Abstract
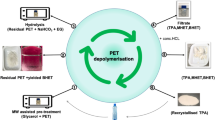
Chemical recycling of polyethylene terephthalate (PET) to produce terephthalic acid (TPA) was studied using in situ hydrolysis with sodium methoxide in methanol and dimethyl sulfoxide (DMSO) as solvent under microwave irradiation. The microwave-assisted reaction was carried out at different temperatures, and reaction time between 5 to 30 min. High degrees of depolymerization were examined at temperature near 70°C at microwave power 300 W. The reaction was carried out in a sealed microwave reactor in which the time and temperature were controlled and recorded. The products were mainly monomers such as TPA and ethylene glycol (EG) which were isolated and purified for further analysis. Monomethyl terephthalate, dimethyl terephthalate, and terephthalic acid were formed initially then converted to TPA as a single monomer product. Purified, TPA was analyzed and identified by NMR, TGA, DSC and FTIR. It was observed that the reaction greatly depends on the amount of sodium methoxide, the volume of methanol and DMSO used, the reaction time, and temperature. Compared to conventional heating methods, the time needed to achieve complete degradation of PET was significantly reduced to 5 min by using microwave irradiation and sodium methoxide catalyst. This has led to substantial saving in energy and cost supporting the conclusion that this proposed recycling process is very beneficial for the recycling of PET wastes.
Similar content being viewed by others
References
Awaja, F. and Pavel, D., Eur. Polym. J., 2005, vol. 41, no. 7, pp. 1453–1477.
Zhang, H. and Wen, Z.-G., Waste Manage., 2014, vol. 34, no. 6, pp. 987–998.
Siddiqui, M.N., Redhwi, H.H., and Achilias, D.S., J. Anal. Appl. Pyrolysis, 2012, vol. 98, pp. 214–220.
Tuna, Ö., Bal, A., and Güçlü, G., Polym. Eng. Sci., 2013, vol. 53, no. 1, pp. 176–182.
Torres, N., Robin, J.J., and Boutevin, B., Eur. Polym. J., 2000, vol. 36, no. 10, pp. 2075–2080.
Paszun, D. and Spychaj, T., Ind. Eng. Chem. Res., 1997, vol. 36, no. 4, pp. 1373–1383.
Karayannidis, G.P. and Achilias, D.S., Macromol. Mater. Eng., 2007, vol. 292, no. 2, pp. 128–146.
Pingale, N.D. and Shukla, S.R., Eur. Polym. J., 2008, vol. 44, no. 12, pp. 4151–4156.
Nikles, D.E. and Farahat, M.S., Macromol. Mater. Eng., 2005, vol. 290, no. 1, pp. 13–30.
Achilias, D.S., Tsintzou, G.P., Nikolaidis, A.K., Bikiaris, D.N., and Karayannidis, G.P., Polym. Int., 2011, vol. 60, no. 3, pp. 500–506.
Siddiqui, M.N., Achilias, D.S., Redhwi, H.H., Bikiaris, D.N., Katsogiannis, K.-A.G., and Karayannidis, G.P., Macromol. Mater. Eng., 2010, vol. 295, no. 6, pp. 575–584.
Grause, G., Handa, T., Kameda, T., Mizoguchi, T., and Yoshioka, T., Chem. Eng. J., 2011, vol. 166, no. 2, pp. 523–528.
Mishra, S., Zope, V.S., and Goje, A.S., J. Appl. Polym. Sci., 2003, vol. 90, no. 12, pp. 3305–3309.
Yang, Y., Lu, Y., Xiang, H., Xu, Y., and Li, Y., Polym. Degrad. Stab., 2002, vol. 75, no. 1, pp. 185–191.
Chen, W. and McCarthy, T.J., Macromolecules, 1998, vol. 31, no. 11, pp. 3648–3655.
Yoshioka, T., Ota, M., and Okuwaki, A., Ind. Eng. Chem. Res., 2003, vol. 42, no. 4, pp. 675–679.
Liu, F., Cui, X., Yu, S., Li, Z., and Ge, X., J. Appl. Polym. Sci., 2009, vol. 114, no. 6, pp. 3561–3565.
Yue, Q.F., Wang, C.X., Zhang, L.N., Ni, Y., and Jin, Y.X., Polym. Degrad. Stab., 2011, vol. 96, no. 4, pp. 399–403.
Wang, H., Liu, Y., Li, Z., Zhang, X., Zhang, S., and Zhang, Y., Eur. Polym. J., 2009, vol. 45, no. 5, pp. 1535–1544.
Genta, M., Iwaya, T., Sasaki, M., and Goto, M., Waste Manage., 2007, vol. 27, no. 9, pp. 1167–1177.
Kurokawa, H. and Ohshima, M.-A., Sugiyama, K., and Miura, H., Polym. Degrad. Stab., 2003, vol. 79, no. 3, pp. 529–533.
Das, J., Halgeri, A.B., Sahu, V., and Parikh, P.A., Indian J. Chem. Technol., 2007, vol. 14, no. 2, pp. 173–177.
Kosmidis, V.A., Achilias, D.S., and Karayannidis, G.P., Macromol. Mater. Eng., 2001, vol. 286, no. 10, pp. 640–647.
Karayannidis, G.P., Chatziavgoustis, A.P., and Achilias, D.S., Adv. Polym. Technol., 2002, vol. 21, no. 4, pp. 250–259.
Oku, A., Hu, L.-C., and Yamada, E., J. Appl. Polym. Sci., 1997, vol. 63, no. 5, pp. 595–601.
Kao, C.-Y., Wan, B.-Z., and Cheng, W.-H., Ind. Eng. Chem. Res., 1998, vol. 37, no. 4, pp. 1228–1234.
Liu, L. and Zhang, D., An, L., Zhang, H., and Tian, Y., J. Appl. Polym. Sci., 2005, vol. 95, no. 3, pp. 719–723.
Yoshioka, T., Motoki, T., and Okuwaki, A., Ind. Eng. Chem. Res., 2001, vol. 40, no. 1, pp. 75–79.
Yoshioka, T., Okayama, N., and Okuwaki, A., Ind. Eng. Chem. Res., 1998, vol. 37, no. 2, pp. 336–340.
Mehrabzadeh, M., Shodjaei, S.T., and Khosravi, M., Iran. Polym. J., 2000, vol. 9, no. 1, pp. 37–40.
López-Fonseca, R., González-Velasco, J.R., and Gutiérrez-Ortiz, J.I., Chem. Eng. J., 2009, vol. 146, no. 2, pp. 287–294.
Paliwal, N.R. and Mungray, A.K., Polym. Degrad. Stab., 2013, vol. 98, no. 10, pp. 2094–2101.
Manju, Roy, P.K., Ramanan, A., and Rajagopal, C., Mater. Lett., 2013, vol. 106, pp. 390–392.
Kržan, A., Polym. Adv. Technol., 1999, vol. 10, no. 10, pp. 603–606.
Nikje, M.M.A. and Nazari, F., Adv. Polym. Technol., 2006, vol. 25, no. 4, pp. 242–246.
Lidström, P., Tierney, J., Wathey, B., and Westman, J., Tetrahedron, 2001, vol. 57, no. 45, pp. 9225–9283.
Fini, A. and Breccia, A., Pure Appl. Chem., 1999, vol. 71, no. 4, pp. 573–579.
Namboori, C.G.G. and Haith, M.S., J. Appl. Polym. Sci., 1968, vol. 12, no. 9, pp. 1999–2005.
Khalaf, H.I. and Hasan, O.A., Chem. Eng. J., 2012, vol. 192, pp. 45–48.
Kimyonok, A.B.E. and Ulutürk, M., J. Energ. Mater., 2016, vol. 34, no. 2, pp. 113–122.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Mohsin, M.A., Alnaqbi, M.A., Busheer, R.M. et al. Sodium Methoxide Catalyzed Depolymerization of Waste Polyethylene Terephthalate Under Microwave Irradiation. Catal. Ind. 10, 41–48 (2018). https://doi.org/10.1134/S2070050418010087
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070050418010087