Abstract
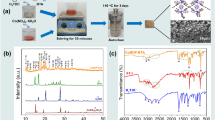
The aim of this paper was to investigate the formation of an amorphous phase in the coating structure due to reduction in porosity and improvement in corrosion resistance of Ti6Al4V alloy during PEO process. The effect of crystallization on the electrochemical corrosion behavior and surface property of Ti6Al4V coating fabricated by plasma electrolytic oxidation (PEO) technique, was investigated in 5 wt %HCl solution. The coating compositions and electrochemical behaviors were examined by X-ray diffraction, Fourier transform infrared spectroscopy (FT-IR), scanning electron microscope (SEM) and electrochemical impedance spectroscopy (EIS) and polarization tests. The results showed that an increase in the ionic strength of electrolyte had a direct effect on the crystal size, the lattice strain and corrosion resistance. An increase in lattice strain due to Ti–O–Si led to a reduction in coating adhesion and corrosion resistance. A significant effect in phase transformation for Titania in the PEO process was not only led to an increase in polarization resistance but also reduced the number and size of porosity.









Similar content being viewed by others
REFERENCES
Zhang, R., Qiao, L., Qu, B., Zhang, S., Chanmg W., and Xiang, J., Mater. Lett., 2015, vol. 153, p. 77.
Rehim, S., Hassan, H., and Amin, M., Corros. Sci., 2004, vol. 46, p. 1921.
Cabrini, M., Lorenzi, S., and Pastore, T., Electrochim. Acta, 2014, vol. 124, p. 156.
Wang, H. and Pilon, L., Electrochim. Acta, 2012, vol. 64, p. 130.
Trdan, U. and Grum, J., Corros. Sci., 2012, vol. 59, p. 324.
Liu, Y., Xia, X., and Liu, H., J. Power Sources, 2004, vol. 130, p. 299.
Zhou, L., Yan, Sh., Tian, B., Zhang, J., and Anpo, M., Mater. Lett., 2006, vol. 60, p. 396.
Shi, X., Wang, Q., Wang, F., and Ge, S., Min. Sci. Technol., 2009, vol. 19, p. 220.
Cimenoglu, H., Gunyuz, M., Kose, G., Baydogan, M., Uğurlu, F., and Sener, C., Mater. Charact., 2011, vol. 62, p. 304.
Wheeler, J., Collier, C., Paillard, J., and Curran, J., Surf. Coat. Technol., 2010, vol. 204, p. 3399.
Teng, H., Yang, C., Lin, J., Huang, Y., and Lu, F., Electrochim. Acta, 2016, vol. 193, p. 216.
Mumjitha, M., and Raj, V., J. Mech. Behav. Biomed. Mater., 2015, vol. 46, p. 205.
Durdu, S. and Usta, M., Ceram. Int., 2014, vol. 40, p. 3627.
Çelik, O., Appl. Surf. Sci., 2013, vol. 274, p. 334.
Arslan, E., Totik, Y., Demirci, E., and Efeoglu, I., Surf. Coat. Technol., 2013, vol. 214, p. 1.
Vangolu, Y., Alsaran, A., and Yildirim, O., Wear, 2011, vol. 271, p. 2322.
Blau, P. and Budinski, K., Wear, 1999, vols. 225–229, p. 1159.
Zhang, P., Zhang, Z., Li, W., and Zhu, M., Appl. Surf. Sci., 2013, vol. 268, p. 381.
Kim, D., Kim, M., Kim, H., Koh, Y., Kim, H., and Jang, J., Acta Biomater., 2009, vol. 5, p. 2196.
Yao, X., Zhang, X., Wu, H., Tian, L., Ma, Y., and Tang, B., Appl. Surf. Sci., 2014, vol. 292, p. 944.
Nie, X., Leyland, A., and Matthews, A., Surf. Coat. Technol., 2000, vol. 125, p. 407.
Mingo, B., Arrabal, R., Mohedano, M., Llamazares, Y., Matykina, E., and Yerokhin, A., Appl. Surf. Sci., 2018, vol. 433, p. 653.
Malinovschi, V., Marin, A., Mihalache, M., and Iosub, I., Surf. Coat. Technol., 2016, vol. 296, p. 96.
Öhman, L., Lövgren, L., Hedlund, T., and Sjöberg, S., Interface Sci. Technol., 2006, vol. 11, p. 1.
Rastogi, P., Sarkar, S., and Mandler, D., Appl. Mater. Today, 2017, vol. 8, p. 44.
Eslamipour, F. and Hejazi, P., J. Mol. Catal. B: Enzym., 2015, vol. 119, p. 1.
Sun, T., Xue, N., Liu, C., Wang, C., and He, J., Appl. Surf. Sci., 2015, vol. 356, p. 599.
Stem, N., Souza, M., Faria, D., and Filho, S., Thin Solid Films, 2014, vol. 558, p. 67.
Sun, Y., Zhang, Z., Liu, L., and Wang, X., J. Non-Cryst. Solids, 2015, vol. 420, p. 26.
Oliveira, C., Gonçalves, L., Almeida, B., Tavares, C., Carvalho, S., and Vaz, F., Surf. Coat. Technol., 2008, vol. 203, p. 490.
Tian, X., Chen, Q., Song, L., Wang, Y., and Li, H., Mater. Lett., 2007, vol. 61, p. 4432.
Jeong, S., Kim, J., Kim, B., Shim, S., and Lee, B., Vacuum, 2004, vol. 76, p. 507.
Wang, S., Guo, X., Xie, Y., Liu, L., Yang, H., and Zhu, R., Surf. Coat. Technol., 2012, vol. 213, p. 192.
Di, S., Guo, Y., Lv, H., Yu, J., and Li, Z., Ceram. Int., 2015, vol. 41, p. 6178.
Venkateswarlu, K., Bose, A., and Rameshbabu, N., Phys. B (Amsterdam, Neth.), 2010, vol. 405, p. 4256.
Malekmohammadi, F., Rouhaghdam, A.S., and Shahrabi, T., J. Non-Cryst. Solids, 2011, vol. 357, p. 1141.
Gawel, L., Nieuzyla, L., Nawrat, G., Darowicki, K., and Slepski, P., J. Alloys Compd., 2017, vol. 722, p. 406.
Zhu, L., Petrova, R.S., Gashinski, J.P., and Yang, Z., Surf. Coat. Technol., 2017, vol. 325, p. 22.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Malekmohammadi, F., Razaghian, A. & Dehghanian, C. The Electrolytic Ionic Strength Effect on Corrosion Resistance and Crystallinity of Plasma Electrolytic Oxidation (PEO) Coating Developed on Ti6Al4V. Prot Met Phys Chem Surf 55, 115–126 (2019). https://doi.org/10.1134/S2070205119010143
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205119010143