Abstract
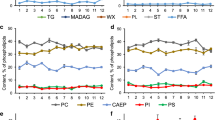
The regularities that shape the composition of low molecular weight organic compounds (LMWOCs) in aquatic macrophytes in response to aquatic environment alterations remain poorly characterized. The aim of the present study consists of a comparative interannual investigation into LMWOC composition in rigid hornwort (Ceratophyllum demersum L.) from a Volga-Akhtuba floodplain lake with a variable trophic state. A high variability of LMWOC composition and individual compound levels in hornwort is detected as different trophic states of the water body are analyzed. Active allelochemicals are the predominant LMWOCs in the case of a “macrophytic” mesotrophic state of the lake, with fatty acids (the free fatty acid fraction) apparently being the most important in this group. Hornwort LMWOC composition in the case of a “cyanobacterial” eutrophic type of lake development is characterized by the predomination of compounds that enhance the protective reactions (manool being the most important) under the conditions of suppression by cyanobacteria, which is also manifested as an almost twofold decrease in the overall intensity of organiccompound biosynthesis.
Similar content being viewed by others
References
Achotegui-Castells, A., Danti, R., Liusià, J., Della Rocca, G., Barberini, S., and Peñuelas, J., Strong induction of minor terpenes in Italian cypress, Cupressus sempervirens, in response to infection by the fungus Seiridium cardinale, J. Chem. Ecol., 2015, vol. 41, pp. 224–243. doi 10.1007/s10886-015-0554-1
Ahel, M., McEvoy, J., and Giger, W., Bioaccumulation of the lipophilic metabolites of nonionic surfactants in fresh-water organisms, Environ. Pollut., 1993, vol. 79, pp. 243–248.
Alekin, O.A., Semenov, A.D., and Skopintsev, B.A., Rukovodstvo po khimicheskomu analizu vod sushi (Manual on Chemical Analysis of Inland Waters), Leningrad: Nauka, 1973.
Anderson, P.M., Hilker, M., Hansson, B.S., Bombosch, S., Klein, B., and Schildknecht, H., Oviposition deterring components in larval frass of Spodoptera littoralis (Boisd.) (Lepidoptera: Noctuidae): a behavioral and electrophysiological evaluation, J. Insect Physiol., 1993, vol. 39, pp. 129–137.
Blackburn, M.A., Kirby, S.J., and Waldock, M.J., Concentrations of alkyphenol polyethoxylates entering UK estuaries, Mar. Pollut. Bull., 1999, vol. 38, no. 2, pp. 109–118.
Carlson, R.E., A trophic state index for lakes, Limnol. Oceanogr., 1977, vol. 22, no. 2, pp. 361–369.
Bul’on, V.V., Pervichnaya produktsiya planktona vnutrennikh vodoemov (Primary Production of Plankton of Inland Reservoirs), Leningrad: Nauka, 1983.
Burakovskii, A.I., Piven’, N.V., and Lukhverchik, L.N., Nonylphenol as a damaging factor in the regulatory systems of the organism, Tr. Beloruss. Gos. Univ., Farmakol. Farm., 2010, vol. 5, no. 1, pp. 243–254.
Bykova, S.N., Borisovskaya, E.V., and Vinogradov, G.A., Influence of some macrophytes and filamentous green alga on succession of microperiphyton communities, Povolzhsk. Ekol. Zh., 2010, no. 3, pp. 241–253.
Czekanowski, J., Coefficient of racial likeness und Durchschnittliche Differenz, Anthropol. Anz., 1922, vol. 9, pp. 227–249.
Demetzos, C. and Dimas, K.S., Labdane-type diterpenes: chemistry and biological activity, Stud. Nat. Prod. Chem., 2001, vol. 25, pp. 235–292.
Dmitriev, V.V., Myakisheva, N.V., Tret’yakov, V.Yu., and Khovanov, N.V., Multicriteria evaluation of ecological status and stability of geological systems based on the method of aggregated indicators, II. Trophic status of aquatic ecosystems, Vestn. S.-Peterb. Univ., Ser. 7: Geol., Geogr., 1997, vol. 1, no. 7, pp. 51–67.
Du, Z.Y. and Benning, C., Triacylglycerol accumulation in photosynthetic cells in plants and algae, in Lipids in Plant and Algae Development, Nakamura, Y. and Li-Beisson, Y., Eds., New York: Springer-Verlag, 2016, pp. 179–205. doi 10.1007/978-3-319-25979-6_8
Dubyna, D.V., Stoiko, S.M., Sytnik, K.M., Tasenkevich, L.A., Shelyag-Sosonko, Yu.R., Geiny, S., Groudova, Z., Gusak, Sh., Otyagelova, G., and Erzhabkova, O., Makrofity—indikatoryizmenenii prirodnoi sredy (Macrophytes as the Indicators of Environmental Changes), Kiev: Naukova Dumka, 1993.
Ekeberg, D., Jablonska, A.M., and Ogner, G., Phytol as a possible indicator of ozone stress by Picea abies, Environ. Pollut., 1995, vol. 89, pp. 55–58.
Ekelund, R., Bergman, A., Granmo, A., and Berggre, M., Bioaccumulation of 4-nonylphenol in marine animals. A re-evaluation, Environ. Pollut., 1990, no. 64, pp. 107–120.
Filippov, O.V., Kochetkova, A.I., Baranova, M.S., and Bryzgalina, E.S., Modern status and problems of water consumption in Volga-Akhtuba floodplain, Grani Poznaniya, 2015, no. 4 (38), pp. 31–41.
Fink, P., Ecological functions of volatile organic compounds in aquatic systems, Mar. Freshwater Behav. Physiol., 2007, vol. 40, no. 3, pp. 155–168.
Fitoplnkton Nizhnei Volgi. Vodokhranilishcha i nizov’e reki (Phytoplankton of Lower Volga. Reservoirs and River Lower Reaches), Trifonov, I.S., Ed., St. Petersburg: Nauka, 2003.
Gening, V.F., Bunyatyan, E.P., Pustovalov, S.Zh., and Rychkov, N.A., Formalizaovanno-statisticheskie metody v arkheologii (analiz pogrebal’nykh pamyatnikov) (Formalized Statistical Methods Applied in Archeology: Analysis of Buried Monuments), Kiev: Naukova Dumka, 1990.
GOST (State Standard) 24027.2-80: Methods for Determination of Moisture, Ash Content, Extractive, and Tannin Materials, Essential Oil, Moscow: Izd. Standartov, 1980.
GOST (State Standard) 17082.4-88: Fruits of Ethereal-Oil Crops. Industrial Raw Material. Methods for Determination of Essential Oil Mass Fraction, Moscow: Izd. Standartov, 1989}, pp. 13–24
Gosudarstvennyi doklad o sostoyanii prirodnykh resursov i okhrane okruzhayushchei sredy Astrakhanskoi oblasti za 2009 god (The State Report of the Natural Resources and Protection of Environment in Astrakhan Oblast in 2009), Astrakhan, 2010.
Gurevich, F.A., Role of phytoncides in inland reservoirs, Vodn. Resur., 1978, no. 2, pp. 133–142.
Hanari, N., Yamamoto, H., and Kuroda, K., Comparison of terpenes in extracts from the resin and the bark of the resinous stem canker of Chamaecyparis obtuse and Thujopsis dolabrata var. hondae, J. Wood. Sci., 2002, vol. 48, no. 1, pp. 56–63.
Hu, H. and Hong, Y., Algal-bloom control by allelopathy of aquatic macrophytes—a review, Front. Environ. Sci. Eng. China, 2008, vol. 2, no. 4, pp. 421–438.
Islam, M.T., De Alencar, M.V.O.B., Da Conceição Machado, K., Da Conceição Machado, K., De Carvalho Melo-Cavalcante, A.A., De Sousa, D.P., and De Freitas, R.M., Phytol in a pharma-medico-stance, Chem.-Biol. Interact., 2015, vol. 240, pp. 60–73.
Jaccard, P., Distribution de la flore alpine dans le bassin des Dranses et dans quelques régions voisines, Bull. Soc. Vaudoise Sci. Nat., 1901, vol. 37, no. 140, pp. 241–272.
Jackson, L.J., Macrophyte-dominated and turbid states of shallow lakes: evidence from Alberta lakes, Ecosystems, 2003, vol. 6, pp. 213–223. doi 10.1007/s10021-002-0001-3
Jüttner, F., Nor-carotenoids as the major volatile excretion products of Cyanidium, Z. Naturforsch., C, 1979, vol. 34, pp. 186–191.
Kim, J.W., Huh, J.E., Kyung, S.H., and Kyung, K.H., Antimicrobial activity of alk(en)yl sulfides found in essential oils of garlic and onion, Food Sci. Biotechnol., 2004, vol. 13, no. 2, pp. 235–239.
Kitaev, S.P., Osnovy limnologii dlya gidrobiologov i ikhtiologov (Basic Limnology for Hydrobiologists and Ichthyologists), Petrozavodsk: Karel. Nauch. Tsentr, Ross. Akad. Nauk, 2007.
Krylova, Yu.V., Kurashov, E.A., and Mitrukova, G.G., Components of volatile low-molecular organic compounds of Ceratophyllu demersum (Ceratophyllaceae) growing in various climatic conditions, Voda: Khim. Ekol., 2016, no. 8, pp. 11–24.
Kurashov, E.A., Krylova, Yu.V., and Mitrukova, G.G., Composition of volatile low-molecular organic substances of Ceratophyllum demersum L. during fruiting, Voda: Khim. Ekol., 2012, no. 6, pp. 107–116.
Kurashov, E.A., Krylova, J.V., Mitrukova, G.G., and Chernova, A.M., Low-molecular-weight metabolites of aquatic macrophytes growing on the territory of Russia and their role in hydroecosystems, Contemp. Probl. Ecol., 2014, vol. 7, no. 4, pp. 433–448.
Kurashov, E.A., Bataeva, Yu.V., Krylova, Yu.V., and Mitrukova, G.G., Biosynthesis of low-molecular organic compounds by aquatic macrophytes and cyanobacteria and their prospective use, in Biotekhnologiya: sostoyanie i perspektivy razvitiya (Biotechnology: Current Status and Prospective Development), Moscow: Ross. Khim.-Tekhnol. Univ. im. D.I. Mendeleeva, 2015, pp. 332–333.
Kurashov, E.A., Fedorova, E.V., Krylova, J.V., and Mitrukova, G.G., Assessment of the potential biological activity of low molecular weight metabolites of freshwater macrophytes with QSAR, Scientifica, 2016, vol. 2016, art. ID 1205680. doi 10.1155/2016/1205680
Lamikanra, O. and Richard, O.A., Effect of storage on some volatile aroma compounds in fresh-cut cantaloupe melon, J. Agric. Food Chem., 2002, vol. 50, pp. 4043–4047.
Lee, L.S. Brooks, L.O., Homer, L.E., Rossetto, M., Henry, R.J., and Baverstock, P.R., Geographic variation in the essential oils and morphology of natural populations of Melaleuca alternifolia (Myrtaceae), Biochem. Syst. Ecol., 2002, vol. 30, no. 4, pp. 343–360.
Liorens, L., Liorens-Molina, J.A., Agnello, S., and Boira, H., Geographical and environment-related variations of essential oils in isolated populations of Thymus richardii Pers. in the Mediterranean basin, Biochem. Syst. Ecol., 2014, vol. 56, pp. 246–254. doi 10.1016/j.bse.2014.05.007
Meteiko, T.Ya., Metabolites of higher aquatic plants and their role in hydrobiocenosises: a review, Gidrobiol. Zh., 1978, vol. 12, no. 4, pp. 3–14.
Metodicheskie rekomendatsii po sboru i obrabotke materialov pri gidrobiologicheskikh issledovaniyakh na presnovodnykh vodoemakh. Fitoplankton i ego produktsiya (Procedure for Collection and Treatment of Materials for Hydrobiological Studies in Freshwater Reservoirs. Phytoplankton and Its Production), Leningrad, 1981.
Milius, A., Concentration of chlorophyll a in phytoplankton of small different-type lakes of Estonia, Izv. Akad. Nauk EstSSR, Biol., 1981, vol. 30, no. 2, pp. 147–157.
Nakai, S., Yamada, S., and Hosomi, M., Anti-cyanobacterial fatty acids released from Myriophyllum spicatum, Hydrobiologia, 2005, vol. 543, pp. 71–78.
Norris, S., Lincoln, K., Abad, M., Scott, M., Eilers, R., Hartsuyker, K., Kindle, K., Hirshberg, J., Karunanandaa, B., Moshiri, F., Stein, J.C., Valentin, H.E., and Venkatesh, T.V., WO Patent 2 004 013 312 A2, 2004.
Novikov, Yu.V., Lastochkina, K.O., and Boldina, Z.N., Metody issledovaniya kachestva vody vodoemov (Manual for Analysis of Water Quality in Reservoirs), Moscow: Meditisna, 1990.
Nürnberg, G.K., Trophic state of clear and colored, softand hard water lakes with special consideration of nutrients, anoxia, phytoplankton and fish, Lake Reservoir Manage., 1996, vol. 12, no. 4, pp. 432–447. doi 10.1080/07438149609354283
Peisker, C., Düggelin, T., Rentsch, D., and Matile, P., Phytol and the breakdown of chlorophyll in senescent leaves, J. Plant Physiol., 1989, vol. 135, pp. 428–432.
Preuss, T.G., Gehrhardt, J., Schirmer, K., Coors, A., Rubach, M., Russ, A., Jones, P.D., Giesy, J.P., and Ratte, H.T., Nonylphenol isomers differ in estrogenic activity, Environ. Sci. Technol., 2006, vol. 40, no. 16, pp. 5147–5153.
Pyrina, I.L., Oxidative analysis of primary production of phytoplankton, in Metodicheskie voprosy izucheniya produktsii planktona vnutrennikh vodoemov (Manual for Analysis of Plankton Production in Inland Reservoirs), St. Petersburg: Gidrometeoizdat, 1993, pp. 10–13.
Rukovodstvo po metodam gidrobiologicheskogo analiza poverkhnostnykh vod i donnykh otlozhenii (Manual for Hydrobiological Analysis of Surface Waters and Bottom Sediments), Leningrad: Gidrometeoizdat, 1983.
Santos, S.A.O., Oliveira, C.S.D., Trindade, S.S., Abreu, M.H., Rocha, S.S.M., and Silvestre, A.J.D., Bioprospecting for lipophilic-like components of five Phaeophyta macroalgae from the Portuguese coast, J. Appl. Phycol., 2016, vol. 28, no. 5, pp. 3151–3158. doi 10.1007/s10811-016-0855-y
Santos, S.A.O., Vilela, C., Freire, C.S.R., Abreu, M.H., Rocha, S.S.M., and Silvestre, A.J.D., Chlorophyta and Rhodophyta macroalgae: a source of health promoting phytochemicals, Food Chem., 2015, vol. 183, pp. 122–128.
Scheffer, M., Alternative attractors of shallow lakes, Sci. World, 2001, vol. 1, pp. 254–263. doi 10.1100/tsw.2001.62
Scheffer, M., Hosper, S.H., Meijer, M.-L., Moss, B., and Jeppesen, E., Alternative equilibria in shallow lakes, Trends Ecol. Evol., 1993, vol. 8, pp. 275–279.
Scheffer, M., Carpenter, S., Foley, J.A., Folke, C., and Walker, B., Catastrophic shifts in ecosystems, Nature, 2001, vol. 413, pp. 591–596.
Sellami, I.H., Rebey, I.B., Sriti, J., Rahali, F.Z., Limam, F., and Marzouk, B., Drying sage (Salvia officinalis L.) plants and its effects on content, chemical composition and radical scavenging activity of the essential oil, Food Bioprocess Technol., 2012, vol. 5, no. 8, pp. 2978–2989.
Sfriso, A., Pavoni, B., Marcomini, A., and Orio, A.A., Macroalgae, nutrient cycles, and pollutants in the Lagoon of Venice, Estuaries, 1992, vol. 15, no. 4, pp. 517–528.
Soares, A., Guieysse, B., Jefferson, B., Cartmell, E., and Lester, J.N., Nonylphenol in the environment: a critical review on occurrence, fate, toxicity and treatment in wastewaters, Environ. Int., 2008, vol. 34, pp. 1033–1049.
Sorensen, T.A., A method of establishing groups of equal amplitude in plant sociology based on similarity of species content, and its application to analyses of the vegetation on Danish commons, K. Dan. Vidensk. Selsk. Biol. Skr., 1948, vol. 5, pp. 1–34.
Souza, A.B., De Souza, M.G.M., Moreira, M.A., Moreira, M.R., Furtado, N.A.J.C., Martins, C.H.G., Bastos, J.K., Dos Santos, R.A., Heleno, V.C.G., Ambrosio, S.R., and Veneziani, R.C.S., Antimicrobial evaluation of diterpenes from Copaifera langsdorffii oleoresin against periodontal anaerobic bacteria, Molecules, 2011, vol. 16, no. 11, pp. 9611–9619.
Srinivasan, R., Devi, K.R., Kannappan, A., Pandian, S.K., and Ravi, A.V., Piper beetle and its bioactive metabolite phytol mitigates quorum sensing mediated virulence factors and biofilm of nosocomial pathogen Serratia marcescens in vitro, J. Ethnopharmacol., 2016, vol. 193, pp. 592–603.
Sun, X., Jin, H., Zhang, L., Hu, W., Li, Y., and Xu, N., Screening and isolation of the algicidal compounds from marine green alga Ulva intestinalis, Chin. J. Ocean. Limnol., 2016, vol. 34, pp. 781–788. doi 10.1007/ s00343-016-4383-z
Tkachev, A.V., Issledovanie letuchikh veshchestv rastenii (Analysis of Volatile Substances of the Plants), Novosibirsk: Ofset, 2008.
Topçu, G. and Gören, A.G., Biological activity of diterpenoids isolated from Anatolian Lamiaceae plants, Rec. Nat. Prod., 2007, vol. 1, no. 1, pp. 1–16.
Ulubelen, A., Topçu, G., Eris, C., Sönmez, U., Kartal, M., Kurucu, S., and Bozok-Johansson, C., Terpenoids from Salvia sclarea, Phytochemistry, 1994, vol. 36, pp. 971–974.
Valentin, H.E., and Qungang, Q., Tocochromanols: biological function and recent advances to engineer plastidial biochemistry for enhanced oil seed vitamin E levels, in The Chloroplast. Advances in Photosynthesis and Respiration, Rebeiz, C.A., Benning, C., Bohnert, H.J., Daniell, H., Hoober, J.K., Lichtenthaler, H.K., Portis, A.R., and Tripathy, B.C., Eds., Dordrecht: Springer-Verlag, 2010, vol 31, pp. 155–170.
Vencl, F.V. and Morton, T.C., The shield defense of the sumac flea beetle, Blepharida rhois (Chrysomelidae: Alticinae), Chemoecology, 1998, vol. 8, pp. 25–32.
Vinberg, G.G., Pervichnaya produktsiya vodoemov (Primary Production of Reservoirs), Minsk, 1960.
Watson, S.B., Cyanobacterial and eukaryotic algal odor compounds: signals or by-products? A review of their biological activity, Phycologia, 2003, vol. 42, no. 4, pp. 332–350.
Watson, S.B., Caldwell, G., and Pohnert, G., Fatty acids and oxylipins as semiochemicals, in Lipids in Aquatic Ecosystems, Dordrecht: Springer-Verlag, 2009, pp. 65–91.
Zhang, K., Lin, T.F., Zhang, T., Li, C., and Gao, N., Characterization of typical taste and odor compounds formed by Microcystis aeruginosa, J. Environ. Sci., 2013a, vol. 25, no. 8, pp. 1539–1548.
Zhang, H., Mallik, A., and Zeng, R.S., Control of Panama disease of banana by rotating and intercropping with Chinese chive (Allium tuberosum Rottler): role of plant volatiles, J. Chem. Ecol., 2013b, vol. 39, no. 2, pp. 243–252. doi 10.1007/s10886-013-0243-x
Zuo, S., Zhou, S., Ye, L., Ding, Y., and Jiang, X., Antialgal effects of five individual allelochemicals and their mixtures in low level pollution conditions, Environ. Sci. Pollut. Res., 2016, vol. 23, pp. 15703–15711. doi 10.1007/s11356-016-6770-6
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.A. Kurashov, G.G. Mitrukova, J.V. Krylova, 2018, published in Sibirskii Ekologicheskii Zhurnal, 2018, No. 2, pp. 207–224.
Rights and permissions
About this article
Cite this article
Kurashov, E.A., Mitrukova, G.G. & Krylova, J.V. Interannual Variability of Low-Molecular Metabolite Composition in Ceratophyllum demersum (Ceratophyllaceae) from a Floodplain Lake with a Changeable Trophic Status. Contemp. Probl. Ecol. 11, 179–194 (2018). https://doi.org/10.1134/S1995425518020063
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1995425518020063