segment of the liver were analyzed: right posterior hepatic lobe (RPHL), anterior hepatic lobe (RAHL), left medium hepatic lobe (LMHL) and left lower hepatic lobe (LLHL). Mean FF was calculated for each ROI. Additionally, a mean value of the fat fraction of the liver was calculated by averaging the fat fraction values of the four ROI.
Statistical analysis
Parametric data were presented as mean ± SE of the mean and non-parametric data were expressed as medians with interquartile values. Student’s t test was used to compare parametric data. Wilcoxon test was used for non-parametric variables and paired Chi-square tests were used to compare frequencies. Prism software (Version 6, http://www.graphpad.com/) was used for statistical analysis. Differences were considered significant with P values <0.05.
Results
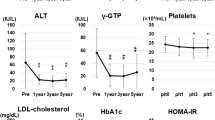
Table 1 show that age and sex distribution was similar in XGB patients and control subjects. Importantly, BMI was in the upper limit of the normal range in both groups at the beginning and end of the study. Fasting serum glucose, insulin, lipids and apoB were also equivalent and within the normal range at the beginning of the study (Table 1). Twenty-four months after XGB, serum apoB levels of control individuals remained within normal ranges at end of the study (68.1 ± 3.4 vs 63.1 ± 2.8 (μg/ml), respectively). Serum apoB concentration of cholecystectomized patients increased from 61.5 ± 3.4 to 79.0 ± 7.8 (μg/ml) in cholecystectomized patients (p < 0.03).
XGB is associated with elevated serum insulin concentration and HOMA-IR index
Serum insulin level increased from 8.1 ± 0.7 to 10.0 ± 1.9 (μU/ml) 24 month after surgery in XGB patients (p < 0.05), whereas no significant changes were detected in control individuals (Table 1). As shown in Fig. 1, median HOMA-IR index of control subjects remained unchanged, from 1.42 (interquartile range, 1.02 – 1.86) to 1.74 (interquartile range, 1.11 - 2.39) during the observational period. Contrariwise, median HOMA-IR index increased from 1.31 (interquartile range, 1.01-1.68) to 2.20 (interquartile range, 1.57 - 2.60) 24 months after XGB, (p < 0.003).We operationally defined IR when HOMA-IR was ≥2.53 for middle-age, non-obese Chilean subjects [24]. Upon this definition, 15% of individuals in both groups had IR at the beginning of the study. Twenty-four months later, 61% of the patients subjected to XGB had HOMA-IR ≥ 2.53, compared with only 12% in the control group of individuals (p < 0.001). Considered together, these results suggest that XGB determines increased circulating insulin levels and favors IR in non-obese individuals.
XGB is associated with higher hepatic fat
At the beginning of the study, all individuals of both groups had hepatic fat (HFC) lower than 8% as assessed by MRI, indicating that they had no, or mild steatosis [21,22]. Twenty-five cholecystectomized patients and 15 control subjects had a second MRI after 24 months of follow–up. Figure 2 shows that HFC of the four segments studied remained unchanged after 24 months of follow up in the control group. Contrariwise, patients subjected to XGB significantly increased HFC in the right posterior hepatic lobe (RPHL) (5.3 ± 0.2% to 6.0 ± 0.2%, p > 0.04) and in the right anterior hepatic lobe (RAHL) (5.8 ± 0.2% to 6.6 ± 0.3%, p < 0.02). The average value of HFC of the 4 hepatic segments, slightly increased 7%, from 5.4 ± 0.2% to 5.8 ± 0.20% (P < 0.05) in cholecystectomized patients. No significant change was found in the control group.
Effect of XGB on hepatic fat content. Columns represent the mean ± SE of the percentage of HFC observed at the beginning and after 24 months of follow-up in control (panel a) and cholecystectomized subjects (panel b). White and black columns represent the values obtained at the beginning and end of the study. Four liver segments were analyzed: right posterior hepatic lobe, RPHL; right anterior hepatic lobe, RAHL; left medium hepatic lobe, LMHL; left lower hepatic lobe, LLHL, and mean value of the four lobes. White columns represent values at the beginning of the study and columns with grey dots represent values obtained at the end of the study. * P < 0.03
Discussion
This prospective pilot study shows that XGB is associated with a significant increase in HFC, serum apoB and insulin levels, as well as in HOMA-IR index, in a cohort of non-obese Hispanic subjects. These findings provide support to the contention that, whereas XGB cures GSD and eliminates the risk of GB cancer, it may have relevant negative metabolic consequences [22, 24], contributing to the development, or worsening of IR and its consequences. Of note, IR is a major pathophysiological determinant of type 2 diabetes, atherosclerotic vascular diseases [44,45,46] and various cancers associated with obesity [47]. Moreover, NAFLD has become the most prevalent liver condition worldwide and has intrinsic links with IR – MS and a number of associated disease conditions, including cardiovascular diseases [9].
The mechanisms underlying the observed metabolic effects of XGB in the present study remain to be clarified. The GB could theoretically regulate whole body IR/sensitivity by direct mechanisms, through signaling factors secreted by its mucosa, or indirectly by regulating the flux mass of BAs through the enterohepatic and systemic circulation during the fast-feeding cycles. One of the GB-derived factor that play a role in this setting could be FGF19. This ileal hormone regulates BA synthesis and GB filling and is highly expressed in GB mucosa and secreted into bile [4]. FGF19 has direct systemic metabolic effects on lipid and carbohydrate metabolism [48, 49]. Of note, serum FGF19 levels decrease after XGB [50] and have been found decreased in patients with NAFLD [51, 52]. Thus, it could be hypothesized that dysregulation of FGF 19 after XGB may in part mediate the metabolic consequences of XGB observed in this study.
With regard to XGB-induced changes in BA physiology, it is generally accepted that XGB determines a decrease in the size of the BA pool [22] and increases the enterohepatic recirculation rates of BA in the fasting state [53, 54]; the BA pool recycles at least twice as often as normal after XGB [55]. BA are recognized as relevant signaling molecules [34] that may be critically involved in NAFLD development through their hepatic and extrahepatic effects regulating lipid and carbohydrate metabolic pathways, as well as energy homeostasis [56]. On these grounds, XGB may theoretically determine elevated exposure of both cell surface and nuclear receptors to BAs, leading to pathological effects on triglycerides and glucose homeostasis [22, 23, 33,34,35].
In our view, three major conclusions can be drawn from this study. The first relates to the observation that XGB favors lipid accumulation in the liver 24 months after surgery. We obtained these results using validated highly sensitive MRI methods [38, 39] that allowed us to evaluate the statistical significance of small changes in the fat fraction of individual liver segments. In fact, although the actual magnitude of the increase in HFC after XGB was small, it is remarkable that these changes occurred in a relatively short time period and reached statistical significance. This result is consistent with the increased prevalence of NAFLD in patients that have underwent XGB reported in two large retrospective population-based studies in North American [25] and Asian populations [26]. Furthermore, a study based on the US National Health and Nutrition Examination Survey (NHANES) showed that XGB, but not GSD, was associated to cirrhosis and elevated serum liver enzymes [57]. Secondly, in our best knowledge, this is the first prospective study showing that GB ablation associates with increased serum insulin levels and HOMA-IR index in humans. These findings are consistent with a number of retrospective cross-sectional epidemiological analyses showing that cholecystectomized patients have increased risks of MS [27], arteriosclerotic vascular diseases [58] and NAFLD [25, 26]. Our third conclusion is that the observed increase in HFC and HOMA-IR index 24 months after XGB supports the hypothesis that NAFLD and IR could develop after XGD due to the recently discovered metabolic roles of GB [22]. In a previous mouse study, we found that XGB increases serum and hepatic triglyceride concentrations along with higher VLDL and apoB production [28]. Although not dynamic measurements of insulin sensitivity/resistance are available for those experiments, the metabolic changes observed are suggestive of hepatic IR [45, 46]. In addition, the elevated levels of apoB, 24 months after XGB is also suggestive of systemic and hepatic IR, since insulin increases the secretion and decreases the clearance of apoB [59].
Based on our current results and the commented experimental and epidemiological evidence, it is theoretically conceivable that a primary altered GB function, even in the absence of GSD, could induce not only gallstone formation, but also potentiate systemic negative metabolic changes. In fact, diabetic patients and individuals with IR with no evidence of gallstones, have abnormal GB motility [60, 61], possibly changing their BA kinetics and exposing enterohepatic and peripheral tissues to changes in BA metabolic effects.
Conclusions
Our results are consistent with previous retrospective epidemiological surveys showing that XGB, but not GBD appears associated as a risk factor of metabolic syndrome-associated complications, particularly to NAFLD. Although this study was performed in a relatively small number of non-obese individuals, it is plausible that XGB could further increase the risk of IR and associated disease conditions in individuals with elevated basal metabolic risk, including obesity and diabetes. Our findings stress the necessity to prevent GSD and perform longer and larger prospective studies to more precisely disclose the metabolic consequences of GB ablation.
Abbreviations
- ALT:
-
Alanine aminotransferase
- Apo B:
-
Apolipoprotein B
- AST:
-
Aspartate aminotransferase
- BA:
-
Bile acid
- BMI:
-
Body mass index
- FF:
-
Fat fraction
- FGF19:
-
Fibroblast growth factor 19
- GSD:
-
Gallstone disease
- HDL:
-
High density lipoprotein
- HFC:
-
Hepatic fat content
- HOMA-IR:
-
Homeostasis model assessment index
- IDEAL:
-
Iterative decomposition of water and fat with echo asymmetry and least squares estimation
- LDL:
-
Low density lipoprotein
- LLHL:
-
Left lower hepatic lobe
- LMHL:
-
Left medium hepatic lobe
- MRI:
-
Magnetic resonance imaging
- ms:
-
Milliseconds
- NAFLD:
-
Non-alcoholic fatty liver disease
- NASH:
-
Non-alcoholic steatohepatitis
- RAHL:
-
Right anterior hepatic lobe
- ROI:
-
Region of interest
- RPHL:
-
Right posterior hepatic lobe
- VLDL:
-
Very low-density lipoprotein
- XGB:
-
Cholecystectomy
References
Bellentani S. The epidemiology of non-alcoholic fatty liver disease. Liver Int. 2017;37(Suppl 1):81–4. PubMed
Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64(1):73–84. PubMed
Harris R, Harman DJ, Card TR, Aithal GP, Guha IN. Prevalence of clinically significant liver disease within the general population, as defined by non-invasive markers of liver fibrosis: a systematic review. Lancet Gastroenterology Hepatology. 2017;2(4):288–97. PubMed
Saab S, Manne V, Nieto J, Schwimmer JB, Chalasani NP. Nonalcoholic Fatty Liver Disease in Latinos. Clin Gastroenterol Hepatol. 2016;14(1):5–12. quiz e9-0. PubMed
Byrne CD, Targher G. NAFLD: a multisystem disease. J Hepatol. 2015;62(1 Suppl):S47–64. PubMed
Dietrich P, Hellerbrand C. Non-alcoholic fatty liver disease, obesity and the metabolic syndrome. Best Pract Res Clin Gastroenterol. 2014;28(4):637–53. PubMed
Ahmed MH, Ali A. Nonalcoholic fatty liver disease and cholesterol gallstones: which comes first? Scand J Gastroenterol. 2014;49(5):521–7. PubMed
Grundy SM. Metabolic syndrome pandemic. Arterioscler Thromb Vasc Biol. 2008;28(4):629–36. PubMed
Yki-Jarvinen H. Non-alcoholic fatty liver disease as a cause and a consequence of metabolic syndrome. Lancet Diabetes Endocrinol. 2014;2(11):901–10. PubMed
Almeda-Valdes P, Aguilar-Olivos N, Uribe M, Mendez-Sanchez N. Common features of the metabolic syndrome and nonalcoholic fatty liver disease. Rev Recent Clin Trials. 2014;9(3):148–58. PubMed
Duseja A, Singh SP, Saraswat VA, Acharya SK, Chawla YK, Chowdhury S, et al. Non-alcoholic Fatty Liver Disease and Metabolic Syndrome-Position Paper of the Indian National Association for the Study of the Liver, Endocrine Society of India, Indian College of Cardiology and Indian Society of Gastroenterology. J Clinical Experimental Hepatology. 2015;5(1):51–68. PubMed Pubmed Central PMCID: 4415196
Lonardo A, Ballestri S, Marchesini G, Angulo P, Loria P. Nonalcoholic fatty liver disease: a precursor of the metabolic syndrome. Dig Liver Dis. 2015;47(3):181–90. doi:10.1016/j.dld.2014.09.020. PubMed
Larrain S, Rinella ME. A myriad of pathways to NASH. Clin. Liver Dis. 2012;16(3):525–48. PubMed
Goh GB, McCullough AJ. Natural History of Nonalcoholic Fatty Liver Disease. Dig Dis Sci. 2016;61(5):1226–33. PubMed
Italian Association for the Study of the L. AISF position paper on nonalcoholic fatty liver disease (NAFLD): Updates and future directions. Dig Liver Dis. 2017;49(5):471–83. PubMed
Kawano Y, Cohen DE. Mechanisms of hepatic triglyceride accumulation in non-alcoholic fatty liver disease. J Gastroenterol. 2013;48(4):434–41. PubMed Pubmed Central PMCID: 3633701
Birkenfeld AL, Shulman GI. Nonalcoholic fatty liver disease, hepatic insulin resistance, and type 2 diabetes. Hepatology. 2014;59(2):713–23. PubMed Pubmed Central PMCID: 3946772
Jelenik T, Kaul K, Sequaris G, Flogel U, Phielix E, Kotzka J, et al. Mechanisms of Insulin Resistance in Primary and Secondary Non-Alcoholic Fatty Liver. Diabetes. 2017; PubMed
EASL. Clinical Practice Guidelines on the prevention, diagnosis and treatment of gallstones. J Hepatol. 2016;65(1):146–81. PubMed Epub 2016/04/18. eng
Csikesz NG, Singla A, Murphy MM, Tseng JF, Shah SA. Surgeon volume metrics in laparoscopic cholecystectomy. Dig Dis Sci. 2010;55(8):2398–405. PubMed
Russo MW, Wei JT, Thiny MT, Gangarosa LM, Brown A, Ringel Y, et al. Digestive and liver diseases statistics, 2004. Gastroenterology. 2004;126(5):1448–53. PubMed Epub 2004/05/08. eng
Housset C, Chretien Y, Debray D, Chignard N. Functions of the Gallbladder. Comprehensive Physiology. 2016;6(3):1549–77. PubMed Epub 2016/06/28. eng
Zanlungo S, Rigotti A, Miquel JF, Nervi F. Abnormalities of lipid metabolism, gallstone disease and gallbladder function. Clinical Lipidology. 2011;6:315–25.
Nervi F, Arrese M. Cholecystectomy and NAFLD: does gallbladder removal have metabolic consequences? Am J Gastroenterol. 2013;108(6):959–61. PubMed Epub 2013/06/06. eng
Ruhl CE, Everhart JE. Relationship of non-alcoholic fatty liver disease with cholecystectomy in the US population. Am J Gastroenterol. 2013;108(6):952–8. PubMed Epub 2013/04/03. eng
Kwak MS, Kim D, Chung GE, Kim W, Kim YJ, Yoon JH. Cholecystectomy is independently associated with nonalcoholic fatty liver disease in an Asian population. World J Gastroenterol. 2015;21(20):6287–95. PubMed Pubmed Central PMCID: 4445106. Epub 2015/06/03. eng
Shen C, Wu X, Xu C, Yu C, Chen P, Li Y. Association of cholecystectomy with metabolic syndrome in a Chinese population. PLoS One. 2014;9(2):e88189. PubMed Pubmed Central PMCID: 3914934
Amigo L, Husche C, Zanlungo S, Lutjohann D, Arrese M, Miquel JF, et al. Cholecystectomy increases hepatic triglyceride content and very-low-density lipoproteins production in mice. Liver Int. 2011;31(1):52–64. PubMed Epub 2010/11/03. eng
Cortes V, Amigo L, Zanlungo S, Galgani J, Robledo F, Arrese M, et al. Metabolic effects of cholecystectomy: gallbladder ablation increases basal metabolic rate through G-protein coupled bile acid receptor Gpbar1-dependent mechanisms in mice. PLoS One. 2015;10(3):e0118478. PubMed Pubmed Central PMCID: 4349594. Epub 2015/03/05. eng
Juvonen T, Kervinen K, Kairaluoma MI, Kesaniemi YA. Effect of cholecystectomy on plasma lipid and lipoprotein levels. Hepato-Gastroenterology. 1995;42(4):377–82. PubMed Epub 1995/07/01. eng
Sonne DP, Hare KJ, Martens P, Rehfeld JF, Holst JJ, Vilsboll T, et al. Postprandial gut hormone responses and glucose metabolism in cholecystectomized patients. Am J Physiol Gastrointest Liver Physiol. 2013;304(4):G413–9. PubMed Epub 2013/01/01. eng
Ali RB, Cahill RA, Watson RG. Weight gain after laparoscopic cholecystectomy. Ir J Med Sci. 2004;173(1):9–12. PubMed Epub 2005/03/01. eng
Schonewille M, de Boer JF, Groen AK. Bile salts in control of lipid metabolism. Curr Opin Lipidol. 2016;27(3):295–301. PubMed Epub 2016/04/01. eng
Chiang JY. Bile acid metabolism and signaling. Comprehensive Physiology. 2013;3(3):1191–212. PubMed Pubmed Central PMCID: 4422175. Epub 2013/07/31. eng
Arab JP, Karpen SJ, Dawson PA, Arrese M, Trauner M. Bile acids and nonalcoholic fatty liver disease: Molecular insights and therapeutic perspectives. Hepatology. 2017;65(1):350–62. PubMed Pubmed Central PMCID: 5191969
Gariani K, Philippe J, Jornayvaz FR. Non-alcoholic fatty liver disease and insulin resistance: from bench to bedside. Diabetes Metab. 2013;39(1):16–26. PubMed Epub 2012/12/26. eng
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412–9. PubMed Epub 1985/07/01. eng
Idilman IS, Aniktar H, Idilman R, Kabacam G, Savas B, Elhan A, et al. Hepatic steatosis: quantification by proton density fat fraction with MR imaging versus liver biopsy. Radiology. 2013;267(3):767–75. PubMed Epub 2013/02/06. eng
Tang A, Tan J, Sun M, Hamilton G, Bydder M, Wolfson T, et al. Nonalcoholic fatty liver disease: MR imaging of liver proton density fat fraction to assess hepatic steatosis. Radiology. 2013;267(2):422–31. PubMed Pubmed Central PMCID: 3632805. Epub 2013/02/06. eng
Acosta AM, Escalona M, Maiz A, Pollak F, Leighton F. Determination of the insulin resistance index by the Homeostasis Model Assessment in a population of Metropolitan Region in Chile. Revista medica de Chile. 2002;130(11):1227–31. PubMed Epub 2003/02/18. Determinacion del indice de resistencia insulinica mediante HOMA en una poblacion de la region metropolitana de Chile. spa.
Reeder SB, Pineda AR, Wen Z, Shimakawa A, Yu H, Brittain JH, et al. Iterative decomposition of water and fat with echo asymmetry and least-squares estimation (IDEAL): application with fast spin-echo imaging. Magn Reson Med. 2005;54(3):636–44. PubMed Epub 2005/08/11. eng
Glover GH. Multipoint Dixon technique for water and fat proton and susceptibility imaging. J Magn Reson Imaging. 1991;1(5):521–30. PubMed Epub 1991/09/01. eng
Arboleda C, Aguirre-Reyes D, Garcia MP, Tejos C, Munoz L, Miquel JF, et al. Total liver fat quantification using three-dimensional respiratory self-navigated MRI sequence. Magn Reson Med. 2016;76(5):1400–9. PubMed
Fotbolcu H, Zorlu E. Nonalcoholic fatty liver disease as a multi-systemic disease. World J Gastroenterol. 2016;22(16):4079–90. PubMed Pubmed Central PMCID: 4837427. Epub 2016/04/29. eng
Abbasi F, Brown BW Jr, Lamendola C, McLaughlin T, Reaven GM. Relationship between obesity, insulin resistance, and coronary heart disease risk. J Am Coll Cardiol. 2002;40(5):937–43. PubMed Epub 2002/09/13. eng
Patel TP, Rawal K, Bagchi AK, Akolkar G, Bernardes N, Dias Dda S, et al. Insulin resistance: an additional risk factor in the pathogenesis of cardiovascular disease in type 2 diabetes. Heart Fail Rev. 2016;21(1):11–23. PubMed Epub 2015/11/07. eng
Iyengar NM, Hudis CA, Dannenberg AJ. Obesity and cancer: local and systemic mechanisms. Annu Rev Med. 2015;66:297–309. PubMed Epub 2015/01/15. eng
Jahn D, Rau M, Hermanns HM, Geier A. Mechanisms of enterohepatic fibroblast growth factor 15/19 signaling in health and disease. Cytokine Growth Factor Rev. 2015;26(6):625–35. PubMed Epub 2015/08/08. eng
Nies VJ, Sancar G, Liu W, van Zutphen T, Struik D, Yu RT, et al. Fibroblast Growth Factor Signaling in Metabolic Regulation. Front Endocrinol. 2015;6:193. PubMed Pubmed Central PMCID: 4718082. Epub 2016/02/03. eng
Barrera F, Azocar L, Molina H, Schalper KA, Ocares M, Liberona J, et al. Effect of cholecystectomy on bile acid synthesis and circulating levels of fibroblast growth factor 19. Ann Hepatol. 2015;14(5):710–21. PubMed Epub 2015/08/11. eng
Wojcik M, Janus D, Dolezal-Oltarzewska K, Kalicka-Kasperczyk A, Poplawska K, Drozdz D, et al. A decrease in fasting FGF19 levels is associated with the development of non-alcoholic fatty liver disease in obese adolescents. J Pediatr Endocrinol Metab. 2012;25(11-12):1089–93. PubMed Epub 2013/01/19. eng
Eren F, Kurt R, Ermis F, Atug O, Imeryuz N, Yilmaz Y. Preliminary evidence of a reduced serum level of fibroblast growth factor 19 in patients with biopsy-proven nonalcoholic fatty liver disease. Clin Biochem. 2012;45(9):655–8. PubMed Epub 2012/04/03. eng
Shaffer EA, Small DM. Biliary lipid secretion in cholesterol gallstone disease. The effect of cholecystectomy and obesity. J Clin Invest. 59(5):1977, 828–40. PubMed Pubmed Central PMCID: 372291. Epub 1977/05/01. eng
Roda E, Aldini R, Mazzella G, Roda A, Sama C, Festi D, et al. Enterohepatic circulation of bile acids after cholecystectomy. Gut. 1978;19(7):640–9. PubMed Pubmed Central PMCID: 1412071. Epub 1978/07/01. eng
Malagelada JR, Go VL, Summerskill WH, Gamble WS. Bile acid secretion and biliary bile acid composition altered by cholecystectomy. Am J Dig Dis. 1973;18(6):455–9. PubMed Epub 1973/06/01. eng
Arab JP, Karpen SJ, Dawson PA, Arrese M, Trauner M. Bile acids & nonalcoholic fatty liver disease: Molecular insights and therapeutic perspectives. Hepatology. 2017;65(1):350–362.
Ioannou GN. Cholelithiasis, cholecystectomy, and liver disease. Am J Gastroenterol. 2010;105(6):1364–73. PubMed Epub 2010/01/14. eng
Mendez-Sanchez N, Bahena-Aponte J, Chavez-Tapia NC, Motola-Kuba D, Sanchez-Lara K, Ponciano-Radriguez G, et al. Strong association between gallstones and cardiovascular disease. Am J Gastroenterol. 2005;100(4):827–30. PubMed Epub 2005/03/24. eng
Haas ME, Attie AD, Biddinger SB. The regulation of ApoB metabolism by insulin. Trends Endocrinol Metab. 2013;24(8):391–7. PubMed Pubmed Central PMCID: 3810413. Epub 2013/06/01. eng
Arshad F, Laway BA, Rather TA, Kuchay MS, Khan SH. Impaired gallbladder motility in adults with newly detected type 2 diabetes and lack of reversibility after achieving euglycemia. Can J Diabetes. 2015;39(2):101–4. PubMed Epub 2014/12/03. eng
Nakeeb A, Comuzzie AG, Al-Azzawi H, Sonnenberg GE, Kissebah AH, Pitt HA. Insulin resistance causes human gallbladder dysmotility. J Gastrointest Surg. 2006;10(7):940–8. discussion 8-9. PubMed Epub 2006/07/18. eng
Acknowledgements
Authors wish to thank Bogomila Batic and Ludwig Amigo for excellent technical assistance.
Availability of data and material
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Funding
This study was supported by the following grants from the Fondo Nacional Científico y Tecnológico (FONDECYT): 1130146 to FN, 11150329 to N.Q., 1141036 to S.U., 1141134 to VC and 1150327 to MA), and by a grant from the Comisión Nacional de Investigación Científica y Tecnológica (CONICYT) (PIA/Basal PFB12/2007, CARE Chile UC to M.A.), and Conicyt Fondef/IDeA, ID15I10284 to SU.
Author information
Authors and Affiliations
Contributions
VC, MA and FN designed the research, performed the statistical analysis; NQ recruited the patients and collected the data; SU performed NMR images and calculated hepatic fat content; VC, NQ, SU, MA, FN analyzed the data; MA and FN wrote the paper. All authors approved this final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was reviewed and approved by the Pontificia Universidad Católica de Chile Review Board. All study participants provided informed written consent prior to study enrollment.
Consent for Publication
In submitting this article to LIPIDS IN HEALTH AND DISEASE published by BioMed Central I certify that;
1. I am authorized by my co-authors to enter into these arrangements.
2. I warrant, on behalf of myself and my co-authors, that:
○ he article is original, has not been formally published in any other peer-reviewed journal, is not under consideration by any other journal and does not infringe any existing copyright or any other third party rights;
○ I am/we are the sole author(s) of the article and have full authority to enter into this agreement and in granting rights to BioMed Central are not in breach of any other obligation; the article contains nothing that is unlawful, libellous, or which would, if published, constitute a breach of contract or of confidence or of commitment given to secrecy;
I/we have taken due care to ensure the integrity of the article. To my/our - and currently accepted scientific - knowledge all statements contained in it purporting to be facts are true and any formula or instruction contained in the article will not, if followed accurately, cause any injury, illness or damage to the user.
3. I, and all co-authors, agree that the article, if editorially accepted for publication, shall be licensed under the Creative Commons Attribution License 4.0. In line with BioMed Central's Open Data Policy, data included in the article shall be made available under the Creative Commons 1.0 Public Domain Dedication waiver, unless otherwise stated. If the law requires that the article be published in the public domain, I/we will notify BioMed Central at the time of submission, and in such cases not only the data but also the article shall be released under the Creative Commons 1.0 Public Domain Dedication waiver. For the avoidance of doubt it is stated that sections 1 and 2 of this license agreement shall apply and prevail regardless of whether the article is published under Creative Commons Attribution License 4.0 or the Creative Commons 1.0 Public Domain Dedication waiver.
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Cortés, V., Quezada, N., Uribe, S. et al. Effect of cholecystectomy on hepatic fat accumulation and insulin resistance in non-obese Hispanic patients: a pilot study. Lipids Health Dis 16, 129 (2017). https://doi.org/10.1186/s12944-017-0525-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12944-017-0525-3