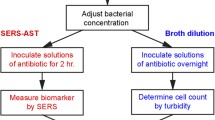
Abstract
Methicillin-resistant Staphylococcus aureus is a bacterium pathogenic to humans and a leading cause of the hospital-acquired infections, causing significant increase in morbidity and mortality. Conventional antibiotic sensitivity testing requires culturing of the isolated pathogen in the presence of antibiotics, and it takes at least 48 hours. Comparatively faster determination of bacterial sensitivity to antibiotics can be achieved with Raman tweezers—an analytical method based on Raman spectroscopy and optical trapping. This article demonstrates the effectiveness of this approach for the discrimination between a methicillin-resistant and a methicillin-sensitive strain of Staphylococcus aureus in about 4 hours from a microliter volume of the bacterial sample. We found that the antibiotic-induced changes in the bacterial cells influenced the ratio of the Raman signals of nucleic acids to phenylalanine. This points to the antibiotic causing cell lysis and the associated loss of nucleic acids from the cytoplasm.





Similar content being viewed by others
References
S. van den Berg, K. P. M. Bonarius, H. P. J.and van Kessel, G. S. Elsinga, N. Kooi, H. Westra, T. Bosma, M.M. van der Kooi-Pol, D. G. A. M. Koedijk, H. Groen, J. M. van Dij, G. Buist, I. A. J. M. Bakker-Woudenberg. Int. J. Med. Microbiol, (2015), 305(11), 55-64
A.J. Kallen, Y. Mu, S. Bulens, A. Reingold, S. Petit, K. Gershman, S.M. Ray, L.H. Harrison, R. Lynfield, G. Dumyati, J.M. Townes, W. Schaffner, P.R. Patel, S.K. Fridkin, JAMA 304(6), 641–648 (2010)
S.E. Cosgrove, Y.L. Qi, K.S. Kaye, S. Harbarth, A.W. Karchmer, Y. Carmeli, Infect. Control. Hosp. Epidemiol 26(2), 166–174 (2005)
D.F.M. Willemse-Erix, M.J. Scholtes-Timmerman, J.-W. Jachtenberg, W.B. van Leeuwen, D. Horst-Kreft, T.C.B. Schut, R.H. Deurenberg, G.J. Puppels, A. van Belkum, M.C. Vos, K. Maquelin, J. Clin. Microbiol. 47(3), 652–659 (2009)
J.H. Jorgensen, J. Clin. Microbiol. 31(11), 2841–2844 (1993)
J. M. Millir, C. M. OHara, Manual of Clinical Microbiology, chapter Manual and automated systems for microbial, (ASM Press,Washington, D.C. 1999)
A. Tannert, R. Grohs, J. Popp, U. Neugebauer, Appl. Microbiol. Biotechnol. 103(2), 549–566 (2019)
J. Kirchhoff, U. Glaser, J.A. Bohnert, M.W. Pletz, J. Popp, U. Neugebauer, Anal. Chem. 90(3), 1811–1818 (2018)
M.G. Bergeron, M. Ouellette, J. Clin. Microbiol. 36(8), 2169–2172 (1998)
M.R. Pulido, M. Garcia-Quintanilla, R. Martin-Pena, J.M. Cisneros, M.J. McConnell, J. Antimicrob. Chemother. 68(12), 2710–2717 (2013)
R.D. Snook, T.J. Harvey, E.C. Faria, P. Gardner, Integr. Biol. 1(1), 43–52 (2009)
H.E. Dekter, C.C. Orelio, M.C. Morsink, S. Tektas, B. Vis, R. te Witt, W.B. van Leeuwen, Eur. J. Clin. Microbiol. Infect. Dis. 36(1), 81–89 (2017)
K. Chen, Y.J. Qin, F. Zheng, M.H. Sun, D.R. Shi, Opt. Lett. 31(13), 2015–2017 (2006)
S. Dochow, Ch. Krafft, U. Neugebauer, T. Bocklitz, T. Henkel, G. Mayer, J. Albert, J. Popp, Lab. Chip. 11(8), 1484–1490 (2011)
A. Ashkin, J. M. Dziedzic, J. E. Bjorkholm, S. Chu, Opt. Lett, (1986), 11(5), 288-290
T.J. Moritz, C.R. Polage, D.S. Taylor, D.M. Krol, S.M. Lane, J.W. Chan, J. Clin. Microbiol. 48(11), 4287–4290 (2010)
Z. Pilát, A. Jonáš, J. Pilátová, T. Klementová, S. Bernatová, M. Šiler, T. Maňka, M. Kizovský, F. Růžička, R. Pantůček, U. Neugebauer, O. Samek, P. Zemánek, Anal. Chem. 92(18), 12304–12311 (2020)
U. Munchberg, P. Rosch, M. Bauer, J. Popp, Anal. Bioanal. Chem. 406(13), 3041–3050 (2014)
S. Raj, M. Marro, M. Wojdyla, D. Petrov, Biomed. Opt. Express 3(4), 753–763 (2012)
K. S. Lee, F. C. Pereira, M. Palatinszky, L. Behrendt, U. Alcolombri, D. Berry, M. Wagner, R. Stocker, Nat. Protoc, (2020), Early Access
K. Maquelin, L.P. Choo-Smith, T. van Vreeswijk, H.P. Endtz, B. Smith, R. Bennett, H.A. Bruining, G.J. Puppels, Anal. Chem. 72(1), 12–19 (2000)
R. Petry, M. Schmitt, J. Popp, ChemPhysChem 4(1), 14–30 (2003)
O. Samek, S. Bernatová, J. Ježek, M. Šiler, M. Šerý, V. Krzyžánek, K. Hrubanová, P. Zemánek, V. Holá, F. Růžička, J. Biomed. Opt. 20(5), 051038 (2015)
J.W. Chan, A.P. Esposito, C.E. Talley, C.W. Hollars, S.M. Lane, T. Huser, Anal. Chem. 76(3), 599–603 (2004)
W.L. Lu, X.Q. Chen, L. Wang, H.F. Li, Y.V. Fu, Anal. Chem. 92(9), 6288–6296 (2020)
C. Xie, J. Mace, M.A. Dinno, Y.Q. Li, W. Tang, R.J. Newton, P.J. Gemperline, PJ. Anal. Chem. 77(14), 4390–4397 (2005)
S. Bernatová, O. Samek, Z. Pilát, M. Šerý, J. Ježek, P. Jákl, M. Šiler, V. Krzyžánek, P. Zemánek, V. Holá, M. Dvořáčková, F. Růžička, Molecules 18(11), 13188–13199 (2013)
Z. Pilát, S. Bernatová, J. Ježek, J. Kirchhoff, A. Tannert, U. Neugebauer, O. Samek, P. Zemánek, Sensors 18(5), 1623 (2018)
K. Rebrošová, M. Šiler, O. Samek, F. Růžička, S. Bernatová, J. Ježek, P. Zemánek, V. Holá, Future Microbiol. 12(10), 881–890 (2017)
I. Notingher, L.L. Hench, Expert Rev. Med. Devices 3(2), 215–234 (2006)
O. Samek, K. Mlynariková, S. Bernatová, J. Ježek, V. Krzyžánek, M. Šiler, P. Zemánek, F. Růžička, V. Holá, M. Mahelová, Int. J. Mol. Sci. 15(12), 23924–23935 (2014)
M.A. Kohanski, D.J. Dwyer, J.J. Collins, Nat. Rev. Microbiol. 8(6), 423–435 (2010)
M.A. Kohanski, D.J. Dwyer, B. Hayete, C.A. Lawrence, J.J. Collins, Cell 130(5), 797–810 (2007)
K.C. Neuman, E.H. Chadd, G.F. Liou, K. Bergman, S.M. Block, Biophys. J. 77(5), 2856–2863 (1999)
N.N. Brandt, O.O. Brovko, A.Y. Chikishev, O.D. Paraschuk, Appl. Spectrosc. 60(3), 288–293 (2006)
N. Schleimer, U. Kaspar, D. Knaack, Ch. von Eiff, S. Molinaro, H. Grallert, E.A. Idelevich, K. Becker, Int. J. Mol. Sci 20(3), 716 (2019)
B.E. Domaracki, A.M. Evans, R.A. Venezia, Antimicrob. Agents. Chemother. 44(5), 1394–1396 (2000)
J. Janardhanan, J.E. Meisel, D. Ding, V.A. Schroeder, W.R. Wolter, S. Mobashery, M. Chang, Antimicrob. Agents. Chemother. 60(9), 5581–5588 (2016)
C. Chuard, J.C. Lucet, P. Rohner, M. Herrmann, R. Auckenthaler, F.A. Waldvogel, D.P. Lew, J. Infect. Dis. 163(6), 1369–1373 (1991)
Funding
The authors acknowledge the support by the Czech Science Foundation (GA19-20697S, GF19-29651L) and European Regional Development Fund (CZ.02.1.01/0.0/0.0/15 003/0000476).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bernatová, S., Rebrošová, K., Pilát, Z. et al. Rapid detection of antibiotic sensitivity of Staphylococcus aureus by Raman tweezers. Eur. Phys. J. Plus 136, 233 (2021). https://doi.org/10.1140/epjp/s13360-021-01152-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1140/epjp/s13360-021-01152-1