Abstract
Background
Campylobacter jejuni is a common cause of acute gastroenteritis, but central nervous system infections are rare manifestations of Campylobacter infection. Therefore, C. jejuni trauma-related subdural hygroma infection in children is poorly described in the literature.
Case presentation
We described a 2-year old boy with lobar holoprosencephaly presenting with subdural hygroma following head trauma. C. jejuni infection was confirmed from a subdural hygroma sample by culture as well as by DNA sequencing of a broad range 16S rDNA PCR product. Cerebrospinal fluid from the ventriculoperitoneal shunt remained sterile. Combined neurosurgical and antimicrobial treatment led to complete recovery. Review of the literature showed that the most common manifestation of Campylobacter central nervous system infection is meningitis, mostly in neonates, and subdural hygroma infection was described for only one case.
Conclusions
Subdural hygroma infection caused by C. jejuni is a rare clinical condition in children. Molecular methods represent an important tool for the detection of rare or unexpected pathogens. No standard recommendations for antimicrobial treatment of C. jejuni subdural space infection in children are available, but meropenem treatment combined with surgery seems to be an effective approach.
Similar content being viewed by others
Background
Campylobacter jejuni is a major global gastrointestinal pathogen causing enteritis in all age groups of which children under 5 years are commonly infected [1]. According to the European Centre for Disease Prevention and Control Camplyobacter spp. has been the most commonly reported gastrointestinal bacterial pathogen since 2005 [2]. Data regarding human campylobacteriosis in Croatia are insufficient and notification of the disease remains manly on physicians reporting clinical cases. Published data from Croatian Institute of Public Health shows relatively consistent number of reported cases in the past 10 years, with notification rate below 49.99 cases per 100,000 [2, 3]. Extraintestinal manifestations of C. jejuni infection in children are rare, and include bacteremia, osteomyelitis and meningitis [1, 4,5,6]. Children with brain abnormalities and previous neurosurgical procedures are at risk for central nervous system (CNS) infections and together with neonates represent the only pediatric population with proven C. jejuni CNS infection. Trauma-related subdural hygroma infection has a wide spectrum of clinical manifestations. Symptoms may not be present until weeks after closed head trauma. Since CNS infections are rare manifestations of C. jejuni infection, correct and early detection of causative agent is challenging. Isolation of Campylobacter in blood and cerebrospinal fluid (CSF) samples requires microaerophilic atmosphere and sometimes enrichment, which is not routinely done for blood and CSF culture [7]. Molecular methods represent another diagnostic option, especially if antimicrobial treatment has been started prior to bacterial sampling. The increasing use of molecular methods for detection of Campylobacter is justifiable because of the limitations of the conventional tests. Rapid bacteriological diagnostics is crucial for adequate treatment, especially in CNS infection. The use of 16S rDNA gene sequencing is common for the identification of biochemically unidentified bacteria and unusual strains [8].
We present a rare case of C. jejuni subdural hygroma infection in a 2-year-old boy along with detailed clinical course, diagnostic and therapeutic approach, and review available literature on CNS infections caused by Campylobacter in children.
Case presentation
Our patient was born from a twin pregnancy with a lobar holoprosencephaly diagnosed on fetal ultrasound and confirmed on magnetic resonance imaging (MRI) at 27 weeks of gestation. Ventriculoperitoneal (VP) shunt was placed on the 16th day of life to treat consequent hydrocephalus. The patient suffered several VP shunt infections during the early infancy and the last replacement of the shunt system was done at 7 months of age. Surprisingly, his motoric and cognitive development was unremarkable and without delay. Vaccinations were followed up according to the Croatian National Immunization Program, including 13-valent pneumococcal conjugated vaccine. At two years of age he was admitted to the University Hospital for Infectious Diseases “Dr.Fran Mihaljević”, Zagreb, Croatia with a 10-day history of high fever without other clinical signs nor symptoms. Previous examinations at the emergency department on the 1st and 6th day of fever revealed no pathological findings and showed normal complete blood counts with slightly elevated C-reactive protein (CRP) of 26.5 mg/L and unremarkable result of urine sediment. Of relevance from the case history was a report of a closed head trauma without loss of consciousness after falling off a swing two weeks before the fever onset.
On admission to the hospital, his vital signs were normal besides the fever (40 °C), and no pathological findings were evident on neither physical nor neurological examination. His general clinical condition was good. The white blood cell count was 23.4 × 109/L (58.6% neutrophils), CRP and procalcitonin were 383.6 mg/L and 1.29 μg/L, respectively. The erythrocyte sedimentation rate was 110 mm/h. Coagulation tests showed elevated fibrinogen (9.6 g/L) and D-dimers (1.89 mg/L) while renal and liver tests were unremarkable.
Microbiological diagnosis and treatment
Blood culture sample was drawn into a pediatric bottle from peripheral vein and remained sterile after five days of incubation at 35 ± 1 °C in automated system (BAC/ALERT 3D instrument, BioMerieux, France). One bag urine sample, obtained and cultivated on the blood agar for 24 h at 35 ± 1 °C, remained sterile. Stool sample tested negative for rotavirus and adenovirus antigen. Microbiological analysis also included the common bacterial gastrointestinal pathogens (Campylobacter spp., Salmonella spp., Shigella spp., Yersinia entereocolitica). The stool sample after cultivation on the Campylobacter Blood-Free Selective Agar Base (Oxoid, UK) at 42 ± 1 °C for 48 h in microaerophilic condition, on the Yersinia Selective Agar Base (Oxoid, UK) at 29 ± 1 °C for 48 h and Xylose, lysine, deoxycolate agar (Oxoid, UK) at 35 ± 1 °C for 24 h remained negative on tested pathogens. To rule out infection in the VP shunt, a sample from the valve reservoir was obtained percutaneously. Initial CSF microscopy showed a white cell count of 5/3 cmm (monocyte 100%), the protein level of 0.17 g/L, and glucose concentration of 4.2 mmol/L. A CSF sample remained sterile after four days of incubation at 35 ± 1 °C on the blood agar in a humidified 5 to10% CO2 atmosphere and in the tryptic soy broth and in room air, respectively. All samples were taken prior to initiation of empirical antimicrobial therapy.
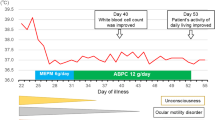
Abdominal ultrasound demonstrated a slightly thickened bladder wall and a small amount of free fluid in the abdomen. Empirical therapy with intravenous ceftriaxone was started. However, despite antimicrobial therapy patient continuously had high fever and on the 3rd hospitalization day parents suspected pain in the child’s left ear. Otoscopy revealed no signs of acute inflammation. A contrast-enhanced brain computer tomography (CT) demonstrated parietooccipital subdural hygroma on the left side with signs of inflammation (Fig. 1a). Antimicrobial treatment was switched to vancomycin and meropenem. The patient was transferred to the Department of Neurosurgery and a subdural external drainage (SED) system was inserted. An additional CSF sample was obtained from the SED system and sent to microbiology laboratory for cultivation and PCR testing. Presence of bacterial DNA was detected in the subdural hygroma sample using broad range PCR targeting 16S rDNA with 16sF557 (5’-GACTCCTACGGGAGGCAG-3’) and 16sR1308 (5’-CGCTCGTTGCGGGACTTAAC-3’) primers. DNA sequencing of a PCR product was performed by ABI PRISM 310 Genetic Analyzer using BigDye v1.1 chemistry (ThermoFisher Scientific, USA). Comparison of obtained sequences to NR/NT database using Nucleotide Basic Local Alignment Search Tool (BLASTn) revealed a match to Campylobacter spp. (the most significant alignment with accession number: CP040607.1) [9]. The colony growth of Campylobacter jejuni was present in the sample of subdural hygroma fluid after 48 h of incubation. Identification was done based on the colony morphology, Gram stain, catalase test, oxidase test and differentiated from Campylobacter coli by positive hippurate hydrolysis test. Following the guidelines of European Committee on Antimicrobial Susceptibility Testing the C. jejuni isolate was found sensitive to erythromycin, gentamycin, and tetracycline, while resistant to ciprofloxacin [10]. The isolate was susceptible to meropenem (MIC 0.023 mg/L) following the recommendation of Clinical and Laboratory Standards Institute; therefore meropenem monotherapy was continued [11]. Despite drainage system insertion and antimicrobial treatment the fever persisted for nine days. Blood samples were taken on the 2nd and 7th day after the drainage system insertion and showed elevated CRP value (169.8 mg/L and 177.9 mg/L, respectively). Brain CT showed partial regression of hygroma with hyperdense areas, more pronounced dorsally and cranially corresponding to the hemorrhagic content (Fig. 1b). On the 22nd day of illness, the patient underwent another neurosurgical procedure. Craniotomy with total subdural hematoma extirpation was performed, VP shunt was removed and an external ventricular drainage (EVD) system was placed (Fig. 2). Meropenem treatment was continued for a total of 4-weeks and a new VP shunt was inserted.
Clinical outcome
Two weeks after the termination of the antimicrobial treatment clinical and laboratory evaluation reported no signs of CNS infection as the CSF samples were unremarkable as well as laboratory findings. Eventually, the child recovered without neurological sequelae.
Literature review
We performed a review of the literature written in English and published between 1980 and July 2020. PubMed database, Web of Science Core Collection and Scopus were searched for the published literature by using the terms: “Campylobacter” OR “Campylobacter jejuni” AND “meningitis” OR “subdural hygroma infection” OR “subdural hematoma infection” AND “children” OR “neonates”. We also searched Google Scholar for other open access resources. We identified and included five articles describing 15 cases of C. jejuni CNS infection in children (Table 1). Fourteen cases of neonatal meningitis are described, three sporadic and 11 in a health-care associated outbreak [5, 12,13,14]. One article described the occurrence of a subdural hygroma infection in 2.5 year old child [15]. The median age of neonates with C. jejuni meningitis was 10.6 days (range 5 to 16 days) [5, 12,13,14]. Only the patient with subdural space infection had underlying brain abnormality; it was a consequence of Sturge-Weber syndrome, with VP shunt inserted to relieve hydrocephalus [15]. Regarding the antimicrobial therapy—the patient with subdural space infection was treated with intravenous chloramphenicol and gentamicin subdural instillation. A subdural-peritoneal shunt was inserted two weeks after the completion of antimicrobial therapy [15]. Patient with meningitis described in 1980 was treated with chloramphenicol and gentamicin for 7 days [12]. Neonates from the nosocomial outbreak were treated with a combination of ampicillin and gentamycin for 7 days, followed by amoxicillin by mouth for 2–3 weeks [5]. Two patients were treated with cefotaxime for 3 weeks [13, 14]. Treatment led to full recovery in all patients except in one who developed moderate dilation of the left ventricle on follow-up [5, 12,13,14,15].
Discussion and conclusion
Infection with Campylobacter spp. most often presents as an acute, self-limited gastrointestinal disease. Extraintestinal manifestations of Campylobacter infection have been described in rare cases, and include meningitis, endocarditis, septic arthritis, osteomyelitis, and neonatal sepsis [7].
Only one case of subdural space infection caused by C. jejuni has been described before—in a child with underlying brain abnormality and VP shunt. The child presented with anorexia, lethargy and vomiting, and C. jejuni was cultivated from subdural fluid sample [15]. Other reported C. jejuni CNS infections were neonatal meningitis [5, 12,13,14]. Clinical presentations were in the majority of patients described as nonspecific, such as convulsions, somnolence and lethargy [5, 14, 15]. Fever was the sole symptom in two neonates [12, 13]. None of the neonates with C. jejuni meningitis had other risk factors. As was described in our study, infection of fluid collection in the subdural space can present as fever of unknown origin (FUO) and produce no neurological symptoms. Prolonged fever with a history of prior head trauma in adults and children should raise suspicion of intracranial infection.
In the reviewed cases, C. jejuni was isolated from the CSF in three patients and in blood culture in four [5, 12, 13, 15]. In one patient C. jejuni was detected in CSF using matrix assisted laser desorption/ionization mass spectrometry with time-of-flight detector (MALDI-TOF MS) [14]. This is the first case report of pediatric CNS Campylobacter infection being diagnosed due to use of 16sDNA sequence analysis. Molecular detection of Campylobacter spp. by 16S rDNA PCR resulted with prompt reduction of empirical antimicrobial therapy and VP shunt removal. This method is rapid and non-specific, and can be especially useful for pathogens that are biochemically inert, fastidious, neglected or slow-growing [8, 16]. Drancourt et al. proposed guidelines for including 16S rDNA sequencing as a reverence method for bacterial identification, especially for slow-growing and fastidious organisms which can remain unidentifiable after the application of all available phenotypic tests [8]. For the genus Campylobacter, 16S DNA sequencing does however lack the sufficient discriminatory power to distinguish C. jejuni and C. coli so the final species identification was performed by hippurate hydrolysis test of the achieved isolate.
World Health Organization does not recommend chloramphenicol treatment for bacterial meningitis as the first-line therapy due to its toxicity and carcinogenic potential in humans [17]. High rates of resistance make cephalosporins poor choice for C. jejuni meningitis [7]. Although C. jejuni is susceptible to gentamicin, its nephrotoxicity and ototoxicity, as well as poor CNS penetration, make it an inappropriate choice for meningitis treatment [18,19,20]. Fluoroquinolones and tetracyclines have demonstrated favorable CNS penetration in adults, however data are limited due to their potential pediatric-specific toxicities [20,21,22,23]. Imipenem and meropenem are the only carbapenems with pediatric data. As imipenem is related to higher seizure risk, meropenem is considered to be a safer option in CNS infection management [20, 24]. Good CNS penetration, low rate of adverse reactions, and good bacterial susceptibility were in favor of meropenem as the best choice in our patient [20]. There are no high-quality evidence guidelines regarding the duration of treatment of bacterial meningitis. Recommended duration for uncomplicated meningitis depends on the causative agent. Three weeks of therapy or a minimum of two weeks beyond the first sterile CSF culture is recommended for gram-negative meningitis, whatever of above is longer [25]. There are only several case reports describing two to three weeks meropenem treatment of C. jejuni meningitis in adults [26,27,28]. We decided for 4-weeks meropenem treatment. Initial treatment failure in our patient could be explained by the size of the hygroma and development of chronic subdural infection. In this setting, the neurosurgical evacuation was necessary for successful treatment.
In conclusion, we report a case of C. jejuni subdural hygroma infection in a patient with holoprosencephaly and VP shunt. Although we didn’t cultivate Campylobacter from stool samples, parents reported regular poultry consumption. Therefore, bacteremia from gastrointestinal tract probably resulted in infection of the posttraumatic subdural hygroma. Congenital brain abnormalities together with the brain injury probably made our patient more prone to C. jejuni CNS invasion. Since C. jejuni meningitis is most commonly diagnosed in neonates, this pathogen is often neglected as a causative agent for CNS infection in older children. Regardless of unspecific clinical presentation, sudden onset of symptoms along with the history of prior closed head trauma, and exclusion of VP shunt meningitis should always raise suspicion of subdural space infection. Early adequate treatment of CNS infections is crucial for satisfying recovery and prompt detection of a pathogen has a significant role in the choice of antimicrobial therapy. Therefore, rapid broad-range molecular methods represent an irreplaceable tool for the detection of rare or unexpected pathogens. Localized subdural infection may require a prolonged antibiotic therapy. In cases of subdural space infection surgical treatment should be considered. In our case, 4-weeks of meropenem treatment combined with surgical evacuation lead to a good clinical outcome.
Availability of data and materials
The datasets generated and/or analysed during the current study are available in the GenBank repository, number OP090654, https://www.ncbi.nlm.nih.gov/nuccore/OP090654.
Abbreviations
- BLASTn:
-
Nucleotide basic local alignment search tool
- CNS:
-
Central nervous system
- CRP:
-
C-reactive protein
- CSF:
-
Cerebrospinal fluid
- CT:
-
Computer tomography
- EVD:
-
External ventricular drainage system
- FUO:
-
Fever of unknown origin
- MALDI-TOF MS:
-
Matrix assisted laser desorption/ionization mass spectrometry with time-of-flight detector
- MIC:
-
Minimal inhibitory concentration
- MRI:
-
Magnetic resonance imaging
- SED:
-
Subdural external drainage system
- VP:
-
Ventriculoperitoneal
References
Bennett JE, Dolin R, Blaser MJ. Mandell, Douglas, And Bennett’s principles and practice of infectious diseases. 9th ed. Philadelphia (PA): Elsevier/Saunders; 2019.
European Centre for Disease Prevention and Control. Campylobacteriosis. In: ECDC. Annual epidemiological report for 2017. Stockholm: ECDC; 2019. https://www.ecdc.europa.eu/en/all-topics-z/campylobacteriosis/surveillance-and-disease-data/annual-epidemiological-reports. Accessed 5 July 2022.
Croatian Institute of Public Health. Croatian Health Statistical Yearbook. https://www.hzjz.hr/sluzba-javno-zdravstvo/hrvatski-zdravstveno-statisticki-ljetopis-za-2020-tablicni-podaci. Accessed 5 July 2022.
Louwen R, Baarlen P, Vliet AHM, Belkum A, Hays JP, Endtz HP. Campylobacter bacteremia: a rare and under-reported event? Eur J Microbiol Immunol. 2012;2:76–87.
Goossens H, Henocque G, Kremp L, Rocque J, Boury R, Alanio G, et al. Nosocomial outbreak of Campylobacter jejuni meningitis in newborn infants. Lancet. 1986;2:146–9.
Vandenberg O, Klein A, Souyah H, Devaster JM, Levy J, Butzler JP. Possible Campylobacter jejuni osteomyelitis in a 4-month-old child. Int J Infect Dis. 2003;7:164–5.
Allos BM. Campylobacter jejuni Infections: update on emerging issues and trends. Clin Infect Dis. 2001;32:1201–6.
Drancourt M, Bollet C, Carlioz A, Martelin R, et al. 16S ribosomal DNA sequence analysis of a large collection of environmental and clinical unidentifiable bacterial isolates. J Clin Microbiol. 2000;38(10):3623–30.
National Center for Biotechnology Information (NCBI). Basic Local Alignment Search Tool (BLAST). https://blast.ncbi.nlm.nih.gov/Blast.cgi. Accessed 20 August 2020.
European Committee on Antimicrobial Susceptibility Testing. Data from the EUCAST MIC distribution 2020. http://www.eucast.org. Accessed 20 August 2020.
Clinical and Laboratory Standards Institute (CLSI) (2020). Performance Standards for Antimicrobial Susceptibility Testing 27th Edition. https://clsi.org/media/1469/m100s27_sample.pdf. Accessed 20 August 2020.
Thomas K, Chan KN, Ribeiro CD. Campylobacter jejuni/coli meningitis in a neonate. Br Med J. 1980;280(6227):1301–2.
Tsoni K, Papadopoulou E, Michailidou E. Campylobacter jejuni meningitis in a neonate: a rare case report. J Neonatal Perinatal Med. 2013;6(2):183–5.
Guerrero García MN, Hernández Magaña R, Rosales García AA, Reyes Hernández KL, Reyes Gómez U, Reyes Hernández D, et al. Meningitis due to Campylobacter jejuni in the neonatal period: case report (in Spanish). Enf Infec Microbiol. 2019;39(4):140–2.
Richie PM, Forbes JC, Steinbok P. Subdural space Campylobacter infection in a child. CMAJ. 1987;131(1):45–6.
Gorkiewicz G, Feierl G, Schober C, Dieber F, Köfer J, Zechner R, et al. Species-specific identification of campylobacter by partial 16S rRNA gene sequencing. J Clin Microbiol. 2003;41(6):2537–46.
World Health Organization (WHO). Report on the comparative effectiveness and safety of chloramphenicol injection. http://archives.who.int/eml/expcom/children/Applications/Chloramphenicol-2.pdf. Accessed 14 August 2020.
Best EJ, Gazarian M, Cohn R, Wilkinson M, Palasanthiran P. Once-daly gentamicin in infants and children: a prospective cohort study evaluating safety and the role therapeutic drug monitoring in minimizing toxicity. Pediatr Infect Dis J. 2011;30(10):827–32.
Martínez-Sagado C, López-Hernández FJ, López-Novoa J. Glomerular nephrotoxcitiy of aminoglycosides. Toxicol Appl Pharmacol. 2007;223(1):86–92.
Sullins AK, Abdel-Rahman SM. Pharmacokinetics of antibacterial agents in the CSF of children and adolescents. Pediatr Drugs. 2013;15(2):93–117.
Brosset A, Nouallie Y, Merle L. Problems raised by the use of tetracyclines in the first year of life. Rev Med Interne. 1987;8(2):218–22.
Stewart DJ. Prevalence of tetracyclines in children’s teeth—study II: a resurvey after five years. Br Med J. 1973;3(5875):320–2.
Sabharwal V, Marchant CD. Fluoroquinolone use in children. Pediatr Infect Dis J. 2006;25(3):257–8.
Miller AD, Ball AM, Bookstaver PB, et al. Epileptogenic potential of carbapenem agents: mechanism of action, seizure rates, and clinical consideration. Pharm. 2011;31(4):408–23.
Feigin RD, McCracken GH Jr, Klein JO. Diagnosis and management of menigitis. Pediatr Infect Dis J. 1992;11(9):785.
Burch KL, Saeed K, Sails AD, Wright PA. Successful treatment by meropenem of Campylobacter jejuni meningitis in a chronic alcoholic following neurosurgery. J Infect. 1999;39(3):241–3.
Kusulja M, Santini M, Margetić K, Gužvinec M, Šopek S, Butić I, et al. Meningitis caused by Campylobacter jejuni: a case presentation and literature review. Acta Clin Belg. 2021;76(4):318–23.
Kogawa S, Furukawa K. Campylobacter jejuni meningitis in an immunocompetent adult male. Rinsho Shinkeigaku. 2010;50(4):262–4.
Acknowledgements
Not applicable.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
IVI, SR, HJ, NK and DD did the literature search, material preparation and data collection. SR, GT and IVI did study conception and design. SR, HJ and GT did data analysis and interpretation and provided guidance for drafting the manuscript. IVI drafted the first manuscript, all authors revised it and commented on the previous versions of the manuscript. IB and MJ did the laboratory diagnostic and pathogen detection. GT concieved the case report and oversaw the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors. The study was approved by the Ethics Committee of the University Hospital for Infectious Diseases “Dr. Fran Mihaljević”, Zagreb (Ethics Committee approval number 01-1931-6-2020). Informed consent was obtained from legal guardians.
Consent for publication
Legal guardians signed informed consent regarding publishing patients data.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Valenčak-Ignjatić, I., Krajcar, N., Didović, D. et al. Campylobacter jejuni subdural hygroma infection in a 2-year old boy: case report and a brief literature review. BMC Infect Dis 22, 700 (2022). https://doi.org/10.1186/s12879-022-07680-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12879-022-07680-0